The Kinesin-8 Member Kif19 Alters Microtubule Dynamics, Suppresses
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
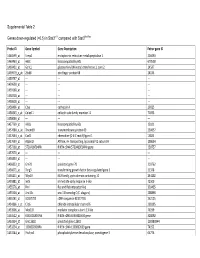
0.5) in Stat3∆/∆ Compared with Stat3flox/Flox
Supplemental Table 2 Genes down-regulated (<0.5) in Stat3∆/∆ compared with Stat3flox/flox Probe ID Gene Symbol Gene Description Entrez gene ID 1460599_at Ermp1 endoplasmic reticulum metallopeptidase 1 226090 1460463_at H60c histocompatibility 60c 670558 1460431_at Gcnt1 glucosaminyl (N-acetyl) transferase 1, core 2 14537 1459979_x_at Zfp68 zinc finger protein 68 24135 1459747_at --- --- --- 1459608_at --- --- --- 1459168_at --- --- --- 1458718_at --- --- --- 1458618_at --- --- --- 1458466_at Ctsa cathepsin A 19025 1458345_s_at Colec11 collectin sub-family member 11 71693 1458046_at --- --- --- 1457769_at H60a histocompatibility 60a 15101 1457680_a_at Tmem69 transmembrane protein 69 230657 1457644_s_at Cxcl1 chemokine (C-X-C motif) ligand 1 14825 1457639_at Atp6v1h ATPase, H+ transporting, lysosomal V1 subunit H 108664 1457260_at 5730409E04Rik RIKEN cDNA 5730409E04Rik gene 230757 1457070_at --- --- --- 1456893_at --- --- --- 1456823_at Gm70 predicted gene 70 210762 1456671_at Tbrg3 transforming growth factor beta regulated gene 3 21378 1456211_at Nlrp10 NLR family, pyrin domain containing 10 244202 1455881_at Ier5l immediate early response 5-like 72500 1455576_at Rinl Ras and Rab interactor-like 320435 1455304_at Unc13c unc-13 homolog C (C. elegans) 208898 1455241_at BC037703 cDNA sequence BC037703 242125 1454866_s_at Clic6 chloride intracellular channel 6 209195 1453906_at Med13l mediator complex subunit 13-like 76199 1453522_at 6530401N04Rik RIKEN cDNA 6530401N04 gene 328092 1453354_at Gm11602 predicted gene 11602 100380944 1453234_at -

Supplementary Table S1. Correlation Between the Mutant P53-Interacting Partners and PTTG3P, PTTG1 and PTTG2, Based on Data from Starbase V3.0 Database
Supplementary Table S1. Correlation between the mutant p53-interacting partners and PTTG3P, PTTG1 and PTTG2, based on data from StarBase v3.0 database. PTTG3P PTTG1 PTTG2 Gene ID Coefficient-R p-value Coefficient-R p-value Coefficient-R p-value NF-YA ENSG00000001167 −0.077 8.59e-2 −0.210 2.09e-6 −0.122 6.23e-3 NF-YB ENSG00000120837 0.176 7.12e-5 0.227 2.82e-7 0.094 3.59e-2 NF-YC ENSG00000066136 0.124 5.45e-3 0.124 5.40e-3 0.051 2.51e-1 Sp1 ENSG00000185591 −0.014 7.50e-1 −0.201 5.82e-6 −0.072 1.07e-1 Ets-1 ENSG00000134954 −0.096 3.14e-2 −0.257 4.83e-9 0.034 4.46e-1 VDR ENSG00000111424 −0.091 4.10e-2 −0.216 1.03e-6 0.014 7.48e-1 SREBP-2 ENSG00000198911 −0.064 1.53e-1 −0.147 9.27e-4 −0.073 1.01e-1 TopBP1 ENSG00000163781 0.067 1.36e-1 0.051 2.57e-1 −0.020 6.57e-1 Pin1 ENSG00000127445 0.250 1.40e-8 0.571 9.56e-45 0.187 2.52e-5 MRE11 ENSG00000020922 0.063 1.56e-1 −0.007 8.81e-1 −0.024 5.93e-1 PML ENSG00000140464 0.072 1.05e-1 0.217 9.36e-7 0.166 1.85e-4 p63 ENSG00000073282 −0.120 7.04e-3 −0.283 1.08e-10 −0.198 7.71e-6 p73 ENSG00000078900 0.104 2.03e-2 0.258 4.67e-9 0.097 3.02e-2 Supplementary Table S2. -

