Annotation-1 Sham Shock Shock + Trauma Trauma
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
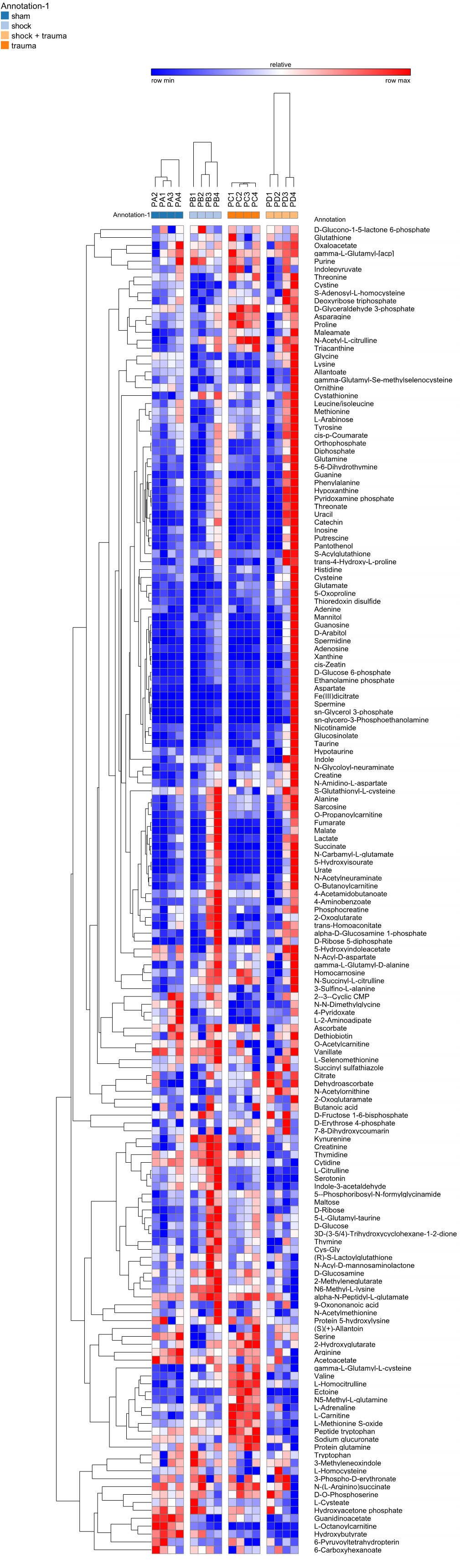
Annotation-1 Annotation-1
Annotation-1 Baseline Resuscitation Normal Saline Resuscitation PFP Shock Annotation-1 Aminoacids Arginine and proline metabolism Carnitine and fatty acid metabolsim Glutamate metabolism Glycerophospholipid biosynthesis Glycolysis and sugars GSH homeostasis GSH homeostasis/Glyoxlate Hexosamine Indole and Tryptophan Nucleotides Other Panthothenate metabolism Pentose Phosphate Pathway Serine biosynthesis and one-carbon metabolism Signaling Sulfur metabolism TCA cycle urea cycle relative row min row max Baseline_14 Baseline_16 Baseline_13 Baseline_15 Baseline_22 Baseline_2 Baseline_12 Baseline_3 Baseline_4 Baseline_9 Baseline_7 Baseline_8 Shock_13 Shock_12 Shock_15 Shock_22 Shock_14 Shock_16 Shock_2 Shock_3 Shock_7 Shock_4 Shock_8 Shock_9 Res_NS_14 Res_NS_13 Res_NS_16 Res_NS_12 Res_NS_22 Res_NS_15 Res_PFP_2 Res_PFP_3 Res_PFP_7 Res_PFP_4 Res_PFP_8 Res_PFP_9 Annotation-1 Annotation-1 Annotation Annotation-1 L-Arginine Aminoacids L-Isoleucine Aminoacids Leucine Aminoacids L-Cysteine Aminoacids L-Alanine Aminoacids L-Aspartate Aminoacids L-Glutamate Aminoacids L-Glutamine Aminoacids L-Histidine Aminoacids L-Lysine Aminoacids L-Methionine Aminoacids L-Tyrosine Aminoacids L-Asparagine Aminoacids L-Threonine Aminoacids L-Cystine Aminoacids L-Serine Aminoacids L-Proline Aminoacids L-Valine Aminoacids L-Tryptophan Aminoacids Glycine Aminoacids L-Kynurenine Aminoacids L-Phenylalanine Aminoacids CMP Nucleotides 6-Hydroxynicotinate Nucleotides 5-6-Dihydrouracil Nucleotides AMP Nucleotides dAMP Nucleotides GMP Nucleotides Guanine Nucleotides 2-5-Dihydroxypyridine -

35 Disorders of Purine and Pyrimidine Metabolism
35 Disorders of Purine and Pyrimidine Metabolism Georges van den Berghe, M.- Françoise Vincent, Sandrine Marie 35.1 Inborn Errors of Purine Metabolism – 435 35.1.1 Phosphoribosyl Pyrophosphate Synthetase Superactivity – 435 35.1.2 Adenylosuccinase Deficiency – 436 35.1.3 AICA-Ribosiduria – 437 35.1.4 Muscle AMP Deaminase Deficiency – 437 35.1.5 Adenosine Deaminase Deficiency – 438 35.1.6 Adenosine Deaminase Superactivity – 439 35.1.7 Purine Nucleoside Phosphorylase Deficiency – 440 35.1.8 Xanthine Oxidase Deficiency – 440 35.1.9 Hypoxanthine-Guanine Phosphoribosyltransferase Deficiency – 441 35.1.10 Adenine Phosphoribosyltransferase Deficiency – 442 35.1.11 Deoxyguanosine Kinase Deficiency – 442 35.2 Inborn Errors of Pyrimidine Metabolism – 445 35.2.1 UMP Synthase Deficiency (Hereditary Orotic Aciduria) – 445 35.2.2 Dihydropyrimidine Dehydrogenase Deficiency – 445 35.2.3 Dihydropyrimidinase Deficiency – 446 35.2.4 Ureidopropionase Deficiency – 446 35.2.5 Pyrimidine 5’-Nucleotidase Deficiency – 446 35.2.6 Cytosolic 5’-Nucleotidase Superactivity – 447 35.2.7 Thymidine Phosphorylase Deficiency – 447 35.2.8 Thymidine Kinase Deficiency – 447 References – 447 434 Chapter 35 · Disorders of Purine and Pyrimidine Metabolism Purine Metabolism Purine nucleotides are essential cellular constituents 4 The catabolic pathway starts from GMP, IMP and which intervene in energy transfer, metabolic regula- AMP, and produces uric acid, a