Año Internacional De La Química 2011
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Almost Forgotten Anniversaries in 2019 Introduction
Almost Forgotten Anniversaries in 2019 Katharina Lodders Department of Earth and Planetary Sciences and Mc Donnell Center for the Space Sciences, Campus Box 1169, Washington University, Saint Louis MO 63130, USA Keywords: history, chemical elements, abundances Abstract: As we celebrate the International Year of the Periodic Table, the 50th anniversary of Apollo 11 and the meteorite falls of Allende and Murchison in 1969, other noteworthy science events with round birthdays seem to be overlooked and almost forgotten Several scientific organizations celebrate the birthdays of their foundation; and key events and discoveries related to meteoritics, astronomy, geo- and cosmochemistry, and nuclear sciences can be commemorated this year, including the anniversaries of the discoveries of eleven chemical elements, and the advancements of our knowledge of the elemental and isotopic abundances. Introduction. Introduction The 150th anniversary of the discovery of the periodic system of the elements by Dmitri Ivanovich Mendeleev (8 Feb. 1834 – 2 Feb. 1907) and independently by Julius Lothar Meyer (19 August 1830 – 11 April 1895) is the reason for celebrating the International Year of the Periodic Table in 2019. Not only that, but several scientific organizations celebrate the birthdays of their foundation: The Astronomical Society of the Pacific (1889), The American Astronomical Society (1899), the American Geophysical Union (1919), the Mineralogical Society of America (1919), and the International Astronomical Union IAU (1919). The anniversaries in 2019 give us reasons to reflect on the major impacts of space exploration. In 1969, the first men landed on the moon and Apollo 11 safely returned with lunar rocks for study. The same year was blessed by the fall of the important carbonaceous chondrites Allende and Murchison. -

Sequencing As a Way of Work
Edinburgh Research Explorer A new insight into Sanger’s development of sequencing Citation for published version: Garcia-Sancho, M 2010, 'A new insight into Sanger’s development of sequencing: From proteins to DNA, 1943-77', Journal of the History of Biology, vol. 43, no. 2, pp. 265-323. https://doi.org/10.1007/s10739-009- 9184-1 Digital Object Identifier (DOI): 10.1007/s10739-009-9184-1 Link: Link to publication record in Edinburgh Research Explorer Document Version: Peer reviewed version Published In: Journal of the History of Biology Publisher Rights Statement: © Garcia-Sancho, M. (2010). A new insight into Sanger’s development of sequencing: From proteins to DNA, 1943-77. Journal of the History of Biology, 43(2), 265-323. 10.1007/s10739-009-9184-1 General rights Copyright for the publications made accessible via the Edinburgh Research Explorer is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy The University of Edinburgh has made every reasonable effort to ensure that Edinburgh Research Explorer content complies with UK legislation. If you believe that the public display of this file breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 28. Sep. 2021 THIS IS AN ADVANCED DRAFT OF A PUBLISHED PAPER. REFERENCES AND QUOTATIONS SHOULD ALWAYS BE MADE TO THE PUBLISHED VERION, WHICH CAN BE FOUND AT: García-Sancho M. -

Historical Development of the Periodic Classification of the Chemical Elements