TITLE PAGE Oxidative Stress and Response to Thymidylate Synthase
Downloaded from molpharm.aspetjournals.org at ASPET Journals on October 2, 2021 -Targeted -Targeted 1 , University of of , University SC K.W.B., South Columbia, (U.O., Carolina, This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. The final version may differ from this version. This article has not been copyedited and formatted. -

Kif18a Regulates Sirt2-Mediated Tubulin Acetylation for Spindle
Tang et al. Cell Div (2018) 13:9 https://doi.org/10.1186/s13008-018-0042-4 Cell Division RESEARCH Open Access Kif18a regulates Sirt2‑mediated tubulin acetylation for spindle organization during mouse oocyte meiosis Feng Tang, Meng‑Hao Pan, Xiang Wan, Yujie Lu, Yu Zhang and Shao‑Chen Sun* Abstract Background: During oocyte meiosis, the cytoskeleton dynamics, especially spindle organization, are critical for chro‑ mosome congression and segregation. However, the roles of the kinesin superfamily in this process are still largely unknown. Results: In the present study, Kif18a, a member of the kinesin-8 family, regulated spindle organization through its efects on tubulin acetylation in mouse oocyte meiosis. Our results showed that Kif18a is expressed and mainly local‑ ized in the spindle region. Knock down of Kif18a caused the failure of frst polar body extrusion, dramatically afecting spindle organization and resulting in severe chromosome misalignment. Further analysis showed that the disrup‑ tion of Kif18a caused an increase in acetylated tubulin level, which might be the reason for the spindle organization defects after Kif18a knock down in oocyte meiosis, and the decreased expression of deacetylase Sirt2 was found after Kif18a knock down. Moreover, microinjections of tubulin K40R mRNA, which could induce tubulin deacetylation, pro‑ tected the oocytes from the efects of Kif18a downregulation, resulting in normal spindle morphology in Kif18a-knock down oocytes. Conclusions: Taken together, our results showed that Kif18a afected Sirt2-mediated tubulin acetylation level for spindle organization during mouse oocyte meiosis. Our results not only revealed the critical efect of Kif18a on micro‑ tubule stability, but also extended our understanding of kinesin activity in meiosis. -

Selective and ATP‐Competitive Kinesin KIF18A Inhibitor Suppresses
Received: 16 October 2019 | Revised: 13 February 2020 | Accepted: 6 March 2020 DOI: 10.1111/jcmm.15200 ORIGINAL ARTICLE Selective and ATP-competitive kinesin KIF18A inhibitor suppresses the replication of influenza A virus Yong-Bin Cho1 | Sungguan Hong2 | Kyung-Won Kang3 | Ji-Hun Kang1 | Sang- Myeong Lee3 | Young-Jin Seo1 1Department of Life Science, Chung-Ang University, Seoul, South Korea Abstract 2Department of Chemistry, Chung-Ang The influenza virus is one of the major public health threats. However, the devel- University, Seoul, South Korea opment of efficient vaccines and therapeutic drugs to combat this virus is greatly 3Division of Biotechnology, College of Environmental and Bioresources, Jeonbuk limited by its frequent genetic mutations. Because of this, targeting the host factors National University, Iksan, South Korea required for influenza virus replication may be a more effective strategy for inhibit- Correspondence ing a broader spectrum of variants. Here, we demonstrated that inhibition of a motor Sang-Myeong Lee, Division of protein kinesin family member 18A (KIF18A) suppresses the replication of the influ- Biotechnology, College of Environmental and Bioresources Sciences, Jeonbuk enza A virus (IAV). The expression of KIF18A in host cells was increased following National University, Gobong-ro 79, Iksan-si, IAV infection. Intriguingly, treatment with the selective and ATP-competitive mitotic Jeollabuk-do 570-752, Korea. Email: [email protected] kinesin KIF18A inhibitor BTB-1 substantially decreased the expression of viral RNAs and proteins, and the production of infectious viral particles, while overexpression Young-Jin Seo, Department of Life Science, College of Natural Sciences, Chung-Ang of KIF18A enhanced the replication of IAV. -