poorly soluble tion, and synthesis of DNA and RNA. Purine metabo- compound, which tends to crystallize once its lism can be divided into three pathways: plasma concentration surpasses 6.5–7 mg/dl (0.38– 4 The biosynthetic pathway, often termed de novo, 0.47 mmol/l). starts with the formation of phosphoribosyl pyro- 4 The salvage pathway utilizes the purine bases, gua- phosphate (PRPP) and leads to the synthesis of nine, hypoxanthine and adenine, which are pro- inosine monophosphate (IMP). -

Clinical Symptoms of Defects in Pyrimidine Metabolism
ClinicalClinical symptomssymptoms ofof DefectsDefects inin pyrimidinepyrimidine metabolismmetabolism Birgit Assmann Department of General Pediatrics Universtiy Children‘s Hospital Düsseldorf, Germany Overview • Biosynthesis: UMP Synthase • Degradation: –– PyrimidinePyrimidine 55‘‘--Nucleotidase(UMPNucleotidase(UMP--Hydrolase)Hydrolase) – [Thymidine-Phosphorylase, mitochondrial] –– DihydropyrimidineDihydropyrimidine DehydrogenaseDehydrogenase –– DihydropyrimidinaseDihydropyrimidinase –– UreidopropionaseUreidopropionase HCO3+gluNH2 carbamoyl-P orotic acid OMP OPRT UMP OD UMPS UMPSUMPS == uridinemonophosphateuridinemonophosphate synthasesynthase Bifunctional enzyme (one gene): a) Orotate phosphoribosyl transferase (OPRT) b) Orotidine decarboxylase (OD) UMPS deficiency • = Hereditary orotic aciduria Hallmarks:Hallmarks: - MegaloblasticMegaloblastic anemiaanemia inin infantsinfants >> IfIf untreateduntreated:: FailureFailure toto thrivethrive PsychomotorPsychomotor retardationretardation • Therapy: uridine (≥100-150 mg/kg/d) Defects of pyrimidine degradation • Pyrimidine 5‘-Nucleotidase deficiency - chronic hemolytic anemia + basophilic stippling of erythrocytes • Thymidine phosphorylase deficiency = MNGIE=Mitoch. NeuroGastroIntestinal Encephalomyopathy Mitochondrial disorder with elevatedelevated urinaryurinary thymidinethymidine excretionexcretion HCO3+gluNH2 carbamoyl-P orotic acid OMP TMP UMP UMPS thymidine cytosolic 5‘- uridine Thym. Nucleotidase phosphor ylase thymine uracil Pyrimidine 5‘-Nucleotidase- SuperactivitySuperactivity • Existence -

Lc and Lc-Free Mass Spectrometry Applications in Non-Targeted Metabolomics for Disease Detection and Early Prediction
LC AND LC-FREE MASS SPECTROMETRY APPLICATIONS IN NON-TARGETED METABOLOMICS FOR DISEASE DETECTION AND EARLY PREDICTION A Dissertation Presented to The Academic Faculty by Xiaoling Zang In Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy in the School of Chemistry and Biochemistry Georgia Institute of Technology August 2018 COPYRIGHT © 2018 BY XIAOLING ZANG LC AND LC-FREE MASS SPECTROMETRY APPLICATIONS IN NON-TARGETED METABOLOMICS FOR DISEASE DETECTION AND EARLY PREDICTION Approved by: Dr. Facundo M. Fernández, Advisor Dr. Ronghu Wu School of Chemistry and Biochemistry School of Chemistry and Biochemistry Georgia Institute of Technology Georgia Institute of Technology Dr. María E. Monge, Co-advisor Dr. Matthew Torres Centro de Investigaciones en School of Biology Bionanociencias (CIBION) Georgia Institute of Technology Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) Dr. Julia Kubanek Dr. Mark Styczynski School of Chemistry and Biochemistry School of Chemical and Biomolecular