Seasonal and Colony Differences in the Foraging Ecology
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Shark Mitigation and Deterrent Measures Submission 64
The Efficacy and Regulation of Shark Mitigation and Deterrent Measures Submission to: Senate Environment and Communications References Committee by Peter Stephenson BSc., ADAS 2815.3, Master Class V February 2017 As a commercial diver and fisherman with over 35 years of diverse experience I write this submission due to my ever-increasing concerns about policies governing management of and research into shark populations. I began snorkelling at the age of 7 and was a keen spear fisherman and surfer for decades although I am currently no longer active in these sports. (partly due to increasing negative shark incidents) I have a BSc. In marine science from Flinders University and have completed a number of years of marine research. Over more than four decades I have spent tens of thousands of hours observing and studying the marine environment. In recent years, particularly after my friends Peter Clarkson and Greg Pickering were attacked by white sharks, I have been researching shark attacks, shark behaviour and the possible factors influencing negative shark/ human interactions. I have also witnessed aggressive shark behaviour first hand but have luckily escaped serious injury…. so far. I currently work as an abalone diver in the South Australian Central Zone Abalone Fishery. THIS IS A MAJOR WORKPLACE SAFETY ISSUE FOR ME! THE BAITING AND HARASSMENT OF SHARKS FOR TOURISM AND SCIENTIFIC RESEARCH Despite legislation deeming the berleying, baiting, approach and harassment of white sharks illegal, governments grant exemptions and licences to tourism operators and scientists to conduct these activities. Despite years of research and observation, the level of conditioning of sharks by repeated berleying and baiting is still poorly understood and documented. -

Three from One = 4000 Magazi
www.mcdoa.org.uk N A V AS MAGAzi totzsin Three from One = 4000 iiiiiiimmommhill111111111111111111111111111111111111111111111101111111111111111111miniiiimnum 11 •_„,,• Siebe Gorman present a now air compressor and cylinder charging decanting set, with an integrated control panel, which can be used for three distinct operations:— To charge large high pressure air storage cylinders to 40001b./sq.in. To decant air from storage cylinders into breathing apparatus or aqualung cylinders. To charge breathing apparatus cylin- ders direct from the compressor. filter and,control panel is mounted In a tubujik.Steel carrying frame and Neptune 4000 weighs-aiiiiroximately 400 lb. It can be Siebe Gorman's new high pressure used independently or incorporated compressor set is designed to provide in a static installation. a versatile unit for charging breathing apparatus or aqualung cylinders with clean, dry air to pressures between ;14,44, 1800 and 4000 p.s.i. Driven by either a `1AN Marineland—see page 9 Ut`, 4 stroke petrol engine or electric 01 ENGLAND -t motor, the air-cooled compressor has For further information, nii, write to 111111111111111141111 1111„i an output of 4.5 cu. ft. of nominal free Siebe Gorman & Co. Ltd., """"""1111111111IM11111111111111111111111 iiiiiiiiiimilimill111191111111111111111111111111111111111111111411 „1040 Neptune Works, Davis Road, F 0,40 air per minute. The complete appara- Chessington, Surrey. -.0.4640 tus, consisting of motor, compressor, Telephone: Lower Hook 8171/8 Printed by Coasby & Co. Ltd., St. James's Road, Southsea, Hai is www.mcdoa.org.uk Vol. 11 No. 1 2/- www.mcdoa.org.uk We specialise in EVERYTHING FOR THE UNDERWATER SPORTSMAN including the latest designs and all the better makes of LUNGS DIVING SUITS SWIMMING GEAR & EQUIPMENT Stainless steel Roles- Oyster, f37. -

