Radium-226 (226Ra)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
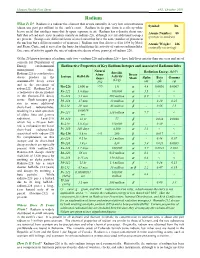
Radium What Is It? Radium Is a Radioactive Element That Occurs Naturally in Very Low Concentrations Symbol: Ra (About One Part Per Trillion) in the Earth’S Crust
Human Health Fact Sheet ANL, October 2001 Radium What Is It? Radium is a radioactive element that occurs naturally in very low concentrations Symbol: Ra (about one part per trillion) in the earth’s crust. Radium in its pure form is a silvery-white heavy metal that oxidizes immediately upon exposure to air. Radium has a density about one- Atomic Number: 88 half that of lead and exists in nature mainly as radium-226, although several additional isotopes (protons in nucleus) are present. (Isotopes are different forms of an element that have the same number of protons in the nucleus but a different number of neutrons.) Radium was first discovered in 1898 by Marie Atomic Weight: 226 and Pierre Curie, and it served as the basis for identifying the activity of various radionuclides. (naturally occurring) One curie of activity equals the rate of radioactive decay of one gram (g) of radium-226. Of the 25 known isotopes of radium, only two – radium-226 and radium-228 – have half-lives greater than one year and are of concern for Department of Energy environmental Radioactive Properties of Key Radium Isotopes and Associated Radionuclides management sites. Natural Specific Radiation Energy (MeV) Radium-226 is a radioactive Abun- Decay Isotope Half-Life Activity decay product in the dance Mode Alpha Beta Gamma (Ci/g) uranium-238 decay series (%) (α) (β) (γ) and is the precursor of Ra-226 1,600 yr >99 1.0 α 4.8 0.0036 0.0067 radon-222. Radium-228 is a radioactive decay product Rn-222 3.8 days 160,000 α 5.5 < < in the thorium-232 decay Po-218 3.1 min 290 million α 6.0 < < series. -
![(12) United States Patent (10) Patent N0.: US 7,887,782 B2 Schwarz Et A]](https://docslib.b-cdn.net/cover/9007/12-united-states-patent-10-patent-n0-us-7-887-782-b2-schwarz-et-a-89007.webp)
(12) United States Patent (10) Patent N0.: US 7,887,782 B2 Schwarz Et A]
US007887782B2 (12) United States Patent (10) Patent N0.: US 7,887,782 B2 Schwarz et a]. (45) Date of Patent: Feb. 15, 2011 (54) RADIOTHERAPEUTIC FORMULATIONS 4,859,431 A * 8/1989 Ehrhardt ............. .. 250/432 PD CONTAINING 224RA AND A METHOD FOR 5,457,323 A 10/1995 Geerlings THEIR PRODUCTION 5,854,968 A * 12/1998 Horwitz et a1. .............. .. 423/2 (75) Inventors: UWe SchWarz, Salzgitter (DE); Rolf Daniels, Salzgitter (DE) OTHER PUBLICATIONS Delikan, O. Health Phys. 1978, 35, 21-24.* (73) Assignee: Altmann Therapie GmbH & Co., Narbutt et al. (Appl. Radiat. lsot. 1998, 49, 89-91).* Salzgitter (DE) Orhan Delikan; Preparation of 224Ra for Therapy of Ankylosing Sp0ndylitis*; Health Physics Vol. 35 (July) pp. 21-24 Pergamon ( * ) Notice: Subject to any disclaimer, the term of this Press Ltd., 1978. patent is extended or adjusted under 35 Radioaktives Arzneimittel; Gebrauchsinformation und Fachinforma U.S.C. 154(b) by 671 days. tion; pp. 1-6, N0 English translation. Alberding, A. et al., “Wirksamkeit und Vertraglichkeit von (21) Appl. No.: 10/362,049 Radiumchlorid in der Behandlung der ankylo sierenden Spondylitis,” Z Rheumatol 2006, 65:245-251. (22) PCT Filed: Aug. 7, 2001 * cited by examiner (86) PCT No.: PCT/EP01/09121 Primary ExamineriMichael G Hartley Assistant ExamineriMelissa Perreira § 371 (0X1)’ (74) Attorney, Agent, or FirmiSam K. Tahmassebi; (2), (4) Date: Jul. 9, 2003 TechLaW LLP (87) PCT Pub. No.: WO02/15943 (57) ABSTRACT PCT Pub. Date: Feb. 28, 2002 The invention relates to novel radiotherapeutic formulations (65) Prior Publication Data containing 224Ra and methods for their production. The US 2004/0028606 A1 Feb. -