Post-Transcriptionally Impaired De Novo Mutations Contribute to The
bioRxiv preprint doi: https://doi.org/10.1101/175844; this version posted November 26, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. 1 Post-transcriptionally impaired de novo mutations 2 contribute to the genetic etiology of four neuropsychiatric 3 disorders 4 5 Fengbiao Mao1,2¶, Lu Wang3¶, Xiaolu Zhao2, Zhongshan Li4, Luoyuan Xiao5, 6 Rajesh C. Rao2, Jinchen Li4, Huajing Teng1*, Xin He6*, and Zhong Sheng Sun1,4* 7 8 1 Beijing Institutes of Life Science, Chinese Academy of Sciences, Beijing 100101, 9 China. 10 2 Department of Pathology, University of Michigan, Ann Arbor, MI 48109, USA. 11 3 Institute of Life Science, Southeast University, Nanjing 210096, China. 12 4 Institute of Genomic Medicine, Wenzhou Medical University, Wenzhou 325027, 13 China 14 5 Department of Computer Science and Technology, Tsinghua University, Beijing 15 100084, China. 16 6 Department of Human Genetics, University of Chicago, Chicago, IL, USA. 17 18 ¶These authors contributed equally to this work 19 * Corresponding authors 20 E-mail: 21 [email protected] (Z.S.S.) 22 [email protected] (X.H.) 23 [email protected] (H.T.) 24 25 1 bioRxiv preprint doi: https://doi.org/10.1101/175844; this version posted November 26, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. -

The Kinesin Spindle Protein Inhibitor Filanesib Enhances the Activity of Pomalidomide and Dexamethasone in Multiple Myeloma
Plasma Cell Disorders SUPPLEMENTARY APPENDIX The kinesin spindle protein inhibitor filanesib enhances the activity of pomalidomide and dexamethasone in multiple myeloma Susana Hernández-García, 1 Laura San-Segundo, 1 Lorena González-Méndez, 1 Luis A. Corchete, 1 Irena Misiewicz- Krzeminska, 1,2 Montserrat Martín-Sánchez, 1 Ana-Alicia López-Iglesias, 1 Esperanza Macarena Algarín, 1 Pedro Mogollón, 1 Andrea Díaz-Tejedor, 1 Teresa Paíno, 1 Brian Tunquist, 3 María-Victoria Mateos, 1 Norma C Gutiérrez, 1 Elena Díaz- Rodriguez, 1 Mercedes Garayoa 1* and Enrique M Ocio 1* 1Centro Investigación del Cáncer-IBMCC (CSIC-USAL) and Hospital Universitario-IBSAL, Salamanca, Spain; 2National Medicines Insti - tute, Warsaw, Poland and 3Array BioPharma, Boulder, Colorado, USA *MG and EMO contributed equally to this work ©2017 Ferrata Storti Foundation. This is an open-access paper. doi:10.3324/haematol. 2017.168666 Received: March 13, 2017. Accepted: August 29, 2017. Pre-published: August 31, 2017. Correspondence: [email protected] MATERIAL AND METHODS Reagents and drugs. Filanesib (F) was provided by Array BioPharma Inc. (Boulder, CO, USA). Thalidomide (T), lenalidomide (L) and pomalidomide (P) were purchased from Selleckchem (Houston, TX, USA), dexamethasone (D) from Sigma-Aldrich (St Louis, MO, USA) and bortezomib from LC Laboratories (Woburn, MA, USA). Generic chemicals were acquired from Sigma Chemical Co., Roche Biochemicals (Mannheim, Germany), Merck & Co., Inc. (Darmstadt, Germany). MM cell lines, patient samples and cultures. Origin, authentication and in vitro growth conditions of human MM cell lines have already been characterized (17, 18). The study of drug activity in the presence of IL-6, IGF-1 or in co-culture with primary bone marrow mesenchymal stromal cells (BMSCs) or the human mesenchymal stromal cell line (hMSC–TERT) was performed as described previously (19, 20). -

