Chapter 11 – Electron Transfer Reactions and Electrochem- Istry Introduction
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Structure-Function Relationship of Metalloproteins
RESEARCH ACTIVITIES Structure-Function Relationship of Metalloproteins Department of Life and Coordination-Complex Molecular Science Division of Biomolecular Functions FUJII, Hiroshi Associate Professor KURAHASHI, Takuya Assistant Professor CONG, Zhiqi IMS Fellow OHTSUKI, Akimichi Post-Doctoral Fellow WANG, Chunlan Graduate Student TANIZAWA, Misako Secretary Metalloproteins are a class of biologically important A d4 high-spin manganese(III) ion forms a Robin-Day Class II macromolecules, which have various functions such as oxygen mixed-valence system, in which electron transfer is occurring transport, electron transfer, oxidation, and oxygenation. These between the localized phenoxyl radical and the phenolate. This diverse functions of metalloproteins have been thought to is in clear contrast to a d8 low-spin nickel(II) ion with the depend on the ligands from amino acid, coordination struc- same salen ligand, which induces a delocalized radical (Robin- tures, and protein structures in immediate vicinity of metal Day Class III) over the two phenolate rings, as previously ions. In this project, we are studying the relationship between reported by others. The present findings point to a fascinating the electronic structures of the metal active sites and reactivity possibility that electron transfer could be drastically modu- of metalloproteins. lated by exchanging the metal ion that bridges the two redox centers. 1. One-Electron Oxidation of One-electron Oxidation Electronically-Diverse Manganese(III) and N N N N Nickel(II) Salen Complexes: Transition from M M R O O R R O O R Localized to Delocalized Mixed-Valence tBu tBu 3+ 2+ tBu tBu Ligand Radicals1) M = Mn , Ni t R = Bu, OCH3, Ph Electron Transfer Ligand radicals from salen complexes are unique mixed- Figure 1. -

Process for Purification of Ethylene Compound Having Fluorine-Containing Organic Group
Office europeen des brevets (fi) Publication number : 0 506 374 A1 @ EUROPEAN PATENT APPLICATION @ Application number: 92302586.0 @ Int. CI.5: C07C 17/38, C07C 21/18 (g) Date of filing : 25.03.92 (30) Priority : 26.03.91 JP 86090/91 (72) Inventor : Kishita, Hirofumi 3-19-1, Isobe, Annaka-shi Gunma-ken (JP) (43) Date of publication of application : Inventor : Sato, Shinichi 30.09.92 Bulletin 92/40 3-5-5, Isobe, Annaka-shi Gunma-ken (JP) Inventor : Fujii, Hideki (84) Designated Contracting States : 3-12-37, Isobe, Annaka-shi DE FR GB Gunma-ken (JP) Inventor : Matsuda, Takashi 791 ~4 YdndSG Anri3k3~shi @ Applicant : SHIN-ETSU CHEMICAL CO., LTD. Gunma-ken (JP) 6-1, Ohtemachi 2-chome Chiyoda-ku Tokyo (JP) @) Representative : Votier, Sidney David et al CARPMAELS & RANSFORD 43, Bloomsbury Square London WC1A 2RA (GB) (54) Process for purification of ethylene compound having fluorine-containing organic group. (57) A process for purifying an ethylene compound having a fluorine-containing organic group (fluorine- containing ethylene compound) by mixing the fluorine-containing ethylene compound with an alkali metal or alkaline earth metal reducing agent, and subjecting the resulting mixture to irradiation with ultraviolet radiation, followed by washing with water. The purification process ensures effective removal of iodides which are sources of molecular iodine, from the fluorine-containing ethylene compound. < h- CO CO o 10 o Q_ LU Jouve, 18, rue Saint-Denis, 75001 PARIS EP 0 506 374 A1 BACKGROUND OF THE INVENTION 1. Field of the Invention 5 The present invention relates to a process for purifying an ethylene compound having a fluorine-containing organic group, and more particularly to a purification process by which iodine contained as impurity in an ethylene compound having a fluorine-containing organic group can be removed effectively. -
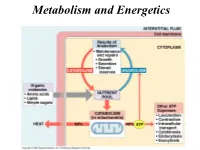
Metabolism and Energetics Oxidation of Carbon Atoms of Glucose Is the Major Source of Energy in Aerobic Metabolism
Metabolism and Energetics Oxidation of carbon atoms of glucose is the major source of energy in aerobic metabolism C6H1206 + 6O2 yields 6 CO2 + H20 + energy Energy released ΔG = - 686 kcal/mol Glucose oxidation requires over 25 discrete steps, with production of 36 ATP. Energy Transformations The mitochondrial synthesis of ATP is not stochiometric. Electron–motive force Proton-motive force Phosphoryl-transfer potential in the form of ATP. Substrate level phosphorylation The formation of ATP by substrate-level phosphorylation ADP ATP P CH O P CH2 O P 2 is used to represent HC OH HC OH a phosphate ester: phosphoglycerate CH OH OH CH2 O P kinase 2 P O bisphosphoglycerate 3-phosphoglycerate OH ADP ATP CH2 CH2 CH3 C O P C OH C O pyruvate kinase non-enzymic COOH COOH COOH phosphoenolpyruvate enolpyruvate pyruvate Why ATP? The reaction of ATP hydrolysis is very favorable ΔGo = - 30.5 kJ/mol = - 7.3 kCal/mol 1. Charge separation of closely packed phosphate groups provides electrostatic relief. Mg2+ 2. Inorganic Pi, the product of the reaction, is immediately resonance-stabilized (electron density spreads equally to all oxygens). 3. ADP immediately ionizes giving H+ into a low [H+] environment (pH~7). 4. Both Pi and ADP are more favorably solvated by water than one ATP molecule. 5. ATP is water soluble. The total body content of ATP and ADP is under 350 mmol – about 10 g, BUT … the amount of ATP synthesized and used each day is about 150 mol – about 110 kg. ATP Production - stage 1 - Glycolysis Glycolysis When rapid production of ATP is needed. -

