Cefuroxime Axetil (Ceftin(R)) a Brief Review
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -

Antimicrobial Stewardship Guidance
Antimicrobial Stewardship Guidance Federal Bureau of Prisons Clinical Practice Guidelines March 2013 Clinical guidelines are made available to the public for informational purposes only. The Federal Bureau of Prisons (BOP) does not warrant these guidelines for any other purpose, and assumes no responsibility for any injury or damage resulting from the reliance thereof. Proper medical practice necessitates that all cases are evaluated on an individual basis and that treatment decisions are patient-specific. Consult the BOP Clinical Practice Guidelines Web page to determine the date of the most recent update to this document: http://www.bop.gov/news/medresources.jsp Federal Bureau of Prisons Antimicrobial Stewardship Guidance Clinical Practice Guidelines March 2013 Table of Contents 1. Purpose ............................................................................................................................................. 3 2. Introduction ...................................................................................................................................... 3 3. Antimicrobial Stewardship in the BOP............................................................................................ 4 4. General Guidance for Diagnosis and Identifying Infection ............................................................. 5 Diagnosis of Specific Infections ........................................................................................................ 6 Upper Respiratory Infections (not otherwise specified) .............................................................................. -
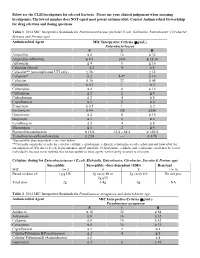
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp. -

Penicillin Allergy Guidance Document
Penicillin Allergy Guidance Document Key Points Background Careful evaluation of antibiotic allergy and prior tolerance history is essential to providing optimal treatment The true incidence of penicillin hypersensitivity amongst patients in the United States is less than 1% Alterations in antibiotic prescribing due to reported penicillin allergy has been shown to result in higher costs, increased risk of antibiotic resistance, and worse patient outcomes Cross-reactivity between truly penicillin allergic patients and later generation cephalosporins and/or carbapenems is rare Evaluation of Penicillin Allergy Obtain a detailed history of allergic reaction Classify the type and severity of the reaction paying particular attention to any IgE-mediated reactions (e.g., anaphylaxis, hives, angioedema, etc.) (Table 1) Evaluate prior tolerance of beta-lactam antibiotics utilizing patient interview or the electronic medical record Recommendations for Challenging Penicillin Allergic Patients See Figure 1 Follow-Up Document tolerance or intolerance in the patient’s allergy history Consider referring to allergy clinic for skin testing Created July 2017 by Macey Wolfe, PharmD; John Schoen, PharmD, BCPS; Scott Bergman, PharmD, BCPS; Sara May, MD; and Trevor Van Schooneveld, MD, FACP Disclaimer: This resource is intended for non-commercial educational and quality improvement purposes. Outside entities may utilize for these purposes, but must acknowledge the source. The guidance is intended to assist practitioners in managing a clinical situation but is not mandatory. The interprofessional group of authors have made considerable efforts to ensure the information upon which they are based is accurate and up to date. Any treatments have some inherent risk. Recommendations are meant to improve quality of patient care yet should not replace clinical judgment. -

CEFTIN® Tablets CEFTIN® for Oral Suspension
PRODUCT INFORMATION CEFTIN® Tablets (cefuroxime axetil tablets) CEFTIN® for Oral Suspension (cefuroxime axetil powder for oral suspension) DESCRIPTION: CEFTIN Tablets and CEFTIN for Oral Suspension contain cefuroxime as cefuroxime axetil. CEFTIN is a semisynthetic, broad-spectrum cephalosporin antibiotic for oral administration. Chemically, cefuroxime axetil, the 1-(acetyloxy) ethyl ester of cefuroxime, is (RS)-1-hydroxyethyl (6R,7R)-7-[2-(2-furyl)glyoxylamido]-3-(hydroxymethyl)-8-oxo-5- thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate, 72-(Z)-(O-methyl-oxime), 1-acetate 3-carbamate. Its molecular formula is C20H22N4O10S, and it has a molecular weight of 510.48. Cefuroxime axetil is in the amorphous form and has the following structural formula: CEFTIN Tablets are film-coated and contain the equivalent of 125, 250, or 500 mg of cefuroxime as cefuroxime axetil. CEFTIN Tablets contain the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, FD&C Blue No. 1 (250- and 500-mg tablets only), hydrogenated vegetable oil, hydroxypropyl methylcellulose, methylparaben, microcrystalline cellulose, propylene glycol, propylparaben, sodium benzoate (125-mg tablets only), sodium lauryl sulfate, and CEFTIN® Tablets (cefuroxime axetil tablets) CEFTIN® for Oral Suspension (cefuroxime axetil powder for oral suspension) titanium dioxide. CEFTIN for Oral Suspension, when reconstituted with water, provides the equivalent of 125 mg or 250 mg of cefuroxime (as cefuroxime axetil) per 5 mL of suspension. CEFTIN for Oral Suspension contains the inactive ingredients povidone K30, stearic acid, sucrose, and tutti-frutti flavoring. CLINICAL PHARMACOLOGY: Absorption and Metabolism: After oral administration, cefuroxime axetil is absorbed from the gastrointestinal tract and rapidly hydrolyzed by nonspecific esterases in the intestinal mucosa and blood to cefuroxime. -

