(12) Patent Application Publication (10) Pub. No.: US 2002/0082444 A1 Bonnard Et Al
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Chemical Threat Agents Call Poison Control 24/7 for Treatment Information 1.800.222.1222 Blood Nerve Blister Pulmonary Metals Toxins
CHEMICAL THREAT AGENTS CALL POISON CONTROL 24/7 FOR TREATMENT INFORMATION 1.800.222.1222 BLOOD NERVE BLISTER PULMONARY METALS TOXINS SYMPTOMS SYMPTOMS SYMPTOMS SYMPTOMS SYMPTOMS SYMPTOMS • Vertigo • Diarrhea, diaphoresis • Itching • Upper respiratory tract • Cough • Shock • Tachycardia • Urination • Erythema irritation • Metallic taste • Organ failure • Tachypnea • Miosis • Yellowish blisters • Rhinitis • CNS effects • Cyanosis • Bradycardia, bronchospasm • Flu-like symptoms • Coughing • Shortness of breath • Flu-like symptoms • Emesis • Delayed eye irritation • Choking • Flu-like symptoms • Nonspecific neurological • Lacrimation • Delayed pulmonary edema • Visual disturbances symptoms • Salivation, sweating INDICATIVE LAB TESTS INDICATIVE LAB TESTS INDICATIVE LAB TEST INDICATIVE LAB TESTS INDICATIVE LAB TESTS INDICATIVE LAB TESTS • Increased anion gap • Decreased cholinesterase • Thiodiglycol present in urine • Decreased pO2 • Proteinuria None Available • Metabolic acidosis • Increased anion gap • Decreased pCO2 • Renal assessment • Narrow pO2 difference • Metabolic acidosis • Arterial blood gas between arterial and venous • Chest radiography samples DEFINITIVE TEST DEFINITIVE TEST DEFINITIVE TEST DEFINITIVE TESTS DEFINITIVE TESTS • Blood cyanide levels • Urine nerve agent • Urine blister agent No definitive tests available • Blood metals panel • Urine ricinine metabolites metabolites • Urine metals panel • Urine abrine POTENTIAL AGENTS POTENTIAL AGENTS POTENTIAL AGENTS POTENTIAL AGENTS POTENTIAL AGENTS POTENTIAL AGENTS • Hydrogen Cyanide -

(12) Patent Application Publication (10) Pub. No.: US 2005/0065361A1 Deshmukh Et Al
US 2005OO65361A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2005/0065361A1 Deshmukh et al. (43) Pub. Date: Mar. 24, 2005 (54) PROCESS FOR PREPARING ALKYLARYL (22) Filed: Sep. 22, 2003 CHLOROFORMATES Publication Classification (76) Inventors: Abdul Rakeeb Abdul Subhan Deshmukh, Maharashtra (IN); Vikas (51) Int. Cl." ........................... C07C 69/74; C07C 69/96 Kalyanrao Gumaste, Maharashtra (IN) (52) U.S. Cl. .............................................................. 558/280 Correspondence Address: (57) ABSTRACT NIXON & VANDERHYE, PC The present invention discloses an improved method for the 1100 N GLEBE ROAD preparation of alky/aryl chloroformates directly from alco 8TH FLOOR hols and triphosgene. This method is simple, mild and ARLINGTON, VA 22201-4714 (US) efficient avoids use of hazardous phosgene. It can be used for the preparation of various aryl as well as alkyl chlorofor (21) Appl. No.: 10/665,410 mates in excellent yields. US 2005/0065361 A1 Mar. 24, 2005 PROCESS FOR PREPARING ALKYLARYL Maligres, K. C. Nicolau, W. Wrasidio Bioorg. Med. Chem. CHLOROFORMATES Lett. 1993, 3, 1051. (c) D. C. Horwell, J. Hughes, J. Hunter, M. C. Pritchard, R. S. Richardson, E. Roberts, G. N. FIELD OF THE INVENTION Woodruff J. Med. Chem., 1991, 34, 404 and tertiary amines as base H. Eckert, B. Forster, Angew. Chem. Int. Ed. Engl., 0001. The present invention relates to a process for 1987,26,894). Hydroquinone is also used in the preparation preparing alkyl/aryl chloroformates. More particularly, the of chloroformates from triphosgene G. Van den Mooter, C. present invention relates to a process for preparing com Samyn, R. Kinget Int. J. Pharm., 1993, 97, 133). -