Reductions and Reducing Agents
REDUCTIONS AND REDUCING AGENTS 1 Reductions and Reducing Agents • Basic definition of reduction: Addition of hydrogen or removal of oxygen • Addition of electrons 9:45 AM 2 Reducible Functional Groups 9:45 AM 3 Categories of Common Reducing Agents 9:45 AM 4 Relative Reactivity of Nucleophiles at the Reducible Functional Groups In the absence of any secondary interactions, the carbonyl compounds exhibit the following order of reactivity at the carbonyl This order may however be reversed in the presence of unique secondary interactions inherent in the molecule; interactions that may 9:45 AM be activated by some property of the reacting partner 5 Common Reducing Agents (Borohydrides) Reduction of Amides to Amines 9:45 AM 6 Common Reducing Agents (Borohydrides) Reduction of Carboxylic Acids to Primary Alcohols O 3 R CO2H + BH3 R O B + 3 H 3 2 Acyloxyborane 9:45 AM 7 Common Reducing Agents (Sodium Borohydride) The reductions with NaBH4 are commonly carried out in EtOH (Serving as a protic solvent) Note that nucleophilic attack occurs from the least hindered face of the 8 carbonyl Common Reducing Agents (Lithium Borohydride) The reductions with LiBH4 are commonly carried out in THF or ether Note that nucleophilic attack occurs from the least hindered face of the 9:45 AM 9 carbonyl. Common Reducing Agents (Borohydrides) The Influence of Metal Cations on Reactivity As a result of the differences in reactivity between sodium borohydride and lithium borohydride, chemoselectivity of reduction can be achieved by a judicious choice of reducing agent. 9:45 AM 10 Common Reducing Agents (Sodium Cyanoborohydride) 9:45 AM 11 Common Reducing Agents (Reductive Amination with Sodium Cyanoborohydride) 9:45 AM 12 Lithium Aluminium Hydride Lithium aluminiumhydride reacts the same way as lithium borohydride. -

Chapter 20 Electrochemistry
Chapter 20 Electrochemistry Learning goals and key skills: Identify oxidation, reduction, oxidizing agent, and reducing agent in a chemical equation Complete and balance redox equations using the method of half-reactions. Sketch a voltaic cell and identify its cathode, anode, and the directions in which electrons and ions move. o Calculate standard emfs (cell potentials), E cell, from standard reduction potentials. Use reduction potentials to predict whether a redox reaction is spontaneous. o o Relate E cell to DG and equilibrium constants. Calculate emf under nonstandard conditions. Identify the components of common batteries. Describe the construction of a lithium-ion battery and explain how it works. Describe the construction of a fuel cell and explain how it generates electrical energy. Explain how corrosion occurs and how it is prevented by cathodic protection. Describe the reactions in electrolytic cells. Relate the amounts of products and reactants in redox reactions to electrical charge. Electrochemistry Electrochemistry is the study of the relationships between electricity and chemical reactions. • It includes the study of both spontaneous and nonspontaneous processes. 1 Redox reactions: assigning oxidation numbers Oxidation numbers help keep track of what species loses electrons and what species gains them. • An element is oxidized when the oxidation number increases • An element is reduced when the oxidation number decreases • an oxidizing agent causes another element to be oxidized • a reducing agent causes another element to be reduced. Assigning oxidation numbers (sect. 4.4) 1. Elemental form, each atom has ox. # = 0. Zn O2 O3 I2 S8 P4 2. Simple ions, = charge on ion. -1 for Cl-, +2 for Mg2+ 3. -

