Riverine and Marine Ecotypes of Sotalia Dolphins Are Different Species
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Inia Araguaiaensisand Inia Geoffrensis
Interactions between children, teenagers and botos (Inia araguaiaensis and Inia geoffrensis) in markets and fairs of Eastern Amazon Angélica Lúcia Figueiredo Rodrigues, Gabriel Melo-Santos, Iara Ramos-Santos, Ana Marta Andrade, Danilo Leal Arcoverde, Leonardo Sena, Maria Luisa da Silva Date of deposit 09 05 2019 Document version Author’s accepted manuscript Access rights © 2019, Elsevier Ltd. All rights reserved. This work is made available online in accordance with the publisher’s policies. This is the author created, accepted version manuscript following peer review and may differ slightly from the final published version. Citation for Figueiredo Rodrigues, A.L., Melos-Santos, G., and Ramos-Santos, published version I. (2019). Interactions between children, teenagers and botos (Inia araguaiaensis and Inia geoffrensis) in markets and fairs of Eastern Amazon. Ocean and Coastal Management, 172: pp137- 145. Link to published https://doi.org/j.ocecoaman.2019.02.005 version Full metadata for this item is available in St Andrews Research Repository at: https://research-repository.st-andrews.ac.uk/ 1 Abstract In this study we present the first record of interactions (ludic behavior, swimming and induced feeding) involving river dolphins known as botos (Inia sp.) and children/adolescents (from eight to 17 years old) at markets and fairs at the river front of two localities of Pará State, Brazil. We registered the reports of the participants using semi-structured interviews while they were working at the markets or when they were in the water to interact with river dolphins. We registered dolphins and human behavior using the ad libitum method. Most of the children interacting with botos were boys - we observed only two girls swimming with the dolphins. -

Living Between Rapids: Genetic Structure and Hybridization in Botos (Cetacea: Iniidae: Inia Spp.) of the Madeira River, Brazil
bs_bs_banner Biological Journal of the Linnean Society, 2015, 114, 764–777. With 5 figures Living between rapids: genetic structure and hybridization in botos (Cetacea: Iniidae: Inia spp.) of the Madeira River, Brazil WALESKA GRAVENA1,2*, VERA M. F. DA SILVA2, MARIA N. F. DA SILVA3, IZENI P. FARIAS1 and TOMAS HRBEK1 1Laboratório de Evolução e Genética Animal (LEGAL), Departamento de Genética, Universidade Federal do Amazonas, Av. General Rodrigo Otávio Jordão, 3000, 69077-000, Manaus, Amazonas, Brazil 2Laboratório de Mamíferos Aquáticos, Instituto Nacional de Pesquisas da Amazônia, Av. André Araújo, 2936, 69011-970, Manaus, Amazonas, Brazil 3Coleção de Mamíferos, Instituto Nacional de Pesquisas da Amazônia, Av. André Araújo, 2936, 69011-970, Manaus, Amazonas, Brazil Received 2 September 2014; revised 14 November 2014; accepted for publication 15 November 2014 Until the recent construction of hydroelectric dams, a series of 18 rapids divided the upper and lower Madeira River, and these rapids were thought to separate two species of Amazonian freshwater dolphins (boto): Inia boliviensis (above) and I. geoffrensis (below). Some reports and articles, however, mention the occurrence of botos within the rapids region and that they occasionally cross the rapids, but without mentioning the species concerned. Based on our previous studies, it is likely that I. boliviensis occurs in the region of the rapids. To test this supposition, we sampled 18 individuals from this region, and collected mitochondrial (control region, cytochrome b and cytochrome oxidase I) and nuclear (10 microsatellite loci) DNA data, in order to test if there is connectivity between the dolphins that were found within the rapids region and dolphins collected upstream and downstream of the rapids, and investigate population structuring between these localities. -