Decreased DNA Methylation at Promoters and Gene-Specific Neuronal
medRxiv preprint doi: https://doi.org/10.1101/2020.12.10.20246405; this version posted December 11, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. It is made available under a CC-BY-NC-ND 4.0 International license . Decreased DNA methylation at promoters and gene-specific neuronal hypermethylation in the prefrontal cortex of patients with bipolar disorder Miki Bundo1†, Junko Ueda1,2†, Yutaka Nakachi1, Kiyoto Kasai3,4, Tadafumi Kato2,5#, Kazuya Iwamoto1# 1Department of Molecular Brain Science, Graduate School of Medical Sciences, Kumamoto University, Japan 2Laboratory for Molecular Dynamics of Mental Disorders, RIKEN Center for Brain Science, Japan 3Department of Neuropsychiatry, Graduate School of Medicine, The University of Tokyo, Japan 4The International Research Center for Neurointelligence (WPI-IRCN) at The University of Tokyo Institutes for Advanced Study (UTIAS), 5Department of Psychiatry and Behavioral Science, Graduate School of Medicine, Juntendo University †contributed equally to this work #corresponding authors: Kazuya Iwamoto Department of Molecular Brain Science, Graduate School of Medical Sciences, Kumamoto University. 1-1-1 Honjo, Chuo-ku, Kumamoto, 860-8556, Japan [email protected] Tadadumi Kato Department of Psychiatry and Behavioral Science, Graduate School of Medicine, Juntendo University. 2-1-1 Hongo, Bunkyo, Tokyo 113-8421, Japan [email protected]. Running title: DNA methylation analysis in bipolar disorder 1 NOTE: This preprint reports new research that has not been certified by peer review and should not be used to guide clinical practice. medRxiv preprint doi: https://doi.org/10.1101/2020.12.10.20246405; this version posted December 11, 2020. -

Tubular P53 Regulates Multiple Genes to Mediate AKI
BASIC RESEARCH www.jasn.org Tubular p53 Regulates Multiple Genes to Mediate AKI † † † † † Dongshan Zhang,* Yu Liu,* Qingqing Wei, Yuqing Huo, Kebin Liu, Fuyou Liu,* and † Zheng Dong* *Departments of Emergency Medicine and Nephrology, Second Xiangya Hospital, Central South University, Changsha, Hunan, China; and †Department of Cellular Biology and Anatomy, Vascular Biology Center and Department of Biochemistry and Molecular Biology, Georgia Regents University and Charlie Norwood Veterans Affairs Medical Center, Augusta, Georgia ABSTRACT A pathogenic role of p53 in AKI was suggested a decade ago but remains controversial. Indeed, recent work indicates that inhibition of p53 protects against ischemic AKI in rats but exacerbates AKI in mice. One intriguing possibility is that p53 has cell type-specific roles in AKI. To determine the role of tubular p53, we generated two conditional gene knockout mouse models, in which p53 is specifically ablated from proximal tubules or other tubular segments, including distal tubules, loops of Henle, and medullary collecting ducts. Proximal tubule p53 knockout (PT-p53-KO) mice were resistant to ischemic and cisplatin nephrotoxic AKI, which was indicated by the analysis of renal function, histology, apoptosis, and inflammation. However, other tubular p53 knockout (OT-p53-KO) mice were sensitive to AKI. Mechanis- tically, AKI associated with the upregulation of several known p53 target genes, including Bax, p53- upregulated modulator of apoptosis-a, p21, and Siva, and this association was attenuated in PT-p53-KO mice. In global expression analysis, ischemic AKI induced 371 genes in wild-type kidney cortical tissues, but the induction of 31 of these genes was abrogated in PT-p53-KO tissues. -

Original Article Integrative Analysis of Somatic Mutations and Differential Expression Profiles in Glioblastoma Based on Aging Acceleration
Int J Clin Exp Pathol 2021;14(5):582-595 www.ijcep.com /ISSN:1936-2625/IJCEP0121428 Original Article Integrative analysis of somatic mutations and differential expression profiles in glioblastoma based on aging acceleration Huize Wang1,2, Shiyan Li3, Hongxin Liu3, Shiyu Bian4, Wanjiang Huang5, Chengzhong Xing2, Yin Wang2,3 1Department of Nursing, First Affiliated Hospital of China Medical University, 155# North Nanjing Street, Shenyang 110001, Liaoning, China; 2Tumor Etiology and Screening Department of Cancer Institute and General Surgery, The First Affiliated Hospital of China Medical University, 155# North Nanjing Street, Heping District, Shenyang 110001, Liaoning Province, China; 3Department of Biomedical Engineering, School of Fundamental Sciences, China Medical University, Shenyang 110122, Liaoning Province, China; 4China Medical University - The Queen’s University of Belfast Joint College, China Medical University, Shenyang 110122, Liaoning Province, China; 5No. 10 Middle School, Xiangyang 441021, Hubei Province, China Received August 31, 2020; Accepted February 7, 2021; Epub May 15, 2021; Published May 30, 2021 Abstract: Background: Glioblastoma (GBM) is an aggressive brain tumor and the mechanisms of progression are very complex. Accelerated aging is a driving factor of GBM. However, there has not been thorough research about the mechanisms of GBM progression based on aging acceleration. Methods: The aging predictor was modeled based on normal brain samples. Then an aging acceleration background network was constructed to explore GBM mechanisms. Results: The accelerated aging-related mechanisms provided an innovative way to study GBM, where- in integrative analysis of somatic mutations and differential expression revealed key pathologic characteristics. Moreover, the influence of the immune system, the nervous system and other critical factors on GBM were identi- fied. -