Concepts and Tools for Mechanism and Selectivity Analysis in Synthetic Organic Electrochemistry
Concepts and tools for mechanism and selectivity analysis in synthetic organic electrochemistry Cyrille Costentina,1,2 and Jean-Michel Savéanta,1 aUniversité Paris Diderot, Sorbonne Paris Cité, Laboratoire d’Electrochimie Moléculaire, Unité Mixte de Recherche Université–CNRS 7591, 75205 Paris Cedex 13, France Contributed by Jean-Michel Savéant, April 2, 2019 (sent for review March 19, 2019; reviewed by Robert Francke and R. Daniel Little) As an accompaniment to the current renaissance of synthetic organic sufficient to record a current-potential response but small electrochemistry, the heterogeneous and space-dependent nature of enough to leave the substrates and cosubstrates (of the order of electrochemical reactions is analyzed in detail. The reactions that follow one part per million) almost untouched. Competition of the the initial electron transfer step and yield the products are intimately electrochemical/chemical events with diffusional transport under coupled with reactant transport. Depiction of the ensuing reactions precisely mastered conditions allows analysis of the kinetics profiles is the key to the mechanism and selectivity parameters. within extended time windows (from minutes to submicroseconds). Analysis is eased by the steady state resulting from coupling of However, for irreversible processes, these approaches are blind on diffusion with convection forced by solution stirring or circulation. reaction bifurcations occurring beyond the kinetically determining Homogeneous molecular catalysis of organic electrochemical reactions step, which are precisely those governing the selectivity of the re- of the redox or chemical type may be treated in the same manner. The same benchmarking procedures recently developed for the activation action. This is not the case of preparative-scale electrolysis accom- of small molecules in the context of modern energy challenges lead to panied by identification and quantitation of products. -

Ch. 21.1 Redox Reactions and Electrochemical Cells
Pre-Health Post-Baccalaureate Program Study Guide and Practice Problems Course: CHM2046 Textbook Chapter: 21.1 (Silberberg 6e) Topics Covered: Redox Reactions and Electrochemical Cells Created by Isaac Loy 1. Review Understanding this chapter’s material will depend on an in- depth understanding of redox reactions, which were first covered last semester in ch. 4. We will review redox reactions in this study guide, but it would be wise to review ch. 4 if you are having difficulty with this material. Redox reactions will also be incredibly important moving forward into organic chemistry and biochemistry. 2. Oxidation-Reduction Reactions The mnemonic that you will come back to time and time again for this topic is: “LEO the lion says GER” Where “LEO” stands for Loss of Electrons = Oxidation And “GER” stands for Gain of Electrons = Reduction The oxidizing agent (the substance that is being reduced) pulls electrons from the substance that is being oxidized. The reducing agent (the substance that is being oxidized) gives electrons to the substance that is being reduced. Oxidation and reduction are simultaneous processes. In order for a redox reaction to take place, one substance must be oxidized and the other must be reduced. When working with oxidation numbers to solve problems, the substance being oxidized (LEO -> loss of electrons) becomes more positive. Likewise, the substance being reduced (GER -> gaining electrons) becomes more negative. 3. Using Half-Reactions to Solve Redox Problems Follow the steps below to create half-reactions. It is crucial that you follow the steps in order! A. Split up the overall reaction into two half-reactions, where the species of one reaction is being oxidized, and the species of the other reaction is being reduced. -

