Coronavirus Disease 2019 in Pregnancy
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
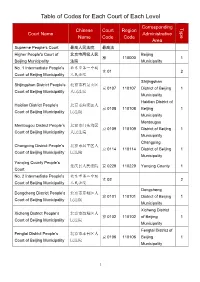
Table of Codes for Each Court of Each Level
Table of Codes for Each Court of Each Level Corresponding Type Chinese Court Region Court Name Administrative Name Code Code Area Supreme People’s Court 最高人民法院 最高法 Higher People's Court of 北京市高级人民 Beijing 京 110000 1 Beijing Municipality 法院 Municipality No. 1 Intermediate People's 北京市第一中级 京 01 2 Court of Beijing Municipality 人民法院 Shijingshan Shijingshan District People’s 北京市石景山区 京 0107 110107 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Haidian District of Haidian District People’s 北京市海淀区人 京 0108 110108 Beijing 1 Court of Beijing Municipality 民法院 Municipality Mentougou Mentougou District People’s 北京市门头沟区 京 0109 110109 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Changping Changping District People’s 北京市昌平区人 京 0114 110114 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Yanqing County People’s 延庆县人民法院 京 0229 110229 Yanqing County 1 Court No. 2 Intermediate People's 北京市第二中级 京 02 2 Court of Beijing Municipality 人民法院 Dongcheng Dongcheng District People’s 北京市东城区人 京 0101 110101 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Xicheng District Xicheng District People’s 北京市西城区人 京 0102 110102 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Fengtai District of Fengtai District People’s 北京市丰台区人 京 0106 110106 Beijing 1 Court of Beijing Municipality 民法院 Municipality 1 Fangshan District Fangshan District People’s 北京市房山区人 京 0111 110111 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Daxing District of Daxing District People’s 北京市大兴区人 京 0115 -

China to Keep Watch on TPP
Table for strangers Hainan helps visitors Memory protection Special police target tourism An app connects amateur chefs industry irregularities in Sanya with willing dining companions Database to be created on > CHINA, PAGE 4 > LIFE, PAGE 9 the Nanjing Massacre > p3 MONDAY, October 12, 2015 chinadailyusa.com $1 COMMERCE China to keep watch on TPP Such trade deals can disrupt non-signatories: offi cial By ZHONG NAN highly unlikely that the TPP would in Beijing lead to the creation of a trade bloc [email protected] that excludes China. “The economic development China will conduct comprehensive mode in China has already changed and systematic assessments of the from low-end product trade to ‘going fallout from the Trans-Pacifi c Part- global’ strategies like setting up or nership, a broad agreement between moving manufacturing facilities and 12 Pacifi c Rim countries, including to more direct investment in over- Japan and the United States, since it seas markets,” said Fan. believes that such deals have disrup- Besides the US, other signatories tive eff ects on non-signatory nations, to the TPP are Australia, Brunei, a top government offi cial said. Canada, Chile, Japan, Malaysia, Mex- Commerce Minister Gao Hucheng ico, New Zealand, Peru, Singapore said China is of the view that changes and Vietnam. in the global trade pattern should be China has to date signed bilateral decided by adjustments in the indus- and multilateral free trade agree- trial structure and through product ments with seven TPP members. competitiveness in global -
A Modi Catory Screening Protocol for ARDS in Patients with Respiratory
A Modicatory Screening Protocol for ARDS in Patients with Respiratory Support Based on SpO2 and FiO2 Yan Xia The First Aliated Hospital of Wannan Medical College (Yijishan Hospital of Wannan Medical College) Qiancheng Xu The First Aliated Hospital of Wannan Medical College (Yijishan Hospital of Wannan Medical College) Zhiyuan Guo The First Aliated Hospital of Wannan Medical College (Yijishan Hospital of Wannan Medical College) Huijuan Zhang The First Aliated Hospital of Wannan Medical College (Yijishan Hospital of Wannan Medical College) Yingya Cao The First Aliated Hospital of Wannan Medical College (Yijishan Hospital of Wannan Medical College) Yupeng Qi The First Aliated Hospital of Wannan Medical College (Yijishan Hospital of Wannan Medical College) Qun Chen The First Aliated Hospital of Wannan Medical College (Yijishan Hospital of Wannan Medical College) Weihua Lu ( [email protected] ) The First Aliated Hospital of Wannan Medical College (Yijishan Hospital of Wannan Medical College) Research Article Keywords: ARDS, PaO2/fraction of inspired oxygen ratio, SpO2/fraction of inspired oxygen ratio, FiO2, Diagnose Posted Date: August 4th, 2021 DOI: https://doi.org/10.21203/rs.3.rs-744649/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License A Modificatory Screening Protocol for ARDS in Patients with Respiratory Support Based on SpO2 and FiO2 Yan Xia, MD a; Qiancheng Xu, MD a,b; Zhiyuan Guo, MD a,b; Huijuan Zhang, MD a,b; Yingya Cao, MD a,b; Yupeng Qi, MD a,b; Qun Chen, MD a,b;Weihua Lu, MD a,b* a. Department of Critical Care Medicine, The First Affiliated Hospital of Wannan Medical College (Yijishan Hospital of Wannan Medical College), No.2, West road of Zheshan, Jinghu District, Wuhu, Anhui, 241000, China. -