Georgia Institute of Technology Engineering Georgia Institute of Technology Date Approved: July 23, 2018 Never give up on what you really want to do. The person with big dreams is more powerful than one with all the facts. -Albert Einstein, The World as I See It1 ACKNOWLEDGEMENTS There are many people who have helped and supported me a lot during my 6-year Ph.D. life at Georgia Tech. First of all, I want to express my deepest gratitude to my advisor, Dr. Facundo M. Fernández, for his guidance, support, patience and encouragement during my whole Ph.D. study. Under his mentorship over the last 6 years, I have gained so much knowledge and experience in mass spectrometry and have grown to become more independent and capable in handling the research projects. -

Behavior of Activities of Thymidine Metabolizing Enzymes in Human Leukemia-Lymphoma Cells1
[CANCER RESEARCH 49. 1090-1094. March I, 1989] Behavior of Activities of Thymidine Metabolizing Enzymes in Human Leukemia-Lymphoma Cells1 Taiichi Shiotani, Yasuko Hashimoto, Terukazu Tanaka, and Shozo Irino First Department of Internal Medicine, Kagawa Medical School, Ikenobe, Miki, Kagawa, 761-07, Japan ABSTRACT 7). These observations indicate the importance of this catabolic enzyme for dThd utilization. Thymidylate, an important pre The behavior of the activities of thymidine metabolizing enzymes, cursor of DNA synthesis, also may be produced by the de novo dihydrothymine dehydrogenase (EC 1.3.1.2) and thymidine phosphoryl- pathway through dTMP synthase. The correlation between cell ase (EC 2.4.2.4) for thymidine degradation, thymidine kinase (EC proliferation and dThd kinase (3, 4, 6, 8, 9) or dTMP synthase 2.7.1.75) and thymidylate synthase (EC 2.1.1.45) for DNA synthesis, was elucidated in cytosolic extracts from normal human lymphocytes and (8, 10, 11) has been demonstrated. The enhanced capacity for 13 human leukemia-lymphoma cell lines. In the normal human lympho the salvage pathway in leukemia (12, 13) appears to limit the cytes, the activities of dihydrothymine dehydrogenase, thymidine phos- antitumor effectiveness of antimetabolites of de novo pyrimidine phorylase, thymidine kinase, and thymidylate synthase were 6.88, 796, biosynthesis. The enzymic capacities of the de novo and salvage 0.30, and 0.29 nmol/h/mg protein, respectively. In leukemia-lymphoma pathways and dThd catabolism have not been determined si cell lines, the activities of synthetic enzymes, thymidine kinase, and multaneously in human leukemia-lymphoma cells. Therefore, thymidylate synthase, increased two- to 79-fold and 22- to 407-fold of the elucidation of the behavior of the enzymic capacities in the the normal lymphocyte values. -

Possible Prediction of Adverse Reactions to Pyrimidine Chemotherapy from Urinary Pyrimidine Levels and a Case of Asymptomatic Adult Dihydropyrimidinuria1