Gen Chem II Jasperse Ch. 19 Electrochemistry 1
Gen Chem II Jasperse Ch. 19 Electrochemistry 1 Chapter 19 Electrochemistry Math Summary Relating Standard Cell Potential to Standard Half Cell Potentials Eºcell=Eºoxidation + Eºreduction (standard conditions assume 1.0 M concentrations) Relating Half Cell Potentials when Written in Opposite Directions Eºox = -Eºred for half reactions written in opposite directions Relating Standard Cell Potentials to ∆G ∆Gº = -nFE˚cell (to give answer in kJ, use F = 96.485) F = 96,500 C/mol n=number of electrons transferred Relating Actual Cell Potential to Standard Cell Potential when Concentrations aren't 1.0-M Ecell = Eºcell -[0.0592/n] log Q (Q = ratio of actual concentrations) Relating Standard Cell Potential to Equilibrium Constant log K = nEº/0.0592 Relating Actual Cell Potential to Actual Concentrations in Concentration Cells Ecell = -[0.0592/n] log Q for concentration cells, where anode and cathode differ only in concentration, but otherwise have same ions Relating # of Moles of Electrons Transferred as a Function of Time and Current in Electrolysis 1 mol e- = 96,500 C moles of electrons = [current (A)•time (sec)]/96,500 for electrolysis, moles, current, and time are related. rearranged: time (sec)=(moles of electrons)(96500)/current (in A) Note: 3600 sec/hour so time (hours)=(moles of electrons)(26.8)/current (in A) Electrochemistry-Related Units C = Coulomb = quantity of electrical charge = 6.24 • 1018 electrons • 1 mole of electrons = 96,500 C A = amp = rate of charge flow per time = C/sec V = volt = electrical power/force/strength = J/C 96,500C 96.5 kJ F = Faraday = = mole e− mole e− •V € € Gen Chem II Jasperse Ch. -
![Arxiv:1706.09241V1 [Cond-Mat.Mtrl-Sci] 28 Jun 2017 Based Electrolyte Supercapacitors Remain Somewhat Sim- Confinement Might Have a Drastic Influence on the Sol- Ilar](https://docslib.b-cdn.net/cover/0681/arxiv-1706-09241v1-cond-mat-mtrl-sci-28-jun-2017-based-electrolyte-supercapacitors-remain-somewhat-sim-con-nement-might-have-a-drastic-in-uence-on-the-sol-ilar-580681.webp)
Arxiv:1706.09241V1 [Cond-Mat.Mtrl-Sci] 28 Jun 2017 Based Electrolyte Supercapacitors Remain Somewhat Sim- Confinement Might Have a Drastic Influence on the Sol- Ilar
Confinement Effects on an Electron Transfer Reaction in Nanoporous Carbon Electrodes Zhujie Li1;2;3, Guillaume Jeanmairet2;3, Trinidad M´endez-Morales1;2;3, Mario Burbano1;3, Matthieu Haefele1, Mathieu Salanne1;2;3 1Maison de la Simulation, CEA, CNRS, Univ. Paris-Sud, UVSQ, Universit´eParis-Saclay, F-91191 Gif-sur-Yvette, France 2Sorbonne Universit´es,UPMC Univ Paris 06, CNRS, Laboratoire PHENIX, F-75005 Paris, France and 3R´eseau sur le Stockage Electrochimique´ de l'Energie´ (RS2E), FR CNRS 3459, France∗ Nanoconfinement generally leads to drastic effect on the physical and chemical properties of ionic liquids. Here we investigate how the electrochemical reactivity in such media may be impacted inside nanoporous carbon electrodes. To this end, we study a simple electron transfer reaction using molecular dynamics simulations. The electrodes are held at constant electric potential by allowing the atomic charges on the carbon atoms to fluctuate. We show that the Fe3+=Fe2+ couple dissolved in an ionic liquid exhibits a deviation with respect to Marcus theory. This behavior is rationalized by the stabilization of a solvation state of the Fe3+ cation in the disordered nanoporous electrode that is not observed in the bulk. The simulation results are fitted with a recently proposed two solvation state model, which allows us to estimate the effect of such a deviation on the kinetics of electron transfer inside nanoporous electrodes. Nanoconfinement effects strongly impact on many liq- a supercapacitor16. Among the various questions raised uid properties, such as transport, diffusion coefficients, by this study, the most important ones are: How is the phase transitions, and solvation structures1{4. -