Pp-03-25-New Dots.Qxd 10/23/02 2:41 PM Page 778
pp-03-25-new dots.qxd 10/23/02 2:41 PM Page 778 778 PRAESODYMIUM PRAESODYMIUM [7440–10–0] Symbol Pr; atomic number 59; atomic weight 140.908; a lanthanide–series rare earth element; belongs to the cerium group of rare earths; electron con- figuration [Xe] 4f36s2; partially filled f subshell; valence states +3, +4; most 3+ stable oxidation state +3; electrode potential E°/V (aq) for Pr + 3e¯ ↔ Pr is –2.35 V; atomic radius 1.828 Å; first ionization potential 5.46 eV; one natu- rally–occurring isotope, Pr–141; twenty–nine artificial radioactive isotopes known in the mass range 124, 126–140 and 142–154; the longest–lived isotope Pr–143, t1/2 13.57 day, and the shortest–lived isotope Pr–124, t1/2 1.2 second. History, Occurrence, and Uses Mosander extracted from the mineral lanthana a rare earth fraction, named didymia in 1841. In 1879, Boisbaudran separated a rare earth oxide called samaria (samarium oxide) from the didymia fraction obtained from the mineral samarskite. Soon after that in 1885, Baron Auer von Welsbach iso- lated two other rare earths from didymia. He named them as praseodymia (green twin) and neodymia (new twin) after their source didymia (twin). The name praseodymium finally was assigned to this new element, derived from the two Greek words, prasios meaning green and didymos meaning twin. Praseodymium occurs in nature associated with other rare earths in a rel- atively high abundance. It is more abundant than some common metals such as silver, gold, or antimony. The average concentration of this metal in the earth’s crust is estimated to be 8.2 mg/kg. -

The Use of Cosmic-Rays in Detecting Illicit Nuclear Materials
The use of Cosmic-Rays in Detecting Illicit Nuclear Materials Timothy Benjamin Blackwell Department of Physics and Astronomy University of Sheffield This dissertation is submitted for the degree of Doctor of Philosophy 05/05/2015 Declaration I hereby declare that except where specific reference is made to the work of others, the contents of this dissertation are original and have not been submitted in whole or in part for consideration for any other degree or qualification in this, or any other Univer- sity. This dissertation is the result of my own work and includes nothing which is the outcome of work done in collaboration, except where specifically indicated in the text. Timothy Benjamin Blackwell 05/05/2015 Acknowledgements This thesis could not have been completed without the tremendous support of many people. Firstly I would like to express special appreciation and thanks to my academic supervisor, Dr Vitaly A. Kudryavtsev for his expertise, understanding and encourage- ment. I have enjoyed our many discussions concerning my research topic throughout the PhD journey. I would also like to thank my viva examiners, Professor Lee Thomp- son and Dr Chris Steer, for the time you have taken out of your schedules, so that I may take this next step in my career. Thanks must also be given to Professor Francis Liven, Professor Neil Hyatt and the rest of the Nuclear FiRST DTC team, for the initial oppor- tunity to pursue postgraduate research. Appreciation is also given to the University of Sheffield, the HEP group and EPRSC for providing me with the facilities and funding, during this work. -

On the Solubility of Radium Sulfate and Carbonate
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Chalmers Publication Library THESIS FOR THE DEGREE OF LICENTIATE OF ENGINEERING On the solubility of radium sulfate and carbonate Artem Vasilyevich Matyskin Nuclear Chemistry Department of Chemistry and Chemical Engineering CHALMERS UNIVERSITY OF TECHNOLOGY Gothenburg, Sweden 2016 On the solubility of radium sulfate and carbonate © ARTEM VASILYEVICH MATYSKIN, 2016 Technical report number: 2016:04 ISSN number: 1652-943X Nuclear Chemistry Department of Chemistry and Chemical Engineering Chalmers University of Technology SE – 412 96 Göteborg Sweden Telephone +46(0)31-772 1000 Cover: Radium sulfate powder Chalmers Reproservice Gothenburg, Sweden 2016 On the solubility of radium sulfate and carbonate Artem V. Matyskin Nuclear Chemistry Department of Chemistry and Chemical Engineering Chalmers University of Technology Abstract Radium is one of the most toxic elements and its concentration in different human activities and migration from man-made wastes provokes a strong interest in environmental science. To be able to model the migration process, reliable experimental thermodynamic data of radium compounds are needed. In this work details of the safe radium source disassembly which were previously used in brachytherapy are described and different methods for conversion of RaSO4 into aqueous solution are reviewed. The method of choice included three cycles of RaSO4 heating in 1.5 M Na2CO3 up to 85 ºC, cooling and subsequent removal of supernatant. X-ray diffraction studies showed that the method allows the synthesis of amorphous RaCO3, which can be dissolved in mineral acid. Gamma spectrometric measurements showed that most of the initial RaSO4 was 210 converted into solution and that 7 ± 1 % of the initial Pb was co-precipitated with RaCO3. -