Encyclopedia of Marine Mammals, Second Edition
1188 Tucuxi and Guiana Dolphin continues to grow and in the United States, public support stands Chance , P. ( 1994 ). “ Learning and Behavior , ” 3rd Ed. Brooks/Cole fi rmly behind both the MMPA and marine mammal facilities. More Publishing Company , Belmont . people are now enjoying the benefi ts of new and exciting training Cole , K. C. , Van Tilburg , D. , BurchVernon , A. , and Riccio , D. C. ( 1996). programs, shows, presentations, interaction opportunities, and scien- The importance of context in the US preexposure effect in CTA: Novel tifi c discoveries, all facilitated through behavior management. versus latently inhibited contextual stimuli . Lear. Motiv. 27 , 362 – 374 . Domjan , M. ( 1993 ). “ The Principles of Learning and Behavior , ” 3rd Ed. By maintaining a healthy captive population of various marine Brooks/Cole Publishing Company , Belmont . mammal species, comparative data are generated to assist in under- Honig , W. K. , and Staddon , J. E. R. ( 1977 ). “ The Handbook of Operant standing wild animals, and these facilities continue to give material Behavior . ” Prentice-Hall, Inc , Englewood Cliffs . support to important research and conservation initiatives. In addi- Kazdin , A. E. ( 1994 ). “ Behavior Modifi cation in Applied Settings , ” 5th tion, these facilities act as part of the Marine Mammal Stranding Ed. Brooks/Cole Publishing Company , Belmont . Network, assisting NOAA/NMFS in the rescue, housing, and care Marine Mammal Permits and Authorizations. (2006). [Accessed online of stranded wild animals where expertise in medical care can be July 5, 2007]. Available from World Wide Web: http://www.nmfs. applied. These facilities also develop animal management and hus- noaa.gov/pr/permits/mmpa_permits.htm bandry skills in staff members who are also able to assist in health Marine Mammal Poll. -

Food Consumption and Body Measurements of Amazon River Dolphins (Inia Geoffrensis)
Aquatic Mammals 1999, 25.3, 173–182 Food consumption and body measurements of Amazon river dolphins (Inia geoffrensis) R. A. Kastelein1, B. Neurohr2, S. H. Nieuwstraten1 and P. R. Wiepkema3 1Harderwijk Marine Mammal Park, Strandboulevard Oost 1, 3841 AB Harderwijk, The Netherlands 2Tiergarten Nu¨rnberg, Am Tiergarten 30, D90480 Nu¨rnberg, Germany 3Emeritus Professor of Ethology, Wageningen Agricultural University, Stationsweg 1, 6861 EA Oosterbeek, The Netherlands Abstract dolphin is the most widespread freshwater dolphin in the world, its distribution is limited compared to This report is on the food consumption of 3 male that of most marine odontocetes and it is therefore Amazon river dolphins which were housed in water very likely to become a threatened species. For the of between 27 and 29 C at Duisburg Zoo, management of Amazon river dolphins in the wild, Germany. The food consumption of 2 animals was information is needed about the population size, recorded for 17 successive complete calendar years, age composition and sex ratio, seasonal distribu- that of the third animal for 3 complete successive tion, diet, energy requirements relative to seasonal years. In male 002 the total annual food intake prey distribution and density, and about competi- increased to 1280 kg at the age of 10 years, after tion with other animals and with fisheries. This which it decreased slightly and stabilised at around information could facilitate prey management 1170 kg/year. Male 001 was adult on arrival at the to allow for a certain number of Amazon river zoo. His annual food intake fluctuated at around dolphins in their distribution area. -