Dives of the Bathyscaph Trieste, 1958-1963: Transcriptions of Sixty-One Dictabelt Recordings in the Robert Sinclair Dietz Papers, 1905-1994
Dives of the Bathyscaph Trieste, 1958-1963: Transcriptions of sixty-one dictabelt recordings in the Robert Sinclair Dietz Papers, 1905-1994 from Manuscript Collection MC28 Archives of the Scripps Institution of Oceanography University of California, San Diego La Jolla, California 92093-0219: September 2000 This transcription was made possible with support from the U.S. Naval Undersea Museum 2 TABLE OF CONTENTS INTRODUCTION ...........................................................................................................................4 CASSETTE TAPE 1 (Dietz Dictabelts #1-5) .................................................................................6 #1-5: The Big Dive to 37,800. Piccard dictating, n.d. CASSETTE TAPE 2 (Dietz Dictabelts #6-10) ..............................................................................21 #6: Comments on the Big Dive by Dr. R. Dietz to complete Piccard's description, n.d. #7: On Big Dive, J.P. #2, 4 Mar., n.d. #8: Dive to 37,000 ft., #1, 14 Jan 60 #9-10: Tape just before Big Dive from NGD first part has pieces from Rex and Drew, Jan. 1960 CASSETTE TAPE 3 (Dietz Dictabelts #11-14) ............................................................................30 #11-14: Dietz, n.d. CASSETTE TAPE 4 (Dietz Dictabelts #15-18) ............................................................................39 #15-16: Dive #61 J. Piccard and Dr. A. Rechnitzer, depth of 18,000 ft., Piccard dictating, n.d. #17-18: Dive #64, 24,000 ft., Piccard, n.d. CASSETTE TAPE 5 (Dietz Dictabelts #19-22) ............................................................................48 #19-20: Dive Log, n.d. #21: Dr. Dietz on the bathysonde, n.d. #22: from J. Piccard, 14 July 1960 CASSETTE TAPE 6 (Dietz Dictabelts #23-25) ............................................................................57 #23-25: Italian Dive, Dietz, Mar 8, n.d. CASSETTE TAPE 7 (Dietz Dictabelts #26-29) ............................................................................64 #26-28: Italian Dive, Dietz, n.d. -

Elasmobranch Biodiversity, Conservation and Management Proceedings of the International Seminar and Workshop, Sabah, Malaysia, July 1997
The IUCN Species Survival Commission Elasmobranch Biodiversity, Conservation and Management Proceedings of the International Seminar and Workshop, Sabah, Malaysia, July 1997 Edited by Sarah L. Fowler, Tim M. Reed and Frances A. Dipper Occasional Paper of the IUCN Species Survival Commission No. 25 IUCN The World Conservation Union Donors to the SSC Conservation Communications Programme and Elasmobranch Biodiversity, Conservation and Management: Proceedings of the International Seminar and Workshop, Sabah, Malaysia, July 1997 The IUCN/Species Survival Commission is committed to communicate important species conservation information to natural resource managers, decision-makers and others whose actions affect the conservation of biodiversity. The SSC's Action Plans, Occasional Papers, newsletter Species and other publications are supported by a wide variety of generous donors including: The Sultanate of Oman established the Peter Scott IUCN/SSC Action Plan Fund in 1990. The Fund supports Action Plan development and implementation. To date, more than 80 grants have been made from the Fund to SSC Specialist Groups. The SSC is grateful to the Sultanate of Oman for its confidence in and support for species conservation worldwide. The Council of Agriculture (COA), Taiwan has awarded major grants to the SSC's Wildlife Trade Programme and Conservation Communications Programme. This support has enabled SSC to continue its valuable technical advisory service to the Parties to CITES as well as to the larger global conservation community. Among other responsibilities, the COA is in charge of matters concerning the designation and management of nature reserves, conservation of wildlife and their habitats, conservation of natural landscapes, coordination of law enforcement efforts as well as promotion of conservation education, research and international cooperation. -

