List Item Assessment Report for Zinacef
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
28912 Oxoid FDA Cartridge Tables:1
* Adapted in part from CLSI document M100-S23 (M02-A11) : “Disc diffusion supplemental tablesʼʼ Performance standards for antimicrobial susceptibility testing. The complete standard may be obtained from the Clinical and Laboratory Standards Institute, 940 West Valley Road, Suite 1400, Wayne, PA 19807. Test Cultures (zone diameters in mm) Antimicrobial Agent Disc Code Potency Resistant Intermediate Susceptible Amikacin AK 30 μg EnterobacteriaceaeK, P. aeruginosa, Acinetobacter spp., and Staphylococcus spp. ≤14 15-16 ≥17 Amoxycillin - Clavulanic Acid AMC 20/10 μg EnterobacteriaceaeE ≤13 14-17 ≥18 Staphylococcus spp.A,Q ≤19 — ≥20 Haemophilus spp.A,Y ≤19 — ≥20 AmpicillinC,n AMP 10 μg EnterobacteriaceaeE and Vibrio choleraef ≤13 14-16 ≥17 Staphylococcus spp.A,Q ≤28 — ≥29 Enterococcus spp.A,V,W,k,m ≤16 — ≥17 Haemophilus spp.Y ≤18 19-21 ≥22 Streptococcus spp. ß-Hemolytic GroupA,d — — ≥24 Ampicillin – Sulbactam SAM 10/10 μg EnterobacteriaceaeE, Acinetobacter spp., and Staphylococcus spp. ≤11 12-14 ≥15 Haemophilus spp.A,Y ≤19 — ≥20 Azithromycin AZM 15 μg Staphylococcus spp., Streptococcus spp Viridans Groupn, ß-Hemolytic Group, and S. pneumoniae) ≤13 14-17 ≥18 Neisseria meningitidisA,i — — ≥20 Haemophilus spp.A — — ≥12 Aztreonam ATM 30 μg EnterobacteriaceaeE ≤17 18-20 ≤21 P. aeruginosa ≤15 16-21 ≥22 Haemophilus spp.A — — ≥26 CAR 100 μg Carbenicillin Enterobacteriaceae ≤19 20-22 ≥23 Pseudomonas aeruginosaP ≤13 14-16 ≥17 CEC 30 μg Cefaclor Enterobacteriaceae and Staphylococcus spp. ≤14 15-17 ≥18 Haemophilus spp.Y ≤16 17-19 ≥20 MA 30 μg Cefamandole EnterobacteriaceaeD,E and Staphylococcus spp. ≤14 15-17 ≥18 CefazolinG KZ 30 μg Staphylococcus spp. -

Prevalence of Urinary Tract Infection and Antibiotic Resistance Pattern in Pregnant Women, Najran Region, Saudi Arabia
Vol. 13(26), pp. 407-413, August, 2019 DOI: 10.5897/AJMR2019.9084 Article Number: E3F64FA61643 ISSN: 1996-0808 Copyright ©2019 Author(s) retain the copyright of this article African Journal of Microbiology Research http://www.academicjournals.org/AJMR Full Length Research Paper Prevalence of urinary tract infection and antibiotic resistance pattern in pregnant women, Najran region, Saudi Arabia Ali Mohamed Alshabi1*, Majed Saeed Alshahrani2, Saad Ahmed Alkahtani1 and Mohammad Shabib Akhtar1 1Department of Clinical Pharmacy, College of Pharmacy, Najran University, Najran, Saudi Arabia. 2Department of Obstetics and Gyneocology, Faculty of Medicine, Najran University, Najran, Saudi Arabia. Received 25 February, 2019; Accepted August 5, 2019 Urinary Tract Infection (UTI) is one of the commonest infectious disease in pregnancy, and in pregnancy we have very limited number of antibiotics to treat the UTI. This study was conducted on 151 patients who attended the gynecology clinic during the study period. Nineteen UTI proven cases of UTI were studied for prevalence of microorganism and sensitivity pattern against different antibiotics. Among the bacteria isolated, Escherichia coli (73.68%) and Staphylococcus aureus (10.52%) were the most prevalent Gram negative and Gram positive bacteria respectively. To know the resistance pattern of microorganism we used commercially available discs of different antibiotics. Gram negative bacteria showed more resistance as compared to Gram positive one. It is observed that the most effective antibiotic for Gram negative isolates is Ceftriaxone (87.5%), followed by Amoxicillin + Clavulanic acid (81.25%), Amikacin (75%), Cefuroxime (75%), Cefixime (68.75%) and Mezlocillin (62.5%). For the Gram positive bacteria, Ceftriaxone, Amikacin and Amoxicillin + Clavulanic acid were the most effective antimicrobials (100%). -