Taxonomic Status of the Genus Sotalia: Species Level Ranking for “Tucuxi” (Sotalia Fluviatilis) and “Costero” (Sotalia Guianensis) Dolphins
MARINE MAMMAL SCIENCE, **(*): ***–*** (*** 2007) C 2007 by the Society for Marine Mammalogy DOI: 10.1111/j.1748-7692.2007.00110.x TAXONOMIC STATUS OF THE GENUS SOTALIA: SPECIES LEVEL RANKING FOR “TUCUXI” (SOTALIA FLUVIATILIS) AND “COSTERO” (SOTALIA GUIANENSIS) DOLPHINS S. CABALLERO Laboratory of Molecular Ecology and Evolution, School of Biological Sciences, University of Auckland, Private Bag 92019, Auckland, New Zealand and Fundacion´ Omacha, Diagonal 86A #30–38, Bogota,´ Colombia F. TRUJILLO Fundacion´ Omacha, Diagonal 86A #30–38, Bogota,´ Colombia J. A. VIANNA Sala L3–244, Departamento de Biologia Geral, ICB, Universidad Federal de Minas Gerais, Avenida Antonio Carlos, 6627 C. P. 486, 31270–010 Belo Horizonte, Brazil and Escuela de Medicina Veterinaria, Facultad de Ecologia y Recursos Naturales, Universidad Andres Bello Republica 252, Santigo, Chile H. BARRIOS-GARRIDO Laboratorio de Sistematica´ de Invertebrados Acuaticos´ (LASIA), Postgrado en Ciencias Biologicas,´ Facultad Experimental de Ciencias,Universidad del Zulia, Avenida Universidad con prolongacion´ Avenida 5 de Julio, Sector Grano de Oro, Maracaibo, Venezuela M. G. MONTIEL Laboratorio de Ecologıa´ y Genetica´ de Poblaciones, Centro de Ecologıa,´ Instituto Venezolano de Investigaciones Cientıficas´ (IVIC), San Antonio de los Altos, Carretera Panamericana km 11, Altos de Pipe, Estado Miranda, Venezuela S. BELTRAN´ -PEDREROS Laboratorio de Zoologia,´ Colec¸ao˜ Zoologica´ Paulo Burheim, Centro Universitario´ Luterano de Manaus, Manaus, Brazil 1 2 MARINE MAMMAL SCIENCE, VOL. **, NO. **, 2007 M. MARMONTEL Sociedade Civil Mamiraua,´ Rua Augusto Correa No.1 Campus do Guama,´ Setor Professional, Guama,´ C. P. 8600, 66075–110 Belem,´ Brazil M. C. SANTOS Projeto Atlantis/Instituto de Biologia da Conservac¸ao,˜ Laboratorio´ de Biologia da Conservac¸ao˜ de Cetaceos,´ Departamento de Zoologia, Universidade Estadual Paulista (UNESP), Campus Rio Claro, Sao˜ Paulo, Brazil M. -

Morphometrics of the Dolphin Genus Lagenorhynchus: Deciphering A
Morphometrics of the dolphin genus Lagenorhynchus: deciphering a contested phylogeny Allison Galezo1,2 and Nicole Vollmer1,3 1 Department of Vertebrate Zoology, Smithsonian Institution National Museum of Natural History 2 Department of Biology, Georgetown University 3 NOAA National Systematics Laboratory Background Results & Analysis Discussion • Our morphological data support the hypothesis that Figure 1. Phenogram from cluster analysis of dolphin skull Figure 2. Species symbols key with sample sizes. measurements. Calculated using Euclidian distances and the genus Lagenorhynchus is not monophyletic, evident Height l La. acutus (24) p C. commersonii (5) a b c Ward’s method. from the separation in the phenogram of La. albirostris Height p C. eutropia (2) Recent phylogenetic studies1-7 have indicated that the Distance l La. albirostris (10) and La. acutus from the other Lagenorhynchus species, 0 2 4 6 8 p genus Lagenorhynchus, currently containing the species 0 2 4 6 8 l La. australis (7) C. heavisidii (1) u Li. borealis (11) and the mix of genera in the lowermost clade (Figure 1). L. obliquidensa, L. acutusb , L. albirostrisc, L. obscurusd , L. l La. obliquidens (28) p C. hectori (2) n Unknown (1) • Our results show that La. obscurus and La. obliquidens e f cruciger , and L. australis , is not monophyletic. These C. commersonii l La. obscurus (15) are very similar morphologically, which supports the C. commersonii species were originally grouped together because of C.C. cocommemmersoniirsonii C. commeC. hersoniictori hypothesis that they are closely related: they have C. commeC. hersoniictori similarities in external morphology and coloration, but C. commeC. hersoniictori Figure 3. -