080058-89.02.017.Pdf
t9l .Ig6I pup spu?Fr rr"rl?r1mv qnos raq1oaqt dq panqs tou pus 916I uao^\teq sluauennboJ puu surelqord lusue8suuur 1eneds wq sauo8a uc .fu1mpw snorru,r aql uI luar&(oldua ',uq .(tg6l a;oJareqt puuls1oore8ueltr 0t dpo 1u reted lS sr ur saiuzqc aql s,roqs osIB elqeJ srqt usrmoJ 'urpilsny 'V'S) puels tseSrul geu oq; ur 1sa?re1p4ql aql 3o luetugedeq Z alq?J rrr rtr\oqs su padoldua puu prr"lsr aroqsJJorrprJpnsnv qlnos lsJErel aql ruJ ere,u eldoed ZS9 I feqf pa roqs snsseC srllsll?ls dq r ?olp sp qlr^\ puplsl oorp8ue1 Jo neomg u"rl?Jlsnv eql uo{ sorn8g luereJ lsour ,u1 0g€ t Jo eW T86I ul puu 00S € ,{lel?urxorddu sr uoqelndod '(derd ur '8ur,no:8 7r luosaJd eqJ petec pue uoqcnpord ,a uosurqoU) uoqecrJqndro3 peredard Smeq ,{11uermc '(tg6l )potsa^q roJ perualo Suraq puel$ oql Jo qJnu eru sda,r-rnsaseql3o EFSer pa[elop aqJ &usJ qlvrr pedolaaap fuouoce Surqsg puu Surure; e puu pue uosurqo;) pegoder useq aleq pesn spoporu palles-er su,rr prrqsr eql sreo,{ Eurpeet:ns eq1 re,ro prru s{nser druuruqord aqt pue (puep1 ooreSuqtr rnq 698I uI peuopwqs sE^\ elrs lrrrod seaeell eql SumnJcxa) sprrelsl aroqsJJo u"{e4snv qlnos '998I raqueJeo IIl eprelepv Jo tuaruslDesIeuroJ oqt aql Jo lsou uo palelduor ueeq A\ou aaeq s,{e,rrns aro3aq ,(ueduo3 rr"4u4snv qtnos qtgf 'oAE eqt dq ,{nt p:6o1org sree,{009 6 ol 000 L uea r1aq palulosr ur slors8rry1 u,no1paserd 6rll J?eu salaed 'o?e ;o lrrrod ererrirspuulsr Surura sr 3ql Jo dllJolpu aq; sree,{ paqslqplse peuuurad lE s?a\lueurep1os uuedorng y 009 0I spuplsl dpearg pue uosr"ed pup o8e srea,{ 'seruolocuorJ-"3s rel?l puB -
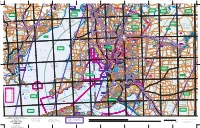
S P E N C E R G U L F S T G U L F V I N C E N T Adelaide
Yatala Harbour Paratoo Hill Turkey 1640 Sunset Hill Pekina Hill Mt Grainger Nackara Hill 1296 Katunga Booleroo "Avonlea" 2297 Depot Hill Creek 2133 Wilcherry Hill 975 Roopena 1844 Grampus Hill Anabama East Hut 1001 Dawson 1182 660 Mt Remarkable SOUTH Mount 2169 440 660 (salt) Mt Robert Grainger Scobie Hill "Mazar" vermin 3160 2264 "Manunda" Wirrigenda Hill Weednanna Hill Mt Whyalla Melrose Black Rock Goldfield 827 "Buckleboo" 893 729 Mambray Creek 2133 "Wyoming" salt (2658±) RANGE Pekina Wheal Bassett Mine 1001 765 Station Hill Creek Manunda 1073 proof 1477 Cooyerdoo Hill Maurice Hill 2566 Morowie Hill Nackara (abandoned) "Bulyninnie" "Oak Park" "Kimberley" "Wilcherry" LAKE "Budgeree" fence GILLES Booleroo Oratan Rock 417 Yeltanna Hill Centre Oodla "Hill Grange" Plain 1431 "Gilles Downs" Wirra Hillgrange 1073 B pipeline "Wattle Grove" O Tcharkuldu Hill T Fullerville "Tiverton 942 E HWY Outstation" N Backy Pt "Old Manunda" 276 E pumping station L substation Tregalana Baroota Yatina L Fitzgerald Bay A Middleback Murray Town 2097 water Ucolta "Pitcairn" E Buckleboo 1306 G 315 water AN Wild Dog Hill salt Tarcowie R Iron Peak "Terrananya" Cunyarie Moseley Nobs "Middleback" 1900 works (1900±) 1234 "Lilydale" H False Bay substation Yaninee I Stoney Hill O L PETERBOROUGH "Blue Hills" LC L HWY Point Lowly PEKINA A 378 S Iron Prince Mine Black Pt Lancelot RANGE (2294±) 1228 PU 499 Corrobinnie Hill 965 Iron Baron "Oakvale" Wudinna Hill 689 Cortlinye "Kimboo" Iron Baron Waite Hill "Loch Lilly" 857 "Pualco" pipeline Mt Nadjuri 499 Pinbong 1244 Iron -