A Therapy with 223Ra-Dichloride
Journal of Nuclear Medicine, published on December 16, 2013 as doi:10.2967/jnumed.112.112482 CONTINUING EDUCATION Bone-Seeking Radiopharmaceuticals for Treatment of Osseous Metastases, Part 1: a Therapy with 223Ra-Dichloride Neeta Pandit-Taskar, Steven M. Larson, and Jorge A. Carrasquillo Molecular Imaging and Therapy Service, Memorial Sloan-Kettering Cancer Center, New York, New York Learning Objectives: On successful completion of this activity, participants should be able to (1) define the advantages and disadvantages of the use of a-emitting radionuclide 223Ra-dichloride in the treatment of painful metastatic osseous metastasis; (2) recognize the indications and contraindications and define the prerequisites for administration of 223Ra-dichloride; and (3) apply and integrate the treatment of osseous metastasis with 223Ra-dichloride in routine clinical practice. Financial Disclosure: The authors of this article have indicated no relevant relationships that could be perceived as a real or apparent conflict of interest. CME Credit: SNMMI is accredited by the Accreditation Council for Continuing Medical Education (ACCME) to sponsor continuing education for physicians. SNMMI designates each JNM continuing education article for a maximum of 2.0 AMA PRA Category 1 Credits. Physicians should claim only credit commensurate with the extent of their participation in the activity. For CE credit, participants can access this activity through the SNMMI Web site (http:// www.snmmi.org/ce_online) through February 2017. and hypercalcemia. These cause significant morbidity and affect Metastatic disease to bone is commonly seen in the advanced performance status and quality of life. The effect of bone metastasis stages of many cancers. The cardinal symptom, pain, is often the is related to altered signals and balance between osteoclastic and cause of significant morbidity and reduced quality of life. -

Effects of Intravenous Injections of Radium Bromide. by R
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by PubMed Central EFFECTS OF INTRAVENOUS INJECTIONS OF RADIUM BROMIDE. BY R. BURTON-OPITZ AND GUSTAVE M. MEYER. (From the Laboratories of Physiology and Physiological Chemistry of Colum- bia University, at the College of Physicians and Surgeons, New York.) PLATE XVI. The present study was undertaken with a view of determining in a general way the effects of intravenous administration of ra- dium upon the circulation and respiration. The problem was sug- gested to us by Dr. William J. Gies, under whose guidance a number of researches, dealing with the more extensive question of the action of radium upon animal and vegetable cells, have re- cently been carried on in the laboratories of Columbia University.1 For the radium used in these experiments we are greatly in- debted to Mr. Hugo Lieber. It was supplied to us in the form of the bromide, in preparations of 240 , iooo, and io,ooo activities. The strength of the solution used was the same in all cases. It contained 45 rag. of the dry substance in 25 c. c. of the solvent; each cubic centimeter of the solution, therefore, contained 1.8 rag. of the radium preparation. The amount of the radium present varies directly with the ra- dio-activity. Preparations of ~,5oo,ooo activity are said to repre- sent pure radium bromide3 Taking this figure as the standard of purity, ~ .8 rag. of the radium preparation of io,ooo activity contained approximately only o.o~ 26 mg., the same quantity of the preparation of ~ooo activity contained o.ooi26 rag. -