Global Patterns in Marine Mammal Distributions
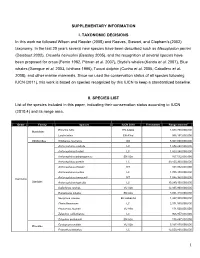
SUPPLEMENTARY INFORMATION I. TAXONOMIC DECISIONS In this work we followed Wilson and Reeder (2005) and Reeves, Stewart, and Clapham’s (2002) taxonomy. In the last 20 years several new species have been described such as Mesoplodon perrini (Dalebout 2002), Orcaella heinsohni (Beasley 2005), and the recognition of several species have been proposed for orcas (Perrin 1982, Pitman et al. 2007), Bryde's whales (Kanda et al. 2007), Blue whales (Garrigue et al. 2003, Ichihara 1996), Tucuxi dolphin (Cunha et al. 2005, Caballero et al. 2008), and other marine mammals. Since we used the conservation status of all species following IUCN (2011), this work is based on species recognized by this IUCN to keep a standardized baseline. II. SPECIES LIST List of the species included in this paper, indicating their conservation status according to IUCN (2010.4) and its range area. Order Family Species IUCN 2010 Freshwater Range area km2 Enhydra lutris EN A2abe 1,084,750,000,000 Mustelidae Lontra felina EN A3cd 996,197,000,000 Odobenidae Odobenus rosmarus DD 5,367,060,000,000 Arctocephalus australis LC 1,674,290,000,000 Arctocephalus forsteri LC 1,823,240,000,000 Arctocephalus galapagoensis EN A2a 167,512,000,000 Arctocephalus gazella LC 39,155,300,000,000 Arctocephalus philippii NT 163,932,000,000 Arctocephalus pusillus LC 1,705,430,000,000 Arctocephalus townsendi NT 1,045,950,000,000 Carnivora Otariidae Arctocephalus tropicalis LC 39,249,100,000,000 Callorhinus ursinus VU A2b 12,935,900,000,000 Eumetopias jubatus EN A2a 3,051,310,000,000 Neophoca cinerea -

SHORT-FINNED PILOT WHALE (Globicephala Macrorhynchus): Western North Atlantic Stock
February 2019 SHORT-FINNED PILOT WHALE (Globicephala macrorhynchus): Western North Atlantic Stock STOCK DEFINITION AND GEOGRAPHIC RANGE There are two species of pilot whales in the western North Atlantic - the long-finned pilot whale, Globicephala melas melas, and the short-finned pilot whale, G. macrorhynchus. These species are difficult to differentiate at sea and cannot be reliably visually identified during either abundance surveys or observations of fishery mortality without high-quality photographs (Rone and Pace 2012); therefore, the ability to separately assess the two species in U.S. Atlantic waters is complex and requires additional information on seasonal spatial distribution. Pilot whales (Globicephala sp.) in the western North Atlantic occur primarily along the continental shelf break from Florida to the Nova Scotia Shelf (Mullin and Fulling 2003). Long-finned and short- finned pilot whales overlap spatially along the mid-Atlantic shelf break between Delaware and the southern flank of Georges Bank (Payne and Heinemann 1993; Rone and Pace 2012). Long-finned pilot whales have occasionally been observed stranded as far south as South Carolina, and short- finned pilot whales have occasionally been observed stranded as far north as Massachusetts (Pugliares et al. 2016). The exact latitudinal ranges of the two species remain uncertain. However, south of Cape Hatteras most pilot whale sightings are expected to be short- Figure 1. Distribution of long-finned (open symbols), short-finned finned pilot whales, while north of (black symbols), and possibly mixed (gray symbols; could be ~42°N most pilot whale sightings are either species) pilot whale sightings from NEFSC and SEFSC expected to be long-finned pilot whales shipboard and aerial surveys during the summers of 1998, 1999, (Figure 1; Garrison and Rosel 2017). -