Vol. 2, 1937-1 941, December 1996 Clinical Cancer Research 1937 Possible Prediction of Adverse Reactions to Pyrimidine Chemotherapy from Urinary Pyrimidine Levels and a Case of Asymptomatic Adult Dihydropyrimidinuria1 Katsuo Hayashi,2 Kiyoshi Kidouchi, INTRODUCTION Satoshi Sumi, Masashi Mizokami, Etsuro Onto, Pyrimidine chemotherapy agents such as 5-FU3 are used widely but can occasionally cause serious adverse reactions. In Kazunoni Kumada, Ryuzo Ueda, and pyrimidine metabolism, uracil and thymine are first converted to Yoshino Wada dihydropyrimidines (dihydrouracil and dihydrothymine) by Departments of Medicine [K. H., K. Ku.] and Pediatrics [K. Ki.], DPD and then are metabolized to 3-ureidopropionic acid and Nagoya City Higashi General Hospital, 2-23 Wakamizu 1-chome, 3-ureidoisobutyric acid, respectively, by dihydropyrimidinase. Chikusa-ku, Nagoya 464, Japan, and Department of Pediatrics [S. S., These compounds are converted subsequently into 3-alanine Y. W.] and Second Department of Medicine [M. M., E. 0., R. U.], Nagoya City University Medical School. Nagoya, Japan and -arninoisobutyric acid, respectively ( 1). Because about 80% of an administered dose of 5-FU is degraded by this pathway, there is a possibility of adverse reactions occurring as ABSTRACT a result of DPD or dihydrophrimidinase deficiency. The value of Deficiency of dihydropyrimidine dehydrogenase or di- measuring peripheral blood monocyte DPD activity before ad- hydropyrimidinase, enzymes that catalyze the breakdown of ministering 5-FU has been suggested in Western countries (2- pyrimidine chemotherapy agents such as 5-fluorouracil, 6), but DPD deficiency has not been reported in Japan. may cause serious adverse reactions to these agents. We Using a method that permits analysis of dihydrouracil and attempted to establish the reference range for urinary pyri- uracil in small volumes of urine, we previously screened Japa- midines in adults to detect individuals with abnormal py- nese infants for abnormalities of pyrimidine metabolism and rimidine metabolism. -

Purine Metabolism Regulates DNA Repair and Therapy Resistance in Glioblastoma
ARTICLE https://doi.org/10.1038/s41467-020-17512-x OPEN Purine metabolism regulates DNA repair and therapy resistance in glioblastoma Weihua Zhou1,14, Yangyang Yao1,2,14, Andrew J. Scott1,3,14, Kari Wilder-Romans1, Joseph J. Dresser1, Christian K. Werner 1, Hanshi Sun1, Drew Pratt4, Peter Sajjakulnukit 5, Shuang G. Zhao1, Mary Davis1, Barbara S. Nelson5, Christopher J. Halbrook5, Li Zhang5, Francesco Gatto 6, Yoshie Umemura3,7, Angela K. Walker8, Maureen Kachman 8, Jann N. Sarkaria 9, Jianping Xiong2, Meredith A. Morgan1,3, Alnawaz Rehemtualla1,3, Maria G. Castro 3,10,11, Pedro Lowenstein 3,10,11, Sriram Chandrasekaran 3,12, ✉ Theodore S. Lawrence1,3, Costas A. Lyssiotis 3,5,13 & Daniel R. Wahl 1,3 1234567890():,; Intratumoral genomic heterogeneity in glioblastoma (GBM) is a barrier to overcoming therapy resistance. Treatments that are effective independent of genotype are urgently needed. By correlating intracellular metabolite levels with radiation resistance across dozens of genomically-distinct models of GBM, we find that purine metabolites, especially guany- lates, strongly correlate with radiation resistance. Inhibiting GTP synthesis radiosensitizes GBM cells and patient-derived neurospheres by impairing DNA repair. Likewise, adminis- tration of exogenous purine nucleosides protects sensitive GBM models from radiation by promoting DNA repair. Neither modulating pyrimidine metabolism nor purine salvage has similar effects. An FDA-approved inhibitor of GTP synthesis potentiates the effects of radiation in flank and orthotopic patient-derived xenograft models of GBM. High expression of the rate-limiting enzyme of de novo GTP synthesis is associated with shorter survival in GBM patients. These findings indicate that inhibiting purine synthesis may be a promising strategy to overcome therapy resistance in this genomically heterogeneous disease. -