Antimicrobial Stewardship Guidance
Antimicrobial Stewardship Guidance Federal Bureau of Prisons Clinical Practice Guidelines March 2013 Clinical guidelines are made available to the public for informational purposes only. The Federal Bureau of Prisons (BOP) does not warrant these guidelines for any other purpose, and assumes no responsibility for any injury or damage resulting from the reliance thereof. Proper medical practice necessitates that all cases are evaluated on an individual basis and that treatment decisions are patient-specific. Consult the BOP Clinical Practice Guidelines Web page to determine the date of the most recent update to this document: http://www.bop.gov/news/medresources.jsp Federal Bureau of Prisons Antimicrobial Stewardship Guidance Clinical Practice Guidelines March 2013 Table of Contents 1. Purpose ............................................................................................................................................. 3 2. Introduction ...................................................................................................................................... 3 3. Antimicrobial Stewardship in the BOP............................................................................................ 4 4. General Guidance for Diagnosis and Identifying Infection ............................................................. 5 Diagnosis of Specific Infections ........................................................................................................ 6 Upper Respiratory Infections (not otherwise specified) .............................................................................. -
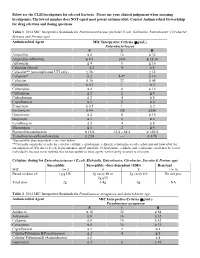
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp. -

Antibacterial Drug Usage Analysis
Department of Health and Human Services Public Health Service Food and Drug Administration Center for Drug Evaluation and Research Office of Surveillance and Epidemiology Drug Use Review Date: April 5, 2012 To: Edward Cox, M.D. Director Office of Antimicrobial Products Through: Gerald Dal Pan, M.D., MHS Director Office of Surveillance and Epidemiology Laura Governale, Pharm.D., MBA Deputy Director for Drug Use Division of Epidemiology II Office of Surveillance and Epidemiology Hina Mehta, Pharm.D. Drug Use Data Analysis Team Leader Division of Epidemiology II Office of Surveillance and Epidemiology From: Tracy Pham, Pharm.D. Drug Use Data Analyst Division of Epidemiology II Office of Surveillance and Epidemiology Drug Name(s): Systemic Antibacterial Drug Products Application Type/Number: Multiple Applicant/sponsor: Multiple OSE RCM #: 2012-544 **This document contains proprietary drug use data obtained by FDA under contract. The drug use data/information in this document has been cleared for public release.** 1 EXECUTIVE SUMMARY The Division of Epidemiology II is providing an update of the drug utilization data in terms of number of kilograms or international units of selected systemic antibacterial drug products sold from manufacturers to various retail and non-retail channels of distribution for years 2010-2011 as a surrogate for nationwide antibacterial drug use in humans. Propriety drug use databases licensed by the FDA were used to conduct this analysis. Data findings are as follows: During years 2010 and 2011, the majority of kilograms of selected systemic antibacterial drug products sold were to outpatient retail pharmacy settings. Approximately 3.28 million kilograms of selected systemic antibacterial drug products were sold during year 2010, and around 3.29 million kilograms were sold during year 2011. -