Two-Proton Radioactivity 2
Two-proton radioactivity Bertram Blank ‡ and Marek P loszajczak † ‡ Centre d’Etudes Nucl´eaires de Bordeaux-Gradignan - Universit´eBordeaux I - CNRS/IN2P3, Chemin du Solarium, B.P. 120, 33175 Gradignan Cedex, France † Grand Acc´el´erateur National d’Ions Lourds (GANIL), CEA/DSM-CNRS/IN2P3, BP 55027, 14076 Caen Cedex 05, France Abstract. In the first part of this review, experimental results which lead to the discovery of two-proton radioactivity are examined. Beyond two-proton emission from nuclear ground states, we also discuss experimental studies of two-proton emission from excited states populated either by nuclear β decay or by inelastic reactions. In the second part, we review the modern theory of two-proton radioactivity. An outlook to future experimental studies and theoretical developments will conclude this review. PACS numbers: 23.50.+z, 21.10.Tg, 21.60.-n, 24.10.-i Submitted to: Rep. Prog. Phys. Version: 17 December 2013 arXiv:0709.3797v2 [nucl-ex] 23 Apr 2008 Two-proton radioactivity 2 1. Introduction Atomic nuclei are made of two distinct particles, the protons and the neutrons. These nucleons constitute more than 99.95% of the mass of an atom. In order to form a stable atomic nucleus, a subtle equilibrium between the number of protons and neutrons has to be respected. This condition is fulfilled for 259 different combinations of protons and neutrons. These nuclei can be found on Earth. In addition, 26 nuclei form a quasi stable configuration, i.e. they decay with a half-life comparable or longer than the age of the Earth and are therefore still present on Earth. -

Women's History Magazine Broadly As Possible
Women’s History Magazine Issue 48, AUTUMN 2004 Issn 1476-6760 Themed Issue: Science, Technology and Education Maria Rentetzi on The Case of the Radium Dial Painters Claire Jones on Grace Chisholm Young in Turn-of-Nineteenth-Century Germany Joyce Goodman on Technical Education, Female Emigration and Nation Building Hull 2004 Conference Report Launch of new WHN Book Prize Clare Evans Prize — Report on 2004 Award and Hypatia Announcement of of Alexandria 2005 Competition c. 370 - 415 Conference 2005 — Mathematician philosopher, Call for Papers teacher and inventor of mechanical devices. th 14 Conference of the Women’s History Network Women, Art and Culture: Historical Perspectives September 2nd- 4th 2005, Southampton Southampton Institute, Sir James Matthews Conference Centre, Southampton, Hants. Papers are welcomed on the following themes: Women and the visual arts; painting, sculpture, architecture, and the decorative arts. Women and the Arts and Crafts Movement/Home Decorating. Women and the performing arts. Women and the literary arts. Women as art objects/images of women. Women as mediators of culture. Women as collectors and benefactors. Plenary Speakers: Frances Borzello on 'Women Artists: Self Portraits' Marina Vaizey on '20th Century Women Collectors' Speakers, papers and a provisional programme will be posted at www.womenshistorynetwork.org as soon as they become available. Papers will be considered for special issues of: Women’s History Magazine & Women’s History Review. Abstracts of 200-300 words should be sent by 30/03/05 to: Dr Anne Anderson, FMAS, Southampton Institute, Southampton, S014 ORF. [email protected] Administrator: Dr. Joyce A. Walker (Women’s History Network) , Department of History, University of Aberdeen, Crombie Annexe, Meston Walk, Old Aberdeen, AB24 3FX E-mail: [email protected] The arrival of this Autumn's Women's History Magazine broadly as possible. -