Evolution of the First Metabolic Cycles
Proc. Natl. Acad. Sci. USA Vol. 87, pp. 200-204, January 1990 Evolution Evolution of the first metabolic cycles (chemoautotrophy/reductive citric acid cycle/origin of life/pyrite) GUNTER WACHTERSHAUSER 8000 Munich 2, Tal 29, Federal Republic of Germany Communicated by Karl Popper, October 12, 1989 (received for review February 28, 1989) ABSTRACT There are two alternatives concerning the genobacter thermophilus (13), and Desulfobacter hydro- origin of life: the origin may be heterotrophic or autotrophic. genophilus (14) and also in the sulfur-associated archaebac- The central problem within the theory of an autotrophic origin teria Thermoproteus neutrophilus (15) and, partly demon- is the first process of carbon fixation. I here propose the strated, in Sulfolobus brierleyi (16). As suggested by Kandler hypothesis that this process is an autocatalytic cycle that can be and Stetter (16) and previously by Hartmann (17), it may be retrodictively constructed from the extant reductive citric acid considered to be of great antiquity and the evolutionary cycle by replacing thioesters by thioacids and by assuming that precursor ofthe oxidative Krebs cycle. It is here conjectured the required reducing power is obtained from the oxidative to be the extant candidate for the reconstruction of the formation of pyrite (FeS2). This archaic cycle is strictly archaic autocatalytic cycle of carbon fixation. chemoautotrophic: photoautotrophy is not required. The cycle The presently accepted form of the extant reductive citric is catalytic for pyrite formation and autocatalytic for its own acid cycle is shown in Fig. 1 in a somewhat unusual repre- multiplication. It is a consequence of this hypothesis that the sentation, twisted in an 8. -

Oxidation-Reduction Reactions
Oxidation-Reduction Reactions What is an Oxidation-Reduction, or Redox, reaction? Oxidation-reduction reactions, or redox reactions, are technically defined as any chemical reaction in which the oxidation number of the participating atom, ion, or molecule of a chemical compound changes. Some common redox reactions include fire, rusting of metals, browning of fruit, and photosynthesis. In simpler terms, redox reactions involve the transfer of electrons from one substance to another. In a redox reaction, electrons can never be “lost”; if one substance loses electrons, another substance must gain an equal number of electrons. Therefore, oxidation and reduction always happen at the same time; they are a matched set. What is Oxidation? Oxidation is the loss of electrons by an atom, ion, or molecule. The atom, ion, or molecule that is oxidized will become more positively charged. Oxidation may also involve the addition of oxygen or the loss of hydrogen. What is Reduction? Reduction is the gain of electrons by an atom, ion, or molecule. The atom, ion, or molecule that is reduced will become more negatively charged. Reduction may also involve the loss of oxygen or the gain of hydrogen. Tips for determining if an atom, ion, or compound has been Oxidized or Reduced: • Remembering one of the two following mnemonics can be of assistance in determining the difference between oxidation and reduction: 1) OIL RIG- Oxidation Is Loss (of electrons), Reduction Is Gain (of electrons) or 2) LEO GER- Loss of Electrons- Oxidation, Gain of Electrons- Reduction • Atoms of metals tend to lose electrons to form positive ions (oxidation) • Atoms of non-metals tend to gain electrons to form negative ions (reduction). -

Design and Fine-Tuning Redox Potentials of Metalloproteins Involved in Electron Transfer in Bioenergetics
1 Design and Fine-tuning Redox Potentials of Metalloproteins Involved in Electron Transfer in Bioenergetics Parisa Hosseinzadeh and Yi Lu Abstract: 1. Introduction A significant portion of biological processes are involved with providing vital energy sources such as ATP, and controlling the flow of energy through living systems. These bioenergetics processes, the most important of which are photosynthesis and respiration, require electron transfer (ET) between different redox partners. Metalloproteins are one of the most widely used ET centers in biology. They can be classified into three major classes: cupredoxins, which include type 1 copper (T1Cu) proteins and CuA centers [1- 10], cytochromes [10-17], and iron-sulfur (FeS) proteins [10,18-24]. Each class of ET proteins transfer electrons between different redox partners which possess different reduction potentials (E°) (Figure 1). Therefore, the ET centers need to adjust their E° in a way that matches those of their redox partners. No single class of protein can cover the entire range of physiological E°, which is between ~ -1V, at which protons are reduced to H2, and 1V, at which water is oxidized to O2. Cupredoxins usually function at the high end of the E°, while FeS proteins are mostly involved in ET reactions possessing relatively low E° [10]. The E°’s of FeS proteins overlap significantly with those of cytochromes that often have intermediate E° among the three classes of ET proteins. Figure 1. Reduction potential range of metal centers in electron transfer metalloprotein. Adapted from ref. [10] 2 In this review, we first describe the importance of tuning E° of ET centers, including the metalloproteins described above.