CEFTIN® Tablets CEFTIN® for Oral Suspension
PRODUCT INFORMATION CEFTIN® Tablets (cefuroxime axetil tablets) CEFTIN® for Oral Suspension (cefuroxime axetil powder for oral suspension) DESCRIPTION: CEFTIN Tablets and CEFTIN for Oral Suspension contain cefuroxime as cefuroxime axetil. CEFTIN is a semisynthetic, broad-spectrum cephalosporin antibiotic for oral administration. Chemically, cefuroxime axetil, the 1-(acetyloxy) ethyl ester of cefuroxime, is (RS)-1-hydroxyethyl (6R,7R)-7-[2-(2-furyl)glyoxylamido]-3-(hydroxymethyl)-8-oxo-5- thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate, 72-(Z)-(O-methyl-oxime), 1-acetate 3-carbamate. Its molecular formula is C20H22N4O10S, and it has a molecular weight of 510.48. Cefuroxime axetil is in the amorphous form and has the following structural formula: CEFTIN Tablets are film-coated and contain the equivalent of 125, 250, or 500 mg of cefuroxime as cefuroxime axetil. CEFTIN Tablets contain the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, FD&C Blue No. 1 (250- and 500-mg tablets only), hydrogenated vegetable oil, hydroxypropyl methylcellulose, methylparaben, microcrystalline cellulose, propylene glycol, propylparaben, sodium benzoate (125-mg tablets only), sodium lauryl sulfate, and CEFTIN® Tablets (cefuroxime axetil tablets) CEFTIN® for Oral Suspension (cefuroxime axetil powder for oral suspension) titanium dioxide. CEFTIN for Oral Suspension, when reconstituted with water, provides the equivalent of 125 mg or 250 mg of cefuroxime (as cefuroxime axetil) per 5 mL of suspension. CEFTIN for Oral Suspension contains the inactive ingredients povidone K30, stearic acid, sucrose, and tutti-frutti flavoring. CLINICAL PHARMACOLOGY: Absorption and Metabolism: After oral administration, cefuroxime axetil is absorbed from the gastrointestinal tract and rapidly hydrolyzed by nonspecific esterases in the intestinal mucosa and blood to cefuroxime. -

Consideration of Antibacterial Medicines As Part Of
Consideration of antibacterial medicines as part of the revisions to 2019 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) Section 6.2 Antibacterials including Access, Watch and Reserve Lists of antibiotics This summary has been prepared by the Health Technologies and Pharmaceuticals (HTP) programme at the WHO Regional Office for Europe. It is intended to communicate changes to the 2019 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) to national counterparts involved in the evidence-based selection of medicines for inclusion in national essential medicines lists (NEMLs), lists of medicines for inclusion in reimbursement programs, and medicine formularies for use in primary, secondary and tertiary care. This document does not replace the full report of the WHO Expert Committee on Selection and Use of Essential Medicines (see The selection and use of essential medicines: report of the WHO Expert Committee on Selection and Use of Essential Medicines, 2019 (including the 21st WHO Model List of Essential Medicines and the 7th WHO Model List of Essential Medicines for Children). Geneva: World Health Organization; 2019 (WHO Technical Report Series, No. 1021). Licence: CC BY-NC-SA 3.0 IGO: https://apps.who.int/iris/bitstream/handle/10665/330668/9789241210300-eng.pdf?ua=1) and Corrigenda (March 2020) – TRS1021 (https://www.who.int/medicines/publications/essentialmedicines/TRS1021_corrigenda_March2020. pdf?ua=1). Executive summary of the report: https://apps.who.int/iris/bitstream/handle/10665/325773/WHO- MVP-EMP-IAU-2019.05-eng.pdf?ua=1. -

WO 2010/025328 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 4 March 2010 (04.03.2010) WO 2010/025328 Al (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every A61K 31/00 (2006.01) kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, (21) International Application Number: CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, PCT/US2009/055306 DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (22) International Filing Date: HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, 28 August 2009 (28.08.2009) KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, (25) Filing Language: English NO, NZ, OM, PE, PG, PH, PL, PT, RO, RS, RU, SC, SD, (26) Publication Language: English SE, SG, SK, SL, SM, ST, SV, SY, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: 61/092,497 28 August 2008 (28.08.2008) US (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (71) Applicant (for all designated States except US): FOR¬ GM, KE, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, ZM, EST LABORATORIES HOLDINGS LIMITED [IE/ ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, —]; 18 Parliament Street, Milner House, Hamilton, TM), European (AT, BE, BG, CH, CY, CZ, DE, DK, EE, Bermuda HM12 (BM). -