THE HISTORICAL DEVELOPMENT OF THE PERIODIC CLASSIFICATION OF THE CHEMICAL ELEMENTS by RONALD LEE FFISTER B. S., Kansas State University, 1962 A MASTER'S REPORT submitted in partial fulfillment of the requirements for the degree FASTER OF SCIENCE Department of Physical Science KANSAS STATE UNIVERSITY Manhattan, Kansas 196A Approved by: Major PrafeLoor ii |c/ TABLE OF CONTENTS t<y THE PROBLEM AND DEFINITION 0? TEH-IS USED 1 The Problem 1 Statement of the Problem 1 Importance of the Study 1 Definition of Terms Used 2 Atomic Number 2 Atomic Weight 2 Element 2 Periodic Classification 2 Periodic Lav • • 3 BRIEF RtiVJiM OF THE LITERATURE 3 Books .3 Other References. .A BACKGROUND HISTORY A Purpose A Early Attempts at Classification A Early "Elements" A Attempts by Aristotle 6 Other Attempts 7 DOBEREBIER'S TRIADS AND SUBSEQUENT INVESTIGATIONS. 8 The Triad Theory of Dobereiner 10 Investigations by Others. ... .10 Dumas 10 Pettehkofer 10 Odling 11 iii TEE TELLURIC EELIX OF DE CHANCOURTOIS H Development of the Telluric Helix 11 Acceptance of the Helix 12 NEWLANDS' LAW OF THE OCTAVES 12 Newlands' Chemical Background 12 The Law of the Octaves. .........' 13 Acceptance and Significance of Newlands' Work 15 THE CONTRIBUTIONS OF LOTHAR MEYER ' 16 Chemical Background of Meyer 16 Lothar Meyer's Arrangement of the Elements. 17 THE WORK OF MENDELEEV AND ITS CONSEQUENCES 19 Mendeleev's Scientific Background .19 Development of the Periodic Law . .19 Significance of Mendeleev's Table 21 Atomic Weight Corrections. 21 Prediction of Hew Elements . .22 Influence -

Cambridge's 92 Nobel Prize Winners Part 2 - 1951 to 1974: from Crick and Watson to Dorothy Hodgkin
Cambridge's 92 Nobel Prize winners part 2 - 1951 to 1974: from Crick and Watson to Dorothy Hodgkin By Cambridge News | Posted: January 18, 2016 By Adam Care The News has been rounding up all of Cambridge's 92 Nobel Laureates, celebrating over 100 years of scientific and social innovation. ADVERTISING In this installment we move from 1951 to 1974, a period which saw a host of dramatic breakthroughs, in biology, atomic science, the discovery of pulsars and theories of global trade. It's also a period which saw The Eagle pub come to national prominence and the appearance of the first female name in Cambridge University's long Nobel history. The Gender Pay Gap Sale! Shop Online to get 13.9% off From 8 - 11 March, get 13.9% off 1,000s of items, it highlights the pay gap between men & women in the UK. Shop the Gender Pay Gap Sale – now. Promoted by Oxfam 1. 1951 Ernest Walton, Trinity College: Nobel Prize in Physics, for using accelerated particles to study atomic nuclei 2. 1951 John Cockcroft, St John's / Churchill Colleges: Nobel Prize in Physics, for using accelerated particles to study atomic nuclei Walton and Cockcroft shared the 1951 physics prize after they famously 'split the atom' in Cambridge 1932, ushering in the nuclear age with their particle accelerator, the Cockcroft-Walton generator. In later years Walton returned to his native Ireland, as a fellow of Trinity College Dublin, while in 1951 Cockcroft became the first master of Churchill College, where he died 16 years later. 3. 1952 Archer Martin, Peterhouse: Nobel Prize in Chemistry, for developing partition chromatography 4. -

RICHARD LAWRENCE MILLINGTON SYNGE BA, Phd(Cantab), Hondsc(Aberd), Hondsc(E.Anglia), Hon Dphil(Uppsala), FRS Nobel Laureate 1952