CHINA VANKE CO., LTD.* 萬科企業股份有限公司 (A Joint Stock Company Incorporated in the People’S Republic of China with Limited Liability) (Stock Code: 2202)
Hong Kong Exchanges and Clearing Limited and The Stock Exchange of Hong Kong Limited take no responsibility for the contents of this announcement, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this announcement. CHINA VANKE CO., LTD.* 萬科企業股份有限公司 (A joint stock company incorporated in the People’s Republic of China with limited liability) (Stock Code: 2202) 2019 ANNUAL RESULTS ANNOUNCEMENT The board of directors (the “Board”) of China Vanke Co., Ltd.* (the “Company”) is pleased to announce the audited results of the Company and its subsidiaries for the year ended 31 December 2019. This announcement, containing the full text of the 2019 Annual Report of the Company, complies with the relevant requirements of the Rules Governing the Listing of Securities on The Stock Exchange of Hong Kong Limited in relation to information to accompany preliminary announcement of annual results. Printed version of the Company’s 2019 Annual Report will be delivered to the H-Share Holders of the Company and available for viewing on the websites of The Stock Exchange of Hong Kong Limited (www.hkexnews.hk) and of the Company (www.vanke.com) in April 2020. Both the Chinese and English versions of this results announcement are available on the websites of the Company (www.vanke.com) and The Stock Exchange of Hong Kong Limited (www.hkexnews.hk). In the event of any discrepancies in interpretations between the English version and Chinese version, the Chinese version shall prevail, except for the financial report prepared in accordance with International Financial Reporting Standards, of which the English version shall prevail. -