Cephalorhynchus Hectori) in Porpoise Bay, New Zealand
NewBejder Zealand & Dawson—Hector’s Journal of Marine dolphins and Freshwater in Porpoise Research, Bay 2001, Vol. 35: 277–287 277 0028–8330/01/3502–0277 $7.00 © The Royal Society of New Zealand 2001 Abundance, residency, and habitat utilisation of Hector’s dolphins (Cephalorhynchus hectori) in Porpoise Bay, New Zealand LARS BEJDER INTRODUCTION Department of Biology Hector’s dolphins (Cephalorhynchus hectori van Dalhousie University Beneden 1881) have been studied on a large scale Halifax, Nova Scotia, B3H 4J1 (c. 1000 km) throughout their geographic distribu- Canada tion (Dawson & Slooten 1988; Bräger 1998). On an STEVE DAWSON† intermediate scale (c. 90 km), one regional popula- Department of Marine Science tion (around Banks Peninsula) has been studied in- University of Otago tensively for more than a decade (Slooten & Dawson P. O. Box 56, Dunedin 1994). Little is known about small populations, how- New Zealand ever, or about habitat utilisation at fine scales (<5km). email: [email protected] Hector’s dolphins are restricted to New Zealand, and have a strictly coastal distribution. Despite wide- ranging survey effort, there is no evidence of alongshore movement of more than a few tens of Abstract Theodolite tracking and boat-based kilometres (Slooten et al. 1993; Bräger 1998). Cur- photo-identification surveys were carried out in the rent distribution is highly localised and fragmented austral summers of 1995/96 and 1996/97 to assess into genetically distinct populations (Pichler et al. abundance, residency, and habitat utilisation of 1998). The species is listed by the International Hector’s dolphins (Cephalorhynchus hectori van Union for the Conservation of Nature as “Endan- Beneden 1881) in Porpoise Bay, on the south-east gered” (2000 IUCN red list of threatened species. -

Feeding Strategies of the Guiana Dolphin Sotalia Guianensis Marcos R
70 The Open Marine Biology Journal, 2009, 3, 70-76 Open Access Feeding Strategies of the Guiana Dolphin Sotalia guianensis Marcos R. Rossi-Santos*,1,§ and Paulo A.C. Flores2 1Centro de Ciências Biológicas, Universidade Federal de Santa Catarina and International Wildlife Coalition, Brasil 2Centro Nacional de Pesquisa, Conservação e Manejo de Mamíferos Aquáticos – CMA, ICMBio CMA SC – Rod. Maurício Sirotsky Sobrinho, s/n, km02, Jurerê, Florianópolis, SC, 88053-700, Brasil Abstract: The aim of this study was to characterize the feeding behavior of the Guiana dolphin, S. guianensis, through description and quantification of the strategies used by the dolphins, and possibly to apply this patterns to other areas in its distribution. Data were collected during systematic boat surveys, between November/1996 and May/1997, with 120 hours and 13 minutes of total observations. Scheffé Test was utilized to determine whether some strategies were used more than others. Six basic strategies of dolphin feeding behavior were identified: Individual Random Feeding (IRF), Group Random Feeding (GRF), Circular Cooperative Feeding (CCF), Front Cooperative feeding (FCF), Crossing Cooperative Feeding (CRF) and Zig-zag Cooperative Feeding (ZCF). The strategy GRF was statistically different from the others (Scheffé 5%). Previous studies suggested that S. guianensis exhibit two basic feeding strategies, while here we show greater diversity on the feeding behavior, with more complex and varied foraging strategies than previously reported for this species. Observed variation in the coordination of individuals, group cohesion, movement patterns, prey availability and environmental features, demonstrates the complexity of the S. guianensis feeding behavior. Keywords: Feeding behavior, Strategies, Guiana dolphin, Sotalia guianensis. -