RICHARD LAWRENCE MILLINGTON SYNGE BA, PhD(Cantab), HonDSc(Aberd), HonDSc(E.Anglia), Hon DPhil(Uppsala), FRS Nobel Laureate 1952 R L M Synge was elected FRSE in 1963. He was born in West Kirby, Cheshire, on 28 October 1914, the son of Katherine (née Swan) and Lawrence Millington Synge, a Liverpool Stockbroker. The family was known to be living in Bridgenorth (Salop) in the early sixteenth century. At that time the name was Millington and there is a story that a member of the family from Millington Hall in Rostherne (Cheshire) sang so beautifully before King Henry VIII that he was told to take the name Synge. There have been various spellings of the name and in the nineteenth century the English branch settled on Sing which they retained until 1920 when both R M and L M Sing (Dick's uncle and father respectively) changed their names by deed poll to Synge. In the 19th and 20th centuries the Sing/Synge family played a considerable part in the life of Liverpool and Dick's father was High Sheriff of Cheshire in 1954. Dick was educated at Old Hall, a prep school in Wellington (Salop) and where he became renowned for his ability in Latin and Greek, subjects which he continued to study with such success at Winchester that in December 1931 when, just turned 17, he was awarded an Exhibition in Classics by Trinity College, Cambridge. His intention was to study science and Trinity allowed Dick to switch from classics to the Natural Sciences Tripos. To prepare for the change he returned to Winchester in January 1932 to study science for the next 18 months and was awarded the senior science prize for 1933. -

A Brief History of Nuclear Astrophysics
A BRIEF HISTORY OF NUCLEAR ASTROPHYSICS PART I THE ENERGY OF THE SUN AND STARS Nikos Prantzos Institut d’Astrophysique de Paris Stellar Origin of Energy the Elements Nuclear Astrophysics Astronomy Nuclear Physics Thermodynamics: the energy of the Sun and the age of the Earth 1847 : Robert Julius von Mayer Sun heated by fall of meteors 1854 : Hermann von Helmholtz Gravitational energy of Kant’s contracting protosolar nebula of gas and dust turns into kinetic energy Timescale ~ EGrav/LSun ~ 30 My 1850s : William Thompson (Lord Kelvin) Sun heated at formation from meteorite fall, now « an incadescent liquid mass » cooling Age 10 – 100 My 1859: Charles Darwin Origin of species : Rate of erosion of the Weald valley is 1 inch/century or 22 miles wild (X 1100 feet high) in 300 My Such large Earth ages also required by geologists, like Charles Lyell A gaseous, contracting and heating Sun 푀⊙ Mean solar density : ~1.35 g/cc Sun liquid Incompressible = 4 3 푅 3 ⊙ 1870s: J. Homer Lane ; 1880s :August Ritter : Sun gaseous Compressible As it shrinks, it releases gravitational energy AND it gets hotter Earth Mayer – Kelvin - Helmholtz Helmholtz - Ritter A gaseous, contracting and heating Sun 푀⊙ Mean solar density : ~1.35 g/cc Sun liquid Incompressible = 4 3 푅 3 ⊙ 1870s: J. Homer Lane ; 1880s :August Ritter : Sun gaseous Compressible As it shrinks, it releases gravitational energy AND it gets hotter Earth Mayer – Kelvin - Helmholtz Helmholtz - Ritter A gaseous, contracting and heating Sun 푀⊙ Mean solar density : ~1.35 g/cc Sun liquid Incompressible = 4 3 푅 3 ⊙ 1870s: J. -
RSC Branding
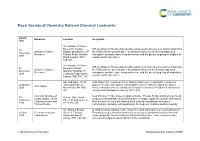