SUNAC CHINA HOLDINGS LIMITED 融創中國控股有限公司 (Incorporated in the Cayman Islands with Limited Liability) (Stock Code: 01918)
Hong Kong Exchanges and Clearing Limited and The Stock Exchange of Hong Kong Limited take no 14.88 responsibility for the contents of this announcement, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this announcement. SUNAC CHINA HOLDINGS LIMITED 融創中國控股有限公司 (Incorporated in the Cayman Islands with limited liability) (Stock Code: 01918) (1) VERY SUBSTANTIAL ACQUISITION ENTERING INTO OF FRAMEWORK AGREEMENT IN RELATION TO THE COOPERATION OF TARGET PROJECT COMPANIES AND TARGET HOTEL ASSETS AND (2) RESUMPTION OF TRADING IN SHARES THE COOPERATION The Board is pleased to announce that, on 10 July 2017, Sunac Real Estate, an indirect wholly-owned subsidiary of the Company as the buyer, and Dalian Wanda Commercial Properties as the seller entered into the Framework Agreement, pursuant to which the Buyer agreed to acquire, and the Seller agreed to dispose of 91% equity interest of the 13 cultural and tourism project companies in the PRC (i.e. the Target Project Companies) and 100% interest of the 76 city hotels (i.e. the Target Hotel Assets) at the consideration of approximately RMB29,575,000,000 and RMB33,595,260,800 respectively. Thus, the total consideration for the Cooperation is approximately RMB63,170,260,800. Before completion of the Cooperation, the Seller held 100% equity interest of the Target Project Companies and 100% interest of the Target Hotel Assets. After completion of the Cooperation, the Buyer will hold 91% equity interest of the Target Project Companies and 100% interest of the Target Hotel Assets, while the Seller will continue to hold 9% equity interest of the Target Project Companies. -

The Prognostic and Predictive Significance of Cytokeratin 5/6 And
1203 Original Article The prognostic and predictive significance of cytokeratin 5/6 and epidermal growth factor receptor in metastatic triple-negative breast cancer treated with maintenance capecitabine Yiping Zhu1#^, Kai Li2#, Jieling Zhang2#, Lu Wang1, Lili Sheng1, Liang Yan2 1Department of Oncology, The First Affiliated Hospital of Wannan Medical College, Wuhu, China; 2Wannan Medical College, Wuhu, China Contributions: (I) Conception and design: Y Zhu, L Yan; (II) Administrative support: L Sheng; (III) Provision of study materials or patients: Y Zhu, L Wang, L Sheng; (IV) Collection and assembly of data: K Li, J Zhang; (V) Data analysis and interpretation: Y Zhu, K Li, J Zhang; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors. #These authors contributed equally to this work. Correspondence to: Lili Sheng. Department of Oncology, The First Affiliated Hospital of Wannan Medical College, No. 2 Zheshan West Road, Jinghu District, Wuhu 241000, China. Email: [email protected]; Liang Yan. Department of Biochemistry, Wannan Medical College, No. 22 Wenchang West Road, Yijiang District, Wuhu 241000, China. Email: [email protected]. Background: Capecitabine is the most widely used agent for maintenance chemotherapy in patients with metastatic triple-negative breast cancer (mTNBC). However, there are no biomarkers for identifying mTNBC patients who could benefit from capecitabine maintenance. Methods: The prognostic roles of cytokeratin 5/6 (CK5/6), epidermal growth factor receptor (EGFR), and maintenance therapy were evaluated in mTNBC patients. Both CK5/6 and EGFR were detected using immunohistochemistry. Of 115 patients who achieved disease control, 56 received capecitabine maintenance therapy and 59 underwent observation. The progression-free survival (PFS) and overall survival (OS) of the patients were evaluated. -

Global Offering
China Conch Venture Holdings Limited 中國海螺創業控股有限公司Limited Holdings Venture Conch China 中國海螺創業控股有限公司 (Incorporated in the Cayman Islands with limited liability) Stock Code : 586 GLOBAL OFFERING China Conch Venture Holdings Limited 中國海螺創業控股有限公司 Sole Sponsor Joint Global Coordinators Joint Bookrunners Joint Lead Managers Conch cover_D15(22mm).indd 1 04/12/2013 8:20 PM IMPORTANT IMPORTANT: If you are in any doubt about any of the contents of this prospectus, you should seek independent professional advice. China Conch Venture Holdings Limited 中 國 海 螺 創 業 控 股 有 限 公 司 (Incorporated in the Cayman Islands with limited liability) GLOBAL OFFERING Number of Offer Shares under : 265,000,000 Shares (subject to the the Global Offering Over-allotment Option) Number of Hong Kong Offer Shares : 26,500,000 Shares (subject to adjustment) Number of International Offer Shares : 238,500,000 Shares (subject to adjustment and the Over-allotment Option) Maximum Offer Price : HK$13.56 per Offer Share, plus brokerage of 1%, SFC transaction levy of 0.003% and Stock Exchange trading fee of 0.005% (payable in full on application in Hong Kong dollars and subject to refund) Nominal value : HK$0.01 per Share Stock code : 586 Sole Sponsor Joint Global Coordinators Joint Bookrunners Joint Lead Managers Hong Kong Exchanges and Clearing Limited, The Stock Exchange of Hong Kong Limited and Hong Kong Securities Clearing Company Limited take no responsibility for the contents of this prospectus, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this prospectus. -