Cell Cycle Arrest Through Indirect Transcriptional Repression by P53: I Have a DREAM
Cell Death and Differentiation (2018) 25, 114–132 Official journal of the Cell Death Differentiation Association OPEN www.nature.com/cdd Review Cell cycle arrest through indirect transcriptional repression by p53: I have a DREAM Kurt Engeland1 Activation of the p53 tumor suppressor can lead to cell cycle arrest. The key mechanism of p53-mediated arrest is transcriptional downregulation of many cell cycle genes. In recent years it has become evident that p53-dependent repression is controlled by the p53–p21–DREAM–E2F/CHR pathway (p53–DREAM pathway). DREAM is a transcriptional repressor that binds to E2F or CHR promoter sites. Gene regulation and deregulation by DREAM shares many mechanistic characteristics with the retinoblastoma pRB tumor suppressor that acts through E2F elements. However, because of its binding to E2F and CHR elements, DREAM regulates a larger set of target genes leading to regulatory functions distinct from pRB/E2F. The p53–DREAM pathway controls more than 250 mostly cell cycle-associated genes. The functional spectrum of these pathway targets spans from the G1 phase to the end of mitosis. Consequently, through downregulating the expression of gene products which are essential for progression through the cell cycle, the p53–DREAM pathway participates in the control of all checkpoints from DNA synthesis to cytokinesis including G1/S, G2/M and spindle assembly checkpoints. Therefore, defects in the p53–DREAM pathway contribute to a general loss of checkpoint control. Furthermore, deregulation of DREAM target genes promotes chromosomal instability and aneuploidy of cancer cells. Also, DREAM regulation is abrogated by the human papilloma virus HPV E7 protein linking the p53–DREAM pathway to carcinogenesis by HPV.Another feature of the pathway is that it downregulates many genes involved in DNA repair and telomere maintenance as well as Fanconi anemia. -

SUPPORTING INFORMATION for Regulation of Gene Expression By
SUPPORTING INFORMATION for Regulation of gene expression by the BLM helicase correlates with the presence of G4 motifs Giang Huong Nguyen1,2, Weiliang Tang3, Ana I. Robles1, Richard P. Beyer4, Lucas T. Gray5, Judith A. Welsh1, Aaron J. Schetter1, Kensuke Kumamoto1,6, Xin Wei Wang1, Ian D. Hickson2,7, Nancy Maizels5, 3,8 1 Raymond J. Monnat, Jr. and Curtis C. Harris 1Laboratory of Human Carcinogenesis, National Cancer Institute, National Institutes of Health, Bethesda, Maryland, U.S.A; 2Department of Medical Oncology, Weatherall Institute of Molecular Medicine, John Radcliffe Hospital, University of Oxford, Oxford, U.K.; 3Department of Pathology, University of Washington, Seattle, WA U.S.A.; 4 Center for Ecogenetics and Environmental Health, University of Washington, Seattle, WA U.S.A.; 5Department of Immunology and Department of Biochemistry, University of Washington, Seattle, WA U.S.A.; 6Department of Organ Regulatory Surgery, Fukushima Medical University, Fukushima, Japan; 7Cellular and Molecular Medicine, Nordea Center for Healthy Aging, University of Copenhagen, Denmark; 8Department of Genome Sciences, University of WA, Seattle, WA U.S.A. SI Index: Supporting Information for this manuscript includes the following 19 items. A more detailed Materials and Methods section is followed by 18 Tables and Figures in order of their appearance in the manuscript text: 1) SI Materials and Methods 2) Figure S1. Study design and experimental workflow. 3) Figure S2. Immunoblot verification of BLM depletion from human fibroblasts. 4) Figure S3. PCA of mRNA and miRNA expression in BLM-depleted human fibroblasts. 5) Figure S4. qPCR confirmation of mRNA array data. 6) Table S1. BS patient and control detail.