How Solitary Are White Sharks: Social Interactions Or Just Spatial Proximity?
Behav Ecol Sociobiol DOI 10.1007/s00265-016-2179-y ORIGINAL ARTICLE How solitary are white sharks: social interactions or just spatial proximity? R. Findlay1 & E. Gennari2,3 & M. Cantor1 & D. P. Tittensor 1,4 Received: 23 October 2015 /Revised: 24 June 2016 /Accepted: 28 June 2016 # Springer-Verlag Berlin Heidelberg 2016 Abstract the evidence that large pelagic shark species are generally White sharks (Carcharodon carcharias) are circumglobally solitary and display limited social behaviour. distributed large apex predators. While ecologically impor- tant, there is very limited study of their social behaviour. Significance statement Although evident in other large, apex marine predators (e.g. Large pelagic shark species are important top predators, but toothed whales) and smaller elasmobranchs (e.g. blacktip reef we know little about their social behaviour. We tested the sharks), the ability of any large pelagic elasmobranch to dem- ability of white sharks (C. carcharias) to form groups and onstrate social preferences, tolerance or grouping behaviour is display social preferences for other individuals when they largely unknown. Here, we test whether white sharks in a congregate at scavenging events in a coastal environment, near-coastal environment form non-random associations with where social interactions may be more likely. We found that other conspecifics or simply share the same space at the same white sharks co-occur at random, displaying no preferred or time. We photo-identified 323 individuals—74 % juvenile avoided associations for other individuals. Nevertheless, there females (175–300 cm)—during chumming events at six dif- was a minor influence of biological traits, with individuals ferent sites in Mossel Bay, South Africa, over a 6-year period aggregating according to gender and, possibly, body size. -

Nswdpigame Fish Tagging Program
NSW DPI GAME FISH TAGGING PROGRAM REPORT 2017-2018 Introduction .......................................................................................................................................... 5 Figure 1. Number of fish recaptured by year, 2017/18. .................................................................................... 5 The Program to date ............................................................................................................................. 6 Species summary of tagging activity for 2017/18 ................................................................................ 7 Black marlin ....................................................................................................................................................... 9 Southern bluefin tuna ..................................................................................................................................... 10 Blue marlin ...................................................................................................................................................... 10 Striped marlin .................................................................................................................................................. 11 Sailfish .............................................................................................................................................................. 11 Yellowfin tuna ................................................................................................................................................. -

Australian Sea Lions Neophoca Cinerea at Colonies in South Australia: Distribution and Abundance, 2004 to 2008
The following supplement accompanies the article Australian sea lions Neophoca cinerea at colonies in South Australia: distribution and abundance, 2004 to 2008 Peter D. Shaughnessy1,*, Simon D. Goldsworthy2, Derek J. Hamer3,5, Brad Page2, Rebecca R. McIntosh4 1South Australian Museum, North Terrace, Adelaide, South Australia 5000, Australia 2South Australian Research and Development Institute, PO Box 120, Henley Beach, South Australia 5022, Australia 3Department of Earth and Environmental Science, University of Adelaide, Adelaide, South Australia 5005, Australia 4Department of Zoology, La Trobe University, Bundoora, Victoria 3068, Australia 5Present address: Australian Antarctic Division, 203 Channel Highway, Kingston, Tasmania 7050, Australia *Email: [email protected] Endangered Species Research 13: 87–98 (2011) Supplement. Information on 26 Neophoca cinerea breeding colonies in South Australia The Australian sea lion Neophoca cinerea is restricted to South Australia and Western Australia. This supplementary material provides information on 26 breeding colonies in South Australia that were visited during this study between 2004 and 2008, with details on pup population estimates from which best estimates are summarized in Table 1 of the main paper. It also summarises data on pup counts available before 2004. Mark-recapture estimates are presented with their 95% confidence limits (CL). Detailed counts of all animals in these colonies have been presented in consultancy reports. Data for the other 13 breeding colonies and 9 haulout sites with occasional pupping (which were not visited in this study) were taken from published literature. In addition, 24 haulout sites of the Australian sea lion visited during the study are listed in Table S1 of this supplementary material, together with their geographical positions and counts of sea lions seen on the dates visited. -