2020 Annual Results Announcement
Hong Kong Exchanges and Clearing Limited and The Stock Exchange of Hong Kong Limited take no responsibility for the contents of this announcement, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this announcement. (A joint stock limited company incorporated in the People’s Republic of China with limited liability) (Stock Code: 6030) 2020 ANNUAL RESULTS ANNOUNCEMENT The Board of Directors of CITIC Securities Company Limited is pleased to announce the audited results of the Company and its subsidiaries for the year ended 31 December 2020. This announcement, containing the full text of the 2020 annual report of the Company, complies with the relevant requirements of the Rules Governing the Listing of Securities on The Stock Exchange of Hong Kong Limited in relation to information to accompany preliminary announcement of annual results. The 2020 annual report of the Company and its printed version will be published and delivered to the H Shareholders of the Company and available for view on the HKExnews website of Hong Kong Exchanges and Clearing Limited at http://www.hkexnews.hk and the website of the Company at http://www.citics.com on or before 30 April 2021. 1 IMPORTANT NOTICE The Board and the Supervisory Committee and the Directors, Supervisors and Senior Management of the Company warrant the truthfulness, accuracy and completeness of the contents of this results announcement and that there is no false representation, misleading statement contained herein or material omission from this results announcement, for which they will assume joint and several liabilities. -

World Bank-Financed Anhui Aged Care System Demonstration Project
EMP for World Bank-Financed Anhui Aged Care System Demonstration Project SFG3916 Zhongzi Huayu Public Disclosure Authorized G. H. P. Z. J. Zi No. 1051 World Bank-Financed Anhui Aged Care System Demonstration Project Public Disclosure Authorized Environment and Social Management Plan (EMP) Public Disclosure Authorized Commissioned by: Department of Civil Affairs of Anhui Province Prepared by: Beijing Zhongzi Huayu Environmental Public Disclosure Authorized Protection Technology Co., Ltd. Prepared in: December 2017 EMP for World Bank-Financed Anhui Aged Care System Demonstration Project Table of Contents Preface .............................................................................................................................................. 1 1 Project overview ..................................................................................................................... 6 1.1 Project background .............................................................................................................. 6 1.2 Construction contractor ....................................................................................................... 7 1.3 Project Objectives ................................................................................................................ 7 1.4 Project details ...................................................................................................................... 8 2 Basis and standard .............................................................................................................. -

Early Awake Prone Position Combined with High-Flow Nasal
Xu et al. Critical Care (2020) 24:250 https://doi.org/10.1186/s13054-020-02991-7 RESEARCH LETTER Open Access Early awake prone position combined with high-flow nasal oxygen therapy in severe COVID-19: a case series Qiancheng Xu1†, Tao Wang1†, Xuemei Qin1†, Yanli Jie2, Lei Zha3 and Weihua Lu1* Keywords: COVID-19, High-flow nasal cannula, Prone positioning, SARS-CoV-2 Coronavirus disease 2019 (COVID-19) has rapidly observation study in three hospitals in Wuhu and Maan- spread around the world in the past 3 months and has shan cities in Anhui Province. now become a global public health crisis. The mortality From January 1 to April 2, 2020, 79 patients with cor- of COVID-19 in some European cities exceeds 11%, and onavirus infection were screened. Ten was severe and all the fatality rate is up to 61.5% in critical patients, espe- of them received early awake PP combined with HFNC cially in mechanically ventilated patients [1]. Once mild to treatment (Table 1). COVID-19 was diagnosed using moderate patients progress to critical illness, the incidence sputum or throat swab determined by real-time reverse of septic shock, intubation, and myocardial injury in- transcription polymerase chain reaction (RT-PCR) assay. creases significantly. Mechanical ventilation patients need The severity of disease was graded according to the more sedative, analgesic, and neuromuscular blocker Guidelines for the Diagnosis and Treatment of Novel drugs, which will affect the patient’s hemodynamic status Coronavirus (2019-nCoV) Infection by the National and airway expectoration ability [2]. Previous studies have Health Commission (trial version 5). -