British Journal of Nutrition (2015), 113, 560–573 Doi:10.1017/S0007114514004000 Q the Authors 2015
Downloaded from British Journal of Nutrition (2015), 113, 560–573 doi:10.1017/S0007114514004000 q The Authors 2015 https://www.cambridge.org/core Absorption and intermediary metabolism of purines and pyrimidines in lactating dairy cows Charlotte Stentoft1*, Betina Amdisen Røjen2, Søren Krogh Jensen1, Niels B. Kristensen2, . IP address: Mogens Vestergaard1 and Mogens Larsen1 1Department of Animal Science, Aarhus University, Foulum, Blichers Alle´ 20, DK-8830 Tjele, Denmark 170.106.35.229 2Knowledge Centre for Agriculture, DK-8200 Aarhus N, Denmark (Submitted 23 June 2014 – Final revision received 27 October 2014 – Accepted 9 November 2014 – First published online 26 January 2015) , on 30 Sep 2021 at 08:56:31 Abstract About 20 % of ruminal microbial N in dairy cows derives from purines and pyrimidines; however, their intermediary metabolism and contribution to the overall N metabolism has sparsely been described. In the present study, the postprandial patterns of net portal-drained viscera (PDV) and hepatic metabolism were assessed to evaluate purine and pyrimidine N in dairy cows. Blood was sampled simul- taneously from four veins with eight hourly samples from four multi-catheterised Holstein cows. Quantification of twenty purines and pyrimidines was performed with HPLC–MS/MS, and net fluxes were estimated across the PDV, hepatic tissue and total splanchnic , subject to the Cambridge Core terms of use, available at tissue (TSP). Concentration differences between veins of fifteen purine and pyrimidine nucleosides (NS), bases (BS) and degradation products (DP) were different from zero (P#0·05), resulting in the net PDV releases of purine NS (0·33–1·3 mmol/h), purine BS (0·0023–0·018 mmol/h), purine DP (7·0–7·8 mmol/h), pyrimidine NS (0·30–2·8 mmol/h) and pyrimidine DP (0·047–0·77 mmol/h). -

Nucleotides, Nucleic Acids: General Information About Structure, Functions and Metabolism
MINISTRY OF HEALTH OF UKRAINE ZAPORIZHZHIA STATE MEDICAL UNIVERSITY Biological Chemistry Department Nucleotides, Nucleic acids: General Information about Structure, Functions and Metabolism A manual for independent work at home and in class for students of second year study of international faculty Speciality “Medicine” Zaporizhzhia, 2016 1 UDC 577.1(075.8) BBC 28.902я73 N92 The manual was approved on the Central Methodological Council of ZSMU on «____» _______________2016, the protocol №________ Reviewers: Prykhodko O.B., Head of Medical Biology, Parasitology and Genetics Department of Zaporizhzhia State Medical University, doctor of biological science Voskoboynik O.Yu., associate professor of Organic and Bioorganic Chemistry Department of Zaporizhzhia State Medical University, PhD Editors: Dr. Hab., professor Aleksandrova K. V. PhD, assoc. professor Ivanchenko D. G. PhD, assoc. professor Krisanova N. V. Nucleotides, Nucleic acids : General Information about Structure, Functions and Metabolism : a manual for independent work at home and in class for students of second year study of international faculty, speciality ―Medicine‖/ ed. : K. V. Aleksandrova, D. G. Ivanchenko, N. V. Krisanova. – Zaporizhzhia : ZSMU, 2016.