Royal Society of Chemistry National Chemical Landmarks Award Honouree Location Inscription Date The Institute of Cancer Research, Chester ICR scientists on this site and elsewhere pioneered numerous new cancer drugs from 10 Institute of Cancer Beatty Laboratories, 237 the 1950s until the present day – including the discovery of chemotherapy drug December Research Fulham Road, Chelsea carboplatin, prostate cancer drug abiraterone and the genetic targeting of olaparib for 2018 Road, London, SW3 ovarian and breast cancer. 6JB, UK The Institute of Cancer ICR scientists on this site and elsewhere pioneered numerous new cancer drugs from 10 Research, Royal Institute of Cancer the 1950s until the present day – including the discovery of chemotherapy drug December Marsden Hospital, 15 Research carboplatin, prostate cancer drug abiraterone and the genetic targeting of olaparib for 2018 Cotswold Road, Sutton, ovarian and breast cancer. London, SM2 5NG, UK Ape and Apple, 28-30 John Dalton Street was opened in 1846 by Manchester Corporation in honour of 26 October John Dalton Street, famous chemist, John Dalton, who in Manchester in 1803 developed the Atomic John Dalton 2016 Manchester, M2 6HQ, Theory which became the foundation of modern chemistry. President of Manchester UK Literary and Philosophical Society 1816-1844. Chemical structure of Near this site in 1903, James Colquhoun Irvine, Thomas Purdie and their team found 30 College Gate, North simple sugars, James a way to understand the chemical structure of simple sugars like glucose and lactose. September Street, St Andrews, Fife, Colquhoun Irvine and Over the next 18 years this allowed them to lay the foundations of modern 2016 KY16 9AJ, UK Thomas Purdie carbohydrate chemistry, with implications for medicine, nutrition and biochemistry. -

Los Premios Nobel De Química
Los premios Nobel de Química MATERIAL RECOPILADO POR: DULCE MARÍA DE ANDRÉS CABRERIZO Los premios Nobel de Química El campo de la Química que más premios ha recibido es el de la Quí- mica Orgánica. Frederick Sanger es el único laurea- do que ganó el premio en dos oca- siones, en 1958 y 1980. Otros dos también ganaron premios Nobel en otros campos: Marie Curie (física en El Premio Nobel de Química es entregado anual- 1903, química en 1911) y Linus Carl mente por la Academia Sueca a científicos que so- bresalen por sus contribuciones en el campo de la Pauling (química en 1954, paz en Física. 1962). Seis mujeres han ganado el Es uno de los cinco premios Nobel establecidos en premio: Marie Curie, Irène Joliot- el testamento de Alfred Nobel, en 1895, y que son dados a todos aquellos individuos que realizan Curie (1935), Dorothy Crowfoot Ho- contribuciones notables en la Química, la Física, la dgkin (1964), Ada Yonath (2009) y Literatura, la Paz y la Fisiología o Medicina. Emmanuelle Charpentier y Jennifer Según el testamento de Nobel, este reconocimien- to es administrado directamente por la Fundación Doudna (2020) Nobel y concedido por un comité conformado por Ha habido ocho años en los que no cinco miembros que son elegidos por la Real Aca- demia Sueca de las Ciencias. se entregó el premio Nobel de Quí- El primer Premio Nobel de Química fue otorgado mica, en algunas ocasiones por de- en 1901 al holandés Jacobus Henricus van't Hoff. clararse desierto y en otras por la Cada destinatario recibe una medalla, un diploma y situación de guerra mundial y el exi- un premio económico que ha variado a lo largo de los años. -

Liste Der Nobelpreisträger Für Chemie - Wikipedia 16.03.2006 13:25 Uhr