Safety and Efficacy of Ceftaroline Fosamil in the Management of Community-Acquired Bacterial Pneumonia Heather F
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Scholarly Papers 2014 Safety and Efficacy of Ceftaroline Fosamil in the Management of Community-Acquired Bacterial Pneumonia Heather F. DeBellis Kimberly L. Barefield Philadelphia College of Osteopathic Medicine, [email protected] Follow this and additional works at: https://digitalcommons.pcom.edu/scholarly_papers Part of the Medicine and Health Sciences Commons Recommended Citation DeBellis, Heather F. and Barefield, Kimberly L., "Safety and Efficacy of Ceftaroline Fosamil in the Management of Community- Acquired Bacterial Pneumonia" (2014). PCOM Scholarly Papers. 1913. https://digitalcommons.pcom.edu/scholarly_papers/1913 This Article is brought to you for free and open access by DigitalCommons@PCOM. It has been accepted for inclusion in PCOM Scholarly Papers by an authorized administrator of DigitalCommons@PCOM. For more information, please contact [email protected]. Open Access: Full open access to Clinical Medicine Reviews this and thousands of other papers at http://www.la-press.com. in Therapeutics Safety and Efficacy of Ceftaroline Fosamil in the Management of Community- Acquired Bacterial Pneumonia Heather F. DeBellis and Kimberly L. Tackett South University School of Pharmacy, Savannah, GA, USA. ABSTR ACT: Ceftaroline fosamil is a new fifth-generation cephalosporin indicated for the treatment of community-acquired bacterial pneumonia (CABP). It possesses antimicrobial effects against both Gram-positive and Gram-negative bacteria, including methicillin-resistant Staphylococcus aureus (MRSA), but not against anaerobes. Organisms covered by this novel agent that are commonly associated with CABP are Streptococcus pneumoniae, Staphylococcus aureus, Haemophilus influenzae, Moraxella catarrhalis, and Klebsiella pneumoniae; however, ceftaroline fosamil lacks antimicrobial activity against Pseudomonas and Acinetobacter species. -

Cephalosporins Can Be Prescribed Safely for Penicillin-Allergic Patients ▲
JFP_0206_AE_Pichichero.Final 1/23/06 1:26 PM Page 106 APPLIED EVIDENCE New research findings that are changing clinical practice Michael E. Pichichero, MD University of Rochester Cephalosporins can be Medical Center, Rochester, NY prescribed safely for penicillin-allergic patients Practice recommendations an allergic reaction to cephalosporins, ■ The widely quoted cross-allergy risk compared with the incidence of a primary of 10% between penicillin and (and unrelated) cephalosporin allergy. cephalosporins is a myth (A). Most people produce IgG and IgM antibodies in response to exposure to ■ Cephalothin, cephalexin, cefadroxil, penicillin1 that may cross-react with and cefazolin confer an increased risk cephalosporin antigens.2 The presence of of allergic reaction among patients these antibodies does not predict allergic, with penicillin allergy (B). IgE cross-sensitivity to a cephalosporin. ■ Cefprozil, cefuroxime, cefpodoxime, Even penicillin skin testing is generally not ceftazidime, and ceftriaxone do not predictive of cephalosporin allergy.3 increase risk of an allergic reaction (B). Reliably predicting cross-reactivity ndoubtedly you have patients who A comprehensive review of the evidence say they are allergic to penicillin shows that the attributable risk of a cross- U but have difficulty recalling details reactive allergic reaction varies and is of the reactions they experienced. To be strongest when the chemical side chain of safe, we often label these patients as peni- the specific cephalosporin is similar to that cillin-allergic without further questioning of penicillin or amoxicillin. and withhold not only penicillins but Administration of cephalothin, cepha- cephalosporins due to concerns about lexin, cefadroxil, and cefazolin in penicillin- potential cross-reactivity and resultant IgE- allergic patients is associated with a mediated, type I reactions. -