- 84 p. This manual is recommended to use for students of International Faculty (the second year of study) for independent work at home and in class. Нуклеотиди, нуклеїнові кислоти : загальне уявлення про структуру, функції та метаболізм : навч. посіб. для самостійної аудиторної та позааудиторної роботи студентів 2 курсу міжнар. ф-ту, спеціальність «Медицина» / ред.. : К. В. Александрова, Д. Г. Іванченко, Н. В. Крісанова. - Запоріжжя : ЗДМУ, 2016. – 84 с. UDC 577.1(075.8) BBC 28.902я73 ©Aleksandrova K.V., IvanchenkoD.G., Krisanova N.V., 2016 ©Zaporizhzhia State Medical University, 2016 2 INTRODUCTION A study of questions for this manual is the basis for learning of all metabolic pathways for nucleotides and nucleic acids. -

1 Synthesis of C-2 and C-6 Functionalized
1 SYNTHESIS OF C-2 AND C-6 FUNCTIONALIZED RIBOFURANOSYLPURINE ANALOGUES AS POTENTIAL ANTIVIRAL AGENTS TARGETING INHIBITION OF INOSINE MONOPHOSPHATE DEHYDROGENASE. by Eric Osei-Tutu Bonsu (Under the Direction of Vasu Nair) ABSTRACT IMPDH is a key enzyme in the de novo biosynthesis of purine nucleotides. It catalyzes the conversion of inosine monophosphate (IMP) to xanthosine monophosphate (XMP) using NAD as a cofactor. XMP is successively converted to deoxyguanosine triphosphate (substrate for DNA synthesis) by GMP synthetase, phosphorylating enzymes and ribonucleotide reductase. IMPDH exists in two isoforms, type I and type II. These isoforms have the same size and share 84% homology. The type I isoform is expressed in both normal and rapidly proliferating cells, whereas type II is preferentially upregulated in proliferating cells. Inhibition of IMPDH has anticancer, antiviral, antibacterial and immunosuppressive effects. Three inhibitors of IMPDH are currently in clinical use: ribavirin (a broad spectrum antiviral), mizoribine (immunosuppressant used in Japan) and mycophenolic mofetil (prodrug of mycophenolic acid, US approved immunosuppressant). None of these inhibitors possess significant selectivity against the type II isoform over the type I, hence there are severe side effects. The quest for specific isozyme inhibitors led to the discovery of the potential antiviral activity of certain C-2 functionalized hypoxanthine and C-2, C-6 modified purine systems against HSV1, HSV2, VV, VSV and RSV. 2 Nair and coworkers have synthesized similar congeners, including 2-vinylinosine (broad spectrum antiviral), which is active due to its C-2 vinyl moiety acting as a Michael Acceptor. This dissertation elucidates the design and synthesis of new Michael Acceptor-type nucleosides. -

Role of Glutathione Metabolism in Host Defense Against Borrelia