Liste der Nobelpreisträger für Chemie - Wikipedia 16.03.2006 13:25 Uhr Liste der Nobelpreisträger für Chemie aus Wikipedia, der freien Enzyklopädie Diese Liste bietet eine chronologische Übersicht der Träger des Nobelpreises für Chemie mit der Begründung der Nobelkommitees. Eine alphabetische Übersicht bietet die Kategorie Nobelpreisträger für Chemie. 1901 bis 1910 · 1911 bis 1920 · 1921 bis 1930 · 1931 bis 1940 · 1941 bis 1950 · 1951 bis 1960 1961 bis 1970 · 1971 bis 1980 · 1981 bis 1990 · 1991 bis 2000 · 2001 bis 2010 1901-1910 1901 Jacobus Henricus van 't "als Anerkennung des außerordentlichen Verdienstes, das er sich durch die Hoff (NL) Entdeckung der Gesetze der chemischen Dynamik und des osmotischen Druckes in Lösungen erworben hat" 1902 Hermann Emil Fischer "als Anerkennung des außerordentlichen Verdienstes, das er sich durch (DE) seine synthetischen Arbeiten auf dem Gebiet der Zucker- und Puringruppen erworben hat" 1903 August Svante Arrhenius "als Anerkennung des außerordentlichen Verdienstes, das er sich durch (SE) seine Theorie über die elektrolytische Dissoziation um die Entwicklung der Chemie erworben hat" 1904 Sir William Ramsay (UK) "als Anerkennung des Verdienstes, das er sich durch die Entdeckung der indifferenten gasförmigen Grundstoffe in der Luft und die Bestimmung ihres Platzes im periodischen System erworben hat" 1905 Adolf von Baeyer (DE) "als Anerkennung des Verdienstes, das er sich um die Entwicklung der organischen Chemie und der chemischen Industrie durch seine Arbeiten über die organischen Farbstoffe und die -

Curriculum Vitae Prof. Dr. Cyril Norman Hinshelwood
Curriculum Vitae Prof. Dr. Cyril Norman Hinshelwood Name: Cyril Norman Hinshelwood Lebensdaten: 19. Juni 1897 ‐ 9. Oktober 1967 Cyril Norman Hinshelwood war ein britischer Chemiker. Er untersuchte Aspekte der chemischen Reaktionskinetik. Nach ihm ist der Langmuir‐Hinshelwood‐Mechanismus benannt. Er beschreibt die Reaktion zweier Ausgangsstoffe an einer Katalysator‐Oberfläche. Für seine Forschungen über die Mechanismen chemischer Reaktion wurde Hinshelwood 1956 mit dem Nobelpreis für Chemie ausgezeichnet. Akademischer und beruflicher Werdegang Cyril Norman Hinshelwood war sehr begabt und bekam ein Stipendium für die University of Oxford zugesprochen. Wegen des Ersten Weltkriegs konnte er dies jedoch nicht in Anspruch nehmen. Stattdessen war er in dieser Zeit als Chemiker in einer Sprengstofffabrik in Queensferry, Schottland tätig. 1919 ging er ans Balliol College in Oxford, wo er einen durch die Nachkriegszeit verkürzten Kurs in Chemie besuchen durfte. Seine Auffassungsgabe der chemischen Prinzipien war so herausragend, dass er erneut für ein Stipendium vorgeschlagen wurde. In Oxford erlangte Hinshelwood den Bachelor‐ und Master‐Grad. Von 1921 bis 1937 arbeitete er am Trinity College in Oxford als Tutor. 1937 erhielt er eine Professur für Chemie an der Universität Oxford. Auch während des Zweiten Weltkriegs war er für die Kriegsproduktion tätig, so unter anderem bei der Herstellung von Gasmasken und Penicillin. Nobelpreis für Chemie 1956 Die chemische Kinetik, die Geschwindigkeit und Mechanismus einer chemischen Reaktion untersucht, war zu Beginn des 20. Jahrhunderts noch wenig erforscht. Hinshelwood hatte großes Interesse an den Mechanismen derartiger chemischer Prozesse. Es wurde während des Ersten Weltkriegs noch verstärkt, als er in einer Munitionsfabrik beschäftigt war, wo er sich mit der Chemie von explosiven Stoffen beschäftigte. -

Named Equations and Laws in Chemistry (H – K)