A Thesis Entitled an Oral Dosage Form of Ceftriaxone Sodium Using Enteric
A Thesis entitled An oral dosage form of ceftriaxone sodium using enteric coated sustained release calcium alginate beads by Darshan Lalwani Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Master of Science Degree in Pharmaceutical Sciences with Industrial Pharmacy Option _________________________________________ Jerry Nesamony, Ph.D., Committee Chair _________________________________________ Sai Hanuman Sagar Boddu, Ph.D, Committee Member _________________________________________ Youssef Sari, Ph.D., Committee Member _________________________________________ Patricia R. Komuniecki, PhD, Dean College of Graduate Studies The University of Toledo May 2015 Copyright 2015, Darshan Narendra Lalwani This document is copyrighted material. Under copyright law, no parts of this document may be reproduced without the expressed permission of the author. An Abstract of An oral dosage form of ceftriaxone sodium using enteric coated sustained release calcium alginate beads by Darshan Lalwani Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Master of Science Degree in Pharmaceutical Sciences with Industrial Pharmacy option The University of Toledo May 2015 Purpose: Ceftriaxone (CTZ) is a broad spectrum semisynthetic, third generation cephalosporin antibiotic. It is an acid labile drug belonging to class III of biopharmaceutical classification system (BCS). It can be solvated quickly but suffers from the drawback of poor oral bioavailability owing to its limited permeability through -

Cefuroxime Axetil
Cefuroxime axetil Zinnat® Tablets PRODUCT DESCRIPTION Cefuroxime (as axetil) (Zinnat®) 250mg tablet:. Each white, film-coated, capsule-shaped tablet engraved with ‘GXES7' on one side tablet contains 250mg of Cefuroxime (as axetil). Cefuroxime (as axetil) (Zinnat®) 500mg tablet: Each white, film-coated, capsule-shaped tablet engraved with 'GXEG2' on one side tablet contains 500mg of Cefuroxime (as axetil). PHARMACOLOGIC PROPERTIES Pharmacodynamics The prevalence of acquired resistance is geographically and time dependent and for select species may be very high. Local information on resistance is desirable, particularly when treating severe infections. In vitro susceptibility of micro-organisms to Cefuroxime Where clinical efficacy of cefuroxime axetil has been demonstrated in clinical trials this is indicated with an asterisk (*). Commonly Susceptible Species Gram-Positive Aerobes: Staphylococcus aureus (methicillin susceptible)* Coagulase negative staphylococcus (methicillin susceptible) Streptococcus pyogenes* Beta-hemolytic streptococci Gram-Negative Aerobes: Haemophilus influenzae* including ampicillin resistant strains Haemophilus parainfluenzae* Moraxella catarrhalis* Neisseria gonorrhoea* including penicillinase and non-penicillinase producing strains Gram-Positive Anaerobes: Peptostreptococcus spp. Propionibacterium spp. Spirochetes: Borrelia burgdorferi* Organisms for which acquired resistance may be a problem Gram-Positive Aerobes: Streptococcus pneumoniae* Gram-Negative Aerobes: Citrobacter spp. not including C. freundii Enterobacter -

List Item Assessment Report for Zinacef
10 September 2012 EMA/CHMP/633654/2012 Assessment report for Zinacef and associated names Pursuant to Article 30 of Directive 2001/83/EC INN : cefuroxime (cefuroxime sodium) Procedure no: EMEA/H/A-30/1158 Assessment Report as adopted by the CHMP with all information of a commercially confidential nature deleted. 7 Westferry Circus ● Canary Wharf ● London E14 4HB ● United Kingdom Telephone +44 (0)20 7418 8400 Facsimile +44 (0)20 7523 7051 E -mail [email protected] Website www.ema.europa.eu An agency of the European Union © European Medicines Agency, 2013. Reproduction is authorised provided the source is acknowledged. Table of contents Table of contents ......................................................................................... 2 1. Background information on the procedure .............................................. 3 1.1. Background information on the basis of the grounds for referral ................................. 3 2.1. Introduction......................................................................................................... 3 2.2. Critical Evaluation ................................................................................................ 4 Section 4.1 - Therapeutic indications ............................................................................. 4 Section 4.2 - Posology and method of administration .................................................... 30 Section 4.3 - Contraindications ................................................................................... 33 Section 4.4 - Special -