Computational Antibiotics Book
Andrew V DeLong, Jared C Harris, Brittany S Larcart, Chandler B Massey, Chelsie D Northcutt, Somuayiro N Nwokike, Oscar A Otieno, Harsh M Patel, Mehulkumar P Patel, Pratik Pravin Patel, Eugene I Rowell, Brandon M Rush, Marc-Edwin G Saint-Louis, Amy M Vardeman, Felicia N Woods, Giso Abadi, Thomas J. Manning Computational Antibiotics Valdosta State University is located in South Georgia. Computational Antibiotics Index • Computational Details and Website Access (p. 8) • Acknowledgements (p. 9) • Dedications (p. 11) • Antibiotic Historical Introduction (p. 13) Introduction to Antibiotic groups • Penicillin’s (p. 21) • Carbapenems (p. 22) • Oxazolidines (p. 23) • Rifamycin (p. 24) • Lincosamides (p. 25) • Quinolones (p. 26) • Polypeptides antibiotics (p. 27) • Glycopeptide Antibiotics (p. 28) • Sulfonamides (p. 29) • Lipoglycopeptides (p. 30) • First Generation Cephalosporins (p. 31) • Cephalosporin Third Generation (p. 32) • Fourth-Generation Cephalosporins (p. 33) • Fifth Generation Cephalosporin’s (p. 34) • Tetracycline antibiotics (p. 35) Computational Antibiotics Antibiotics Covered (in alphabetical order) Amikacin (p. 36) Cefempidone (p. 98) Ceftizoxime (p. 159) Amoxicillin (p. 38) Cefepime (p. 100) Ceftobiprole (p. 161) Ampicillin (p. 40) Cefetamet (p. 102) Ceftoxide (p. 163) Arsphenamine (p. 42) Cefetrizole (p. 104) Ceftriaxone (p. 165) Azithromycin (p.44) Cefivitril (p. 106) Cefuracetime (p. 167) Aziocillin (p. 46) Cefixime (p. 108) Cefuroxime (p. 169) Aztreonam (p.48) Cefmatilen ( p. 110) Cefuzonam (p. 171) Bacampicillin (p. 50) Cefmetazole (p. 112) Cefalexin (p. 173) Bacitracin (p. 52) Cefodizime (p. 114) Chloramphenicol (p.175) Balofloxacin (p. 54) Cefonicid (p. 116) Cilastatin (p. 177) Carbenicillin (p. 56) Cefoperazone (p. 118) Ciprofloxacin (p. 179) Cefacetrile (p. 58) Cefoselis (p. 120) Clarithromycin (p. 181) Cefaclor (p. -

Comparative Activity of Newer Antibiotics Against Gram-Negative Bacilli
CONTRIBUTION • Comparative activity of newer antibiotics against gram-negative bacilli CYNTHIA C. KNAPP, MS AND JOHN A. WASHINGTON, MD • The in vitro activities of cefoperazone, cefotaxime, ceftriaxone, ceftazidime, azlocillin, mezlocillin, piperacillin, ticarcillin/clavulanate, aztreonam, imipenem, and ciprofloxacin were concurrently deter- mined against over 1,000 isolates of gram-negative bacilli from clinical specimens of patients at the Cleve- land Clinic. Cephalosporins, penicillins, and aztreonam were active against species of Enterobacteriaceae other than Citrobacter freundii, Enterobacter aerogenes, and Enterobacter cloacae. Ceftazidime was the most active cephalosporin against Pseudomonas aeruginosa. Against the Enterobacteriaceae, the rank order of activity of penicillins was ticarcillin/clavulanate > piperacillin > mezlocillin > azlocillin. Against P. aer- uginosa, the rank order of activity of penicillins was piperacillin > ticarcillin/clavulanate > azlocillin > mezlocillin. Aztreonam was less active v P. aeruginosa than ceftazidime, cefoperazone, or piperacillin. The most active antimicrobials against all isolates tested were imipenem and ciprofloxacin. • INDEX TERMS: ANTIBIOTICS, LACTAM; GRAM-NEGATIVE BACTERIA • CLEVE CLIN J MED 1989; 56:161-166 HE RECENT introduction of ciprofloxacin for and penicillins, as well as the monobactam, aztreonam, clinical use follows closely a period of active re- and the carbapenem, imipenem.1-3 search in and development of expanded spec- We compared the susceptibility of more than 1,000 trum p-lactam antibiotics, including cephalo- clinical bacterial isolates to four expanded spectrum Tsporins, penicillins, monobactams, and carbapenems. cephalosporins (cefoperazone, cefotaxime, ceftriaxone, Although numerous published studies compare the ac- and ceftazidime), four expanded spectrum penicillins tivity of ciprofloxacin with other quinolones, only a (azlocillin, mezlocillin, piperacillin, and ticarcil- limited number of studies compare the activity of ci- lin/clavulanate), aztreonam, imipenem, and ciproflox- acin. -