Dr. John Andraos, http://www.careerchem.com/NAMED/Named-EQ(H-K).pdf 1 NAMED EQUATIONS AND LAWS IN CHEMISTRY (H – K) © Dr. John Andraos, 2000 - 2011 Department of Chemistry, York University 4700 Keele Street, Toronto, ONTARIO M3J 1P3, CANADA For suggestions, corrections, additional information, and comments please send e-mails to [email protected] http://www.chem.yorku.ca/NAMED/ John Burdon Sanderson Haldane 5 November 1882 - 1 December 1964 British-Scottish, b. Oxford, England Haldane equation Haldane, J.B.S., Enzymes , Longmans, Green, & Co.: London, 1930 Briggs-Haldane solution see George Edward Briggs Biographical References: Daintith, J.; Mitchell, S.; Tootill, E.; Gjersten, D ., Biographical Encyclopedia of Scientists , Institute of Physics Publishing: Bristol, UK, 1994 Millar, D.; Millar, I.; Millar, J.; Millar, M., Chambers Concise Dictionary of Scientists , W. & R. Chambers: Edinburgh, 1989 Pirie, N.W., Biog. Memoirs Fellows Roy. Soc., 1966 , 12 , 219 Louis Plack Hammett 7 April 1894 - 9 February 1987 American, b. Wilmington, Delaware, USA Hammett equation Hammett sigma constants Hammett, L.P., J. Am. Chem. Soc. 1937 , 59 , 96 Hammett, L.P., Chem. Rev. 1935 , 17 , 125 Hammett acidity function Hammett, L.P.; Deyrup, A.J., J. Am. Chem. Soc . 1932 , 54 , 2721 2 Biographical References: Westheimer, F.H., Biog. Memoirs Natl. Acad. Sci. 1997 , 72 , 137 Anon., Chem. Eng. News 1998 , 76 (2), 171 Charles Samuel Hanes 21 May 1903 - 6 July 1990 Canadian, b. Toronto, Ontario, Canada Hanes-Woolf plot Hanes, C.S., Biochem. J. 1932 , 26 , 1406 Woolf's contributions cited in Haldane, J.B.S.; Stern, K.G. Allgemeine Chemie der Enzyme , Steinkopff: Dresden, Leipzig, 1932 though Woolf did not publish his own papers. -

Inhaltsverzeichnis (Textstellen Sind Teilweise Mit Tabellenabschnitten in Den Tafeln Verlinkt)
Inhaltsverzeichnis (Textstellen sind teilweise mit Tabellenabschnitten in den Tafeln verlinkt) GESCHICHTE DER CHEMIE, VOM FEUER BIS ZUR MOLEKULAREN MASCHINE SEITE 001 CHEMIEGESCHICHTE AUS VIELEN BLICKWINKELN SEITE 002 DIE ZEITLICHEN EPOCHEN SEITE 002 UR- UND FRÜHGESCHICHTE SEITE 002 ANTIKE SEITE 003 MITTELALTER (600 -1400) SEITE 004 RENAISSANCE (1400 – 1650) SEITE 007 VON DER ALCHEMIE ZUR MODERNEN CHEMIE (1650-1850) SEITE 008 CHEMIE BIS ZUR MODERNE SEITE 031 ELEMENTARE GESCHICHTEN SEITE 039 AUF ATOMARER EBENE SEITE 045 CHEMISCHE PRODUKTE SEITE 050 DER SCHREI NACH SODA SEITE 050 DAS BLUT DER CHEMIE SEITE 053 BROT UND KRIEG AUS DER LUFT SEITE 055 GELBGRÜNES GIFT SEITE 058 DURCH ZUFALL STOLPERT DAS NEUE IN DIE WELT Seite 063 FARBCHEMIE AM GARTENZAUN SEITE 063 VON EXPLODIERENDEN SCHÜRZEN UND BILLARDKUGELN SEITE 063 DIE DURCHSICHTIGE HAND SEITE 064 SEGENREICHER SCHIMMEL SEITE 066 BICYCLE DAY SEITE 067 LITERATUR SEITE 068 ZEITTAFELN SEITE 070 UR- UND FRÜHGESCHICHTE / AM ANFANG WAR DAS FEUER T01 SEITE 070 ANTIKE (800 V. CHR. – 600 N.CHR.) T01 SEITE 070 Mittelalter (600 – 1400) T01/T02 Seite 071 RENAISSANCE (1400-1650) T02 SEITE 072 VON DER ALCHEMIE ZUR MODERNEN CHEMIE (1650-1850 / AUFKLÄRUNG) T02-T05 SEITE 073 1850 - > MODERNE CHEMIE T05-T14 SEITE 079 ÜBERSICHT 1: VON DER ANTIKE BIS ZUR NEUZEIT SEITE 098 ÜBERSICHT 2: DER WEG IN DIE MODERNE CHEMIE SEITE 099 ERKLÄRUNGEN ZU DEN SYMBOLEN UND FARBEN IN DEN ZEITTAFELN SEITE 100 01 GESCHICHTE DER CHEMIE, VOM FEUER BIS ZUR MOLEKULAREN MASCHINE Der kurze Überblick zur Geschichte der Chemie wurde im Hinblick auf unterschiedliche Aspekte zusammengestellt. Studierende der Chemie lernen im Laufe des Studiums etliche Namensreaktionen und Gleichungen kennen, die Namen von Wissenschaftlern tragen.