4. Environmental Monitoring Plan
Public Disclosure Authorized Anhui Medium Cities Urban Transport Project Environment Management Plan (Wuhu) Public Disclosure Authorized Public Disclosure Authorized Anhui Normal University Public Disclosure Authorized No. 2106 by Level B Environment Impact Assessment Agency November, 2014 Table of Content 1. INTRODUCTION ........................................................................................................... 1 1.1 PROJECT BACKGROUND ............................................................................................. 1 1.2 PROJECT COMPONENT ................................................................................................ 2 1.3 TARGET OF THE EMP .................................................................................................. 5 1.4 EVALUATION CRITERIA .............................................................................................. 5 1.5 ENVIRONMENTAL PROTECTION GOAL ....................................................................... 7 1.5.1 Goal for Noise Environment ................................................................................... 7 1.5.2 Goal for Water Environment ................................................................................... 8 2. ENVIRONMENTAL IMPACT AND MITIGATION MEASURES ............................ 9 3. ENVIRONMENTAL MANAGEMENT AND MONITORING ................................. 18 4. ENVIRONMENTAL MONITORING PLAN ............................................................. 21 4.1 ENVIRONMENTAL MONITORING PLAN .................................................................... -

Yushan District
Yushan District Yushan District. Quite the same Wikipedia. Just better. Yushan District is a district of Anhui Province, China. It is under the administration of Ma'anshan city. Places adjacent to Yushan District. Langya District. Luyang District. District in Anhui, People's Republic of China Yushan雨山区 District Coordinates: 31°40â²43â³N 118°29â²29â³E / 31.67861°N 118. Places adjacent to Yushan District. Langya District. Luyang District. Yushan District. Jinghu District. Xuanzhou District. v. *Xishan Mountains, a mountain range in western Beijing, China *Xishan District, Kunming, a district in Kunming, Yunnan Province, ChinaXishan may also refer to: *Xishan District, Wuxi (锡山) ⦠Wikipedia. Kunming ┠昆明 Prefecture level city 昆明市 Clockwise from top: Kunming s Skyli ⦠Xishan District (simplified Chinese: 锡山区; traditional Chinese: 錫山å; pinyin: XÄ«shÄn QÅ«) is one of five urban districts of Wuxi, Jiangsu Province, People's Republic of China. With a total area of 454 km2 (175 sq mi), the district's 2007 population was 400,000. Administrative divisions[]. Xishan includes the eight towns of DÅngtÃng, Anzhèn, YángjiÄn, HóngshÄn, Ehú, DÅngbÄ›i Táng, XÄ«bÄ›i and DÅnggÇŽng. External links[]. Wuxi Xishan District site. v. t. Xishan District is one of five urban districts of Wuxi, Jiangsu Province, People's Republic of China. With a total area of 454 km2 (175 sq mi), the district's 2007 population was 400,000. Xishan includes the eight towns of DÅngtÃng, Anzhèn, YángjiÄn, HóngshÄn, Ehú, DÅngbÄ›i Táng, XÄ«bÄ›i and DÅnggÇŽng.