Role of glutathione metabolism in host defense PNAS PLUS against Borrelia burgdorferi infection Mariska Kerstholta,b, Hedwig Vrijmoetha,b, Ekta Lachmandasa,b, Marije Oostinga,b, Mihaela Lupsec, Mirela Flontac, Charles A. Dinarelloa,b,d,1, Mihai G. Neteaa,b,e, and Leo A. B. Joostena,b,1 aDepartment of Internal Medicine, Radboud University Medical Center, 6525 GA Nijmegen, The Netherlands; bRadboud Center for Infectious Diseases, Radboud University Medical Center, 6525 GA Nijmegen, The Netherlands; cDepartment of Infectious Diseases, University of Medicine and Pharmacy “Iuliu Hatieganu,” 400349 Cluj-Napoca, Romania; dDepartment of Medicine, University of Colorado Denver, Aurora, CO 80045; and eHuman Genomics Laboratory, Craiova University of Medicine and Pharmacy, 200349 Craiova, Romania Contributed by Charles A. Dinarello, January 18, 2018 (sent for review December 7, 2017; reviewed by Pietro Ghezzi and Georg Schett) Pathogen-induced changes in host cell metabolism are known to central glucose metabolism in host mononuclear cells which was be important for the immune response. In this study, we in- crucial for cytokine production (13). vestigated how infection with the Lyme disease-causing bacterium In the present study, we aimed to further explore the metabolic Borrelia burgdorferi (Bb) affects host metabolic pathways and pathways induced by Bb and analyze their role in immune cell how these metabolic pathways may impact host defense. First, function. To achieve this, we performed metabolomic analysis metabolome analysis was performed on human primary mono- of primary human monocytes stimulated with Bb or other in- cytes from healthy volunteers, stimulated for 24 h with Bb at flammatory stimuli. Identified pathways were then further validated low multiplicity of infection (MOI). -

DPYD Gene Dihydropyrimidine Dehydrogenase
DPYD gene dihydropyrimidine dehydrogenase Normal Function The DPYD gene provides instructions for making an enzyme called dihydropyrimidine dehydrogenase, which is involved in the breakdown of molecules called uracil and thymine when they are not needed. Uracil and thymine are pyrimidines, which are one type of nucleotide. Nucleotides are building blocks of DNA, its chemical cousin RNA, and molecules such as ATP and GTP that serve as energy sources in the cell. Dihydropyrimidine dehydrogenase is involved in the first step of the breakdown of pyrimidines. This enzyme converts uracil to another molecule called 5,6-dihydrouracil and converts thymine to 5,6-dihydrothymine. The molecules created when pyrimidines are broken down are excreted by the body or used in other cellular processes. Health Conditions Related to Genetic Changes Dihydropyrimidine dehydrogenase deficiency More than 50 mutations in the DPYD gene have been identified in people with dihydropyrimidine dehydrogenase deficiency. DPYD gene mutations interfere with the breakdown of uracil and thymine and result in excess quantities of these molecules in the blood, urine, and the fluid that surrounds the brain and spinal cord (cerebrospinal fluid). It is unclear how the excess uracil and thymine are related to the specific neurological problems that affect some people with dihydropyrimidine dehydrogenase deficiency. Mutations in the DPYD gene also interfere with the breakdown of drugs with structures similar to the pyrimidines, such as the cancer drugs 5-fluorouracil and capecitabine.