Antibiotic Resistance Pattern to Different Isolates in Al-Hillah City, Iraq
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by International Institute for Science, Technology and Education (IISTE): E-Journals Journal of Natural Sciences Research www.iiste.org ISSN 2224-3186 (Paper) ISSN 2225-0921 (Online) Vo l.3, No.3, 2013 Pseudomonas aeruginosa: Antibiotic resistance pattern to different isolates in Al-Hillah city, Iraq Ali Hussein Al-Marzoqi1* (Corresponding author), and Zahraa Mohammad Al Taee2 1. College of Science for women, Babylon University, PO box 435, Al-Hillah city, Babylon, Iraq. Tel: 009647710336121 E-mail: [email protected] 2. College of Science, Babylon University * E-mail of the corresponding author: [email protected] Abstract Aim: Pseudomonas aeruginosa (Ps. aeruginosa) considered as most important bacteria which can isolated from various kinds of infection. This study tries to survey the infections caused by Ps. aeruginosa especially medical and surgical care units and try to reveal the antimicrobial agents susceptibility against Ps. aeruginosa. Material and Method: This study was conducted during September 2012 to February 2013. During this period total of 285 samples were tested and showed growth of bacteria. The isolates of Pseudomonas aeruginosa were selected on the basis of their growth on Nutrient agar pigmented and non-pigmented colonies with oxidase positive and on routine MacConkey medium which showed lactose Non-fermenting pale colonies. Antimicrobial susceptibility of all the isolates was performed using disc-diffusion (Modified-Kirby Baur method) according to CLSIs guidelines. Result: In present study, maximum isolates of Ps. aeruginosa isolated from various samples. The isolates were obtained from different clinical specimens, including pus, urine, respiratory fluids, blood, tissue, and genitalia. -

Progress in Pharmacokinetics and Pharmacodynamics - I
274 Abstracts Progress in pharmacokinetics and pharmacodynamics - I P1022 Pharmacokinetics of telithromycin in plasma and was higher in young women than in young men (21% difference), soft tissue after single-dose administration in healthy volunteers with only a 4% difference between elderly women and men. At the target clinical dose of 100 mg load infused over 30–60 min fol- R. Gattringer, F. Traunmueller, E. Urbauer, M. Zeitlinger, lowed by 50 mg q12h, Cmax and AUCss (mean Æ SD) were M. Mueller, C. Joukhadar 621 Æ 93 ng/mL and 3069 Æ 381 ng h/mL, respectively. Vienna, A Objectives: Telithromycin was described to reach high concentra- Dose (mg), with MD given q 12h tions levels in inflammatory fluid, in bronchopulmonary tissues and in tonsillar tissue. Because of these data telithromycin is spe- Pk parameter 12.5 25 50 75 100 200 300 culated to be a new option in the therapy of skin and soft tissue infections. To determine the concentration of telithromycin in the SD CLt 0.29 Æ 0.20 0.20 Æ 0.10 0.28 Æ 0.04 0.29 Æ 0.04 0.30 Æ 0.08 0.23 Æ 0.04 0.25 Æ 0.03 interstitial space fluid, the pharmacokinetics of this new antibiotic (L/hr/kg) (n ¼ 6) (n ¼ 6) (n ¼ 6) (n ¼ 6) (n ¼ 57) (n ¼ 24) (n ¼ 12) were assessed after single dose administration in young healthy MD CLt ÁÁÁ 0.20 Æ 0.04 0.20 Æ 0.02 ÁÁÁ 0.24 Æ 0.045 ÁÁÁ ÁÁÁ (L/hr/kg) (n ¼ 5) (n ¼ 5) (n ¼ 3) volunteers by the use of microdialysis.