HYDROGRAPHIC DEPARTMENT Charts, 1769-1824 Reel M406
AUSTRALIAN JOINT COPYING PROJECT HYDROGRAPHIC DEPARTMENT Charts, 1769-1824 Reel M406 Hydrographic Department Ministry of Defence Taunton, Somerset TA1 2DN National Library of Australia State Library of New South Wales Copied: 1987 1 HISTORICAL NOTE The Hydrographical Office of the Admiralty was created by an Order-in-Council of 12 August 1795 which stated that it would be responsible for ‘the care of such charts, as are now in the office, or may hereafter be deposited’ and for ‘collecting and compiling all information requisite for improving Navigation, for the guidance of the commanders of His Majesty’s ships’. Alexander Dalrymple, who had been Hydrographer to the East India Company since 1799, was appointed the first Hydrographer. In 1797 the Hydrographer’s staff comprised an assistant, a draughtsman, three engravers and a printer. It remained a small office for much of the nineteenth century. Nevertheless, under Captain Thomas Hurd, who succeeded Dalrymple as Hydrographer in 1808, a regular series of marine charts were produced and in 1814 the first surveying vessels were commissioned. The first Catalogue of Admiralty Charts appeared in 1825. In 1817 the Australian-born navigator Phillip Parker King was supplied with instruments by the Hydrographic Department which he used on his surveying voyages on the Mermaid and the Bathurst. Archives of the Hydrographic Department The Australian Joint Copying Project microfilmed a considerable quantity of the written records of the Hydrographic Department. They include letters, reports, sailing directions, remark books, extracts from logs, minute books and survey data books, mostly dating from 1779 to 1918. They can be found on reels M2318-37 and M2436-67. -

Conserving Marine Biodiversity in South Australia - Part 1 - Background, Status and Review of Approach to Marine Biodiversity Conservation in South Australia
Conserving Marine Biodiversity in South Australia - Part 1 - Background, Status and Review of Approach to Marine Biodiversity Conservation in South Australia K S Edyvane May 1999 ISBN 0 7308 5237 7 No 38 The recommendations given in this publication are based on the best available information at the time of writing. The South Australian Research and Development Institute (SARDI) makes no warranty of any kind expressed or implied concerning the use of technology mentioned in this publication. © SARDI. This work is copyright. Apart of any use as permitted under the Copyright Act 1968, no part may be reproduced by any process without prior written permission from the publisher. SARDI is a group of the Department of Primary Industries and Resources CONTENTS – PART ONE PAGE CONTENTS NUMBER INTRODUCTION 1. Introduction…………………………………..…………………………………………………………1 1.1 The ‘Unique South’ – Southern Australia’s Temperate Marine Biota…………………………….…….1 1.2 1.2 The Status of Marine Protected Areas in Southern Australia………………………………….4 2 South Australia’s Marine Ecosystems and Biodiversity……………………………………………..9 2.1 Oceans, Gulfs and Estuaries – South Australia’s Oceanographic Environments……………………….9 2.1.1 Productivity…………………………………………………………………………………….9 2.1.2 Estuaries………………………………………………………………………………………..9 2.2 Rocky Cliffs and Gulfs, to Mangrove Shores -South Australia’s Coastal Environments………………………………………………………………13 2.2.1 Offshore Islands………………………………………………………………………………14 2.2.2 Gulf Ecosystems………………………………………………………………………………14 2.2.3 Northern Spencer Gulf………………………………………………………………………...14 -

Mair87 001.Pdf
Departmental investigation into the grounding of the Singaporean bulk carrier SEA CRANE on Tiparra Reef, Spencer Gulf on 2 December 1995 Report No 87 Contents Summary ....................................................................... 1 Sources of Information ................................................ 2 Narrative ....................................................................... 3 Comment and Analysis ............................................. 13 Conclusions................................................................ 19 Submissions ............................................................... 21 ADMIRALTY SAILING DIRECTIONS VOLUME 13 .... 22 Navigation Act 1912 Navigation (Marine Casualty) Regulations investigation into the grounding of the Singaporean bulk carrier SEA CRANE on Tiparra Reef, Spencer Gulf on 2 December 1995 No 87 Published: June 1996 ISBN 0 642 19971 X To increase the value of the safety material presented in this report, readers are encouraged to copy or reprint the material in part or in whole for further distribution, but should acknowledge the source. Additional copies of the report can be obtained from: Inspector of Marine Accidents Marine Incident Investigation Unit Department of Transport and Regional Development P O Box 594 CANBERRA ACT 2601 Phone: 06 274 7324 Fax: 06 274 6699 Email:[email protected] MIIU on the INTERNET Information relating to this report and other marine investigation reports can be located from the Marine Incident Investigation Unit's Internet homepage at our URL: http://www.dot.gov.au/programs/miiu/miiuhome.htm Summary Shortly after 0900 on Saturday, 2 December 1995, the Singapore registered bulk carrier Sea Crane, while bound for the port of Wallaroo to load a part cargo of barley, grounded off Tiparra Reef in the Spencer Gulf, South Australia. The water ballast in the upper wing tanks was dumped and the vessel refloated after about 1½ hours under its own power.