Bone-Seeking Radiopharmaceuticals for Treatment of Osseous Metastases, Part 1: a Therapy with 223Ra-Dichloride
Downloaded from jnm.snmjournals.org by on June 26, 2020. For personal use only. CONTINUING EDUCATION Bone-Seeking Radiopharmaceuticals for Treatment of Osseous Metastases, Part 1: a Therapy with 223Ra-Dichloride Neeta Pandit-Taskar, Steven M. Larson, and Jorge A. Carrasquillo Molecular Imaging and Therapy Service, Memorial Sloan-Kettering Cancer Center, New York, New York Learning Objectives: On successful completion of this activity, participants should be able to (1) define the advantages and disadvantages of the use of a-emitting radionuclide 223Ra-dichloride in the treatment of painful metastatic osseous metastasis; (2) recognize the indications and contraindications and define the prerequisites for administration of 223Ra-dichloride; and (3) apply and integrate the treatment of osseous metastasis with 223Ra-dichloride in routine clinical practice. Financial Disclosure: Dr. Carrasquillo is a consultant/advisor for Bayer and Algeta. Dr. Larson is on the advisory board of ImaginAb, Molecular Imaging, and Perceptive Informatics; is a scientific advisor for Millennium and Progenics Pharmaceuticals, Inc., is a board member and the Chief Medical Officer of Claymore Technologies LLC; and has other ownership interest in Clinical Silica Technologies. The authors of this article have indicated no other relevant relationships that could be perceived as a real or apparent conflict of interest. CME Credit: SNMMI is accredited by the Accreditation Council for Continuing Medical Education (ACCME) to sponsor continuing education for physicians. SNMMI designates each JNM continuing education article for a maximum of 2.0 AMA PRA Category 1 Credits. Physicians should claim only credit commensurate with the extent of their participation in the activity. For CE credit, participants can access this activity through the SNMMI Web site (http:// www.snmmi.org/ce_online) through February 2017. -

Preparation of New Solution Standards of Radium W
Journal of Research of the National Burea u of Sta nda rds Vol. 62, No.1, January 1959 Resea rch Paper 2924 Preparation of New Solution Standards of Radium W. B. Mann, L. L. Stockmann, W . J. Youden, A. Schwebel, P. A. Mullen, and S. B. Garfinkel New radium-solut ion standards have been prepa red in t he r a nges of 10 micrograms a nd also 10- 9 and 10- 11 gram of r adium element. These h a ve been compa red with thc Nationa l Bureau of Standard's 1940 and 1947 series of radium-solut ion standards and, as a result of t hese compar isons, it has been found t hat t he 1940 10- 9 a nd 10- 11 -gram solut ion standa rds contained som e 2 to 3 percent more r adium element t han cer tified. It has been sho wn t hat t his difference pro ba bly arose in t he dilutio n of t he 1940 standa rds. 1. Introduction out using the NBS radiation balance [4 , 5) . These measurements consisted of three m easurem ents of R adium-solution standards have previously been the rate of energy emission from the new radium prepared at. the National Bureau of Standards in 1940 som'ce alone and also one triad of m easurem ents and 1947. The 1940 series consisted of standards in [1 , 2) of the new source relative to both primary the microgram range, ranging from 0.1 to 100.0 J.1.g of standards (No. -

"Indoor Radon and Radon Decay Product Measurement Device Protocols"
"Indoor Radon and Radon Decay Product Measurement Device Protocols" U.S. Environmental Protection Agency Office of Air and Radiation (6604J) EPA 402-R-92-004, July 1992 (revised) Table of Contents Disclaimer Acknowledgements Significant Changes in This Revision Radon and Radon Decay Product Measurement Methods Section 1: General Considerations 1.1 Introduction and Background 1.2 General Guidance on Measurement Strategy, Measurement Conditions, Device Location Selection, and Documentation 1.3 Quality Assurance Section 2: Indoor Radon Measurement Device Protocols 2.1 Protocol for Using Continuous Radon Monitors (CR) to Measure Indoor Radon Concentrations 2.2 Protocol for Using Alpha Track Detectors (AT or ATD) to Measure Indoor Radon Concentrations 2.4 Protocol for Using Activated Charcoal Adsorption Devices (AC) to Measure Indoor Radon Concentrations 2.5 Protocol for Using Charcoal Liquid Scintillation (LS) Devices to Measure Indoor Radon Concentrations 2.6 Protocol for Using Grab Radon Sampling (GB, GC, GS), Pump/Collapsible Bag Devices (PB), and Three-Day Integrating Evacuated Scintillation Cells (SC) to Measure Indoor Radon Concentrations 2.7 Interim Protocol for Using Unfiltered Track Detectors (UT) to Measure Indoor Radon Concentrations Section 3: Indoor Radon Decay Product Measurement Device Protocols 3.1 Protocol for Using Continuous Working Level Monitors (CW) to Measure Indoor Radon Decay Product Concentrations 3.2 Protocol for Using Radon Progeny Integrating Sampling Units (RPISU or RP) to Measure Indoor Radon Decay Product Concentrations 3.3 Protocol for Using Grab Sampling-Working Level (GW) to Measure Indoor Radon Decay Product Concentrations Glossary References Please Note: EPA closed its National Radon Proficiency Program (RPP) in 1998.