Comparison Between Cefuroxime and Amoxicillin+Clavulanate in Patients
International Journal of Surgery Science 2019; 3(1): 154-155 E-ISSN: 2616-3470 P-ISSN: 2616-3462 © Surgery Science Comparison between cefuroxime and www.surgeryscience.com 2019; 3(1): 154-155 amoxicillin+clavulanate in patients of inguinal hernia Received: 25-11-2018 Accepted: 27-12-2018 undergoing mesh hernioplasty, a randomised control Dr. Girish Pandey study Assistant Professor, Department of General Surgery, Heritage Institute of Medical Sciences, Dr. Girish Pandey Varanasi, Uttar Pradesh, India DOI: https://doi.org/10.33545/surgery.2019.v3.i1c.29 Abstract A randomized control study was conducted at Heritage Institute of Medical Sciences (HIMS) Varanasi to evaluate clinical efficacy of cefuroxime compared with amoxicillin+clavulanate for prevention of wound infection in patients undergoing open prolene - mesh hernioplasty in cases of inguinal hernia. There was minimal difference in overall wound infection rates between cefuroxime and amoxicillin+clavulanate group and side–effects in cefuroxime group were slightly less. Therefore, cefuroxime appears to be slightly more effective antibiotic for use as surgical prophylaxis. Keywords: Hernioplasty. Wound infection. Antibiotic prophylaxis. Surgical site infection Introduction Wound infection is very common problem worldwide. Surgical site Infections (SSI) are potential complications associated with any type of procedure and are among the most preventable hospital acquired infection. SSI effect on average 11% of patients undergoing a surgical procedure and second to third most frequent type of hospital acquired infection in US and Europe. At present, no country is free from the burden of diseases caused by HAI. Commonest causative organisms are staphylococci, streptococci etc. Antibiotics are used to prevent infection at surgical site. -

Comparison of Ceftriaxone and Cefazolin Sodium Antibiotic
ORIGINAL RESEARCH General Urology Doi: 10.4274/jus.galenos.2018.2367 Journal of Urological Surgery, 2019;6(2):111-117 Comparison of Ceftriaxone and Cefazolin Sodium Antibiotic Prophylaxis in Terms of SIRS/Urosepsis Rates in Patients Undergoing Percutaneous Nephrolithotomy Perkütan Nefrolitotomi Ameliyatı Olan Hastalara Verilen Seftriakson ve Sefazolin Sodyum Antibiyotik Profilaksilerinin SIRS/Ürosepsis Oranları Açısından Karşılaştırılması Kerem Taken1, Alper Aşık1, Recep Eryılmaz1, Rahmi Aslan1, Muhammet İrfan Dönmez2, Mustafa Güneş1 1Yüzüncü Yıl University Faculty of Medicine, Department of Urology, Van, Turkiye 2Konya Training and Research Hospital, Clinic of Urology, Konya, Turkiye What’s known on the subject? and What does the study add? Percutaneous nephrolithotomy have infectious complications in the form of systemic inflammatory response (SIRS) and urosepsis. Antibiotic prophylaxis in order to prevent this complication has been widely investigated. Our prospective randomized study highlights two commonly used antibiotics in Turkiye and their effects on infectious complications. Our results indicate similar SIRS/urosepsis rates between groups and hence we advise to use cefazolin that has a narrower anti-bacterial spectrum. Abstract Objective: The aim of this study is to compare ceftriaxone and cefazoline sodium antibiotic prophylaxis in terms of development of Systemic Inflammatory Response syndrome (SIRS)/urosepsis in patients undergoing percutaneous nephrolithotomy (PCNL). Materials and Methods: Patients who underwent PCNL between June 2015 and October 2015 in our hospital were prospectively randomized to ceftriaxone (n=30) and cefazoline sodium (n=32) antibiotic prophylaxis groups. Patients with predisposing conditions to SIRS were excluded. Intraoperative urine cultures from renal pelvis and stone cultures were obtained from all patients. Clinical and laboratory findings of the patients who developed postoperative fever were evaluated.