Distribution and Movement Patterns of Killer Whales (Orcinus Orca) in the Northwest Atlantic. ICES CM 2008/B:20
ICES CM 2008/B:20 Distribution and Movement Patterns of Killer Whales (Orcinus orca) in the Northwest Atlantic Tara S. Stevens1,2 and Jack W. Lawson1 (1) Fisheries and Oceans Canada, NAFC, 80 East White Hills Road, St. John’s, Newfoundland and Labrador A1C 5X1, Canada (2) Memorial University of Newfoundland, Cognitive and Behavioural Ecology Program, St. John’s, Newfoundland and Labrador A1B 3X7, Canada Corresponding author e-mail: [email protected] ABSTRACT: Killer whales (Orcinus orca) occur throughout the northwest Atlantic. A sightings database and photographic catalogue, created mostly from opportunistic sources, is used to examine the occurrence of killer whales in Atlantic Canada. A majority of the sightings are from the Newfoundland and Labrador Region despite comparable observer coverage in adjacent areas such as the Gulf of St. Lawrence and Scotian Shelf, which suggests greater abundance in and habitat preference for Newfoundland and Labrador waters. Killer whales occur in all months of the year and in both near- and offshore regions, although particular sighting patterns may represent local observer effort and awareness. The distribution, movement, and residency patterns of killer whales may be closely linked to that of their prey; they have been observed harassing, attacking, and eating marine mammals, including minke whales (Balaenoptera acuterostrata), dolphins, and seals, and potentially eating fish. Some killer whales appear to remain year round in the Newfoundland and Labrador area and have been sighted during the spring within pack ice, potentially in association with breeding harp seals (Phoca groenlandica). Based on photographic records, individual killer whales in this area have been shown to move hundreds of kilometers within a year. -

Lagenorhynchus Obscurus – Dusky Dolphin
Lagenorhynchus obscurus – Dusky Dolphin to be split in due course (LeDuc et al. 1999; Harlin- Cognato 2010). Several recent phylogenies have shown that L. obscurus consistently groups with L. obliquidens (Pacific White-sided Dolphin) and separately to L. australis (Peale’s Dolphin) and L. cruciger (Hourglass Dolphin). The two North Atlantic members of the genus (L. albirostris, the White-beaked Dolphin, and L. acutus, the Atlantic White-sided Dolphin) appear distinct from all other members of the genus. Current thinking would either split L. obscurus and L obliquidens into the genus Sagmatius (LeDuc et al. 1999; May-Collado & Agnarsson 2006) or make it an entirely new genus (Harlin-Cognato 2010). Sara Golaski / Namibian Dolphin Project Three subspecies of Dusky Dolphin are generally suggested based on both molecular (Harlin-Cognato et al. Regional Red List status (2016) Least Concern*† 2007) and morphological (Van Waerebeek 1993a, 1993b) differences: L. o. obscurus in southern Africa, L. o. fitzroyi National Red List status (2004) Data Deficient in southern South America, and an un-named subspecies Reasons for change Non-genuine change: in New Zealand (Perrin 2002). Subspecies classification New information awaits formal description (Cipriano & Webber 2010). Global Red List status (2008) Data Deficient TOPS listing (NEMBA) (2007) None Assessment Rationale Large schools and frequent sightings of Dusky Dolphins in CITES listing (2003) Appendix II both the northern (Namibia) and southern (South Africa) Endemic No Benguela suggest high abundance and more than 10,000 mature individuals. No major threats were identified, *Watch-list Data †Watch-List Threat although an emerging threat of mid-water trawling requires monitoring.