Nerve Agent - Lntellipedia Page 1 Of9 Doc ID : 6637155 (U) Nerve Agent
This document is made available through the declassification efforts and research of John Greenewald, Jr., creator of: The Black Vault The Black Vault is the largest online Freedom of Information Act (FOIA) document clearinghouse in the world. The research efforts here are responsible for the declassification of MILLIONS of pages released by the U.S. Government & Military. Discover the Truth at: http://www.theblackvault.com Nerve Agent - lntellipedia Page 1 of9 Doc ID : 6637155 (U) Nerve Agent UNCLASSIFIED From lntellipedia Nerve Agents (also known as nerve gases, though these chemicals are liquid at room temperature) are a class of phosphorus-containing organic chemicals (organophosphates) that disrupt the mechanism by which nerves transfer messages to organs. The disruption is caused by blocking acetylcholinesterase, an enzyme that normally relaxes the activity of acetylcholine, a neurotransmitter. ...--------- --- -·---- - --- -·-- --- --- Contents • 1 Overview • 2 Biological Effects • 2.1 Mechanism of Action • 2.2 Antidotes • 3 Classes • 3.1 G-Series • 3.2 V-Series • 3.3 Novichok Agents • 3.4 Insecticides • 4 History • 4.1 The Discovery ofNerve Agents • 4.2 The Nazi Mass Production ofTabun • 4.3 Nerve Agents in Nazi Germany • 4.4 The Secret Gets Out • 4.5 Since World War II • 4.6 Ocean Disposal of Chemical Weapons • 5 Popular Culture • 6 References and External Links --------------- ----·-- - Overview As chemical weapons, they are classified as weapons of mass destruction by the United Nations according to UN Resolution 687, and their production and stockpiling was outlawed by the Chemical Weapons Convention of 1993; the Chemical Weapons Convention officially took effect on April 291997. Poisoning by a nerve agent leads to contraction of pupils, profuse salivation, convulsions, involuntary urination and defecation, and eventual death by asphyxiation as control is lost over respiratory muscles. -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -
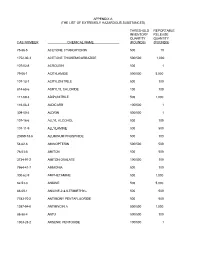
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

Chapter 6 Industrial Applications of Multicomponent Reactions (Mcrs)
University of Groningen Innovative multicomponent reactions and their use in medicinal chemistry Zarganes Tzitzikas, Tryfon IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document version below. Document Version Publisher's PDF, also known as Version of record Publication date: 2017 Link to publication in University of Groningen/UMCG research database Citation for published version (APA): Zarganes Tzitzikas, T. (2017). Innovative multicomponent reactions and their use in medicinal chemistry. University of Groningen. Copyright Other than for strictly personal use, it is not permitted to download or to forward/distribute the text or part of it without the consent of the author(s) and/or copyright holder(s), unless the work is under an open content license (like Creative Commons). Take-down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. Downloaded from the University of Groningen/UMCG research database (Pure): http://www.rug.nl/research/portal. For technical reasons the number of authors shown on this cover page is limited to 10 maximum. Download date: 24-09-2021 CHAPTER 6 INDUSTRIAL APPLICATIONS OF MULTICOMPONENT REACTIONS (MCRS) Chapter contained in the Rodriguez-Bonne book Stereoselectve Multple Bond-Forming Transformatons in Organic Synthesis 2015 by John Wiley & Sons, Inc. Tryfon Zarganes – Tzitzikas, Ahmad Yazbak, Alexander Dömling Chapter 6 INTRODUCTION Multcomponent reactons (MCRs) can be defned as processes in which three or more reactants introduced simultaneously are combined through covalent bonds to form a single product, regardless of the mechanisms and protocols involved.[1] Many basic MCRs are name reactons, for example, Ugi,[2] Passerini,[3] van Leusen,[4] Strecker,[5] Hantzsch,[6] Biginelli,[7] or one of their many variatons. -

Toxic Industrial Chemicals
J R Army Med Corps 2002; 148: 371-381 J R Army Med Corps: first published as 10.1136/jramc-148-04-06 on 1 December 2002. Downloaded from Toxic Industrial Chemicals Introduction location to another. Depending on the The first chemical warfare agent of the available routes of movement, and quantity modern era, chlorine, was released with of chemical to be moved, transport can occur devastating effect on 22 April 1915 at Ypres, by truck or rail tank cars, over water by barge Belgium. Along a 4 mile front, German or boat, over land through above- or below- soldiers opened the valves of 1,600 large and ground pipelines and by air. 4,130 small cylinders containing 168 tons of Toxic chemicals may be produced by the chlorine.The gas formed a thick white cloud burning of materials (e.g., the burning of that crossed the first allied trenches in less Teflon produces perfluoroisobutylene) or by than a minute.The allied line broke, allowing their reaction if spilled into water (e.g. silanes the Germans to advance deep into allied produce hydrogen chloride and cyanides, territory. If the Germans had been fully hydrogen cyanide). prepared to exploit this breakthrough, the course and possibly the outcome of WWI Toxic Industrial Chemicals may have been very different. (TICs) Chlorine is a commodity industrial A Toxic Industrial Chemical (TIC) is defined chemical with hundreds of legitimate uses; it as: is not a "purpose designed" chemical warfare an industrial chemical which has a LCt50 agent. Phosgene, another commodity value of less than 100,000 mg.min/m3 in industrial chemical, accounted for 80% of any mammalian species and is produced in the chemical fatalities during WWI. -

Piratox Sheet N5 Suffocating Agents and Phosphine
Edition of October 2011 Piratox sheet #5: "Suffocating agents and phosphine Indicative list of concerned agents: Toxic compounds with suffocating action - Phosgene (CAS number: 75-44-5) - Chlorine (CAS number: 7782-50-5) - Methyl isocyanate (CAS number: 624-83-9) - But also: o Ammonia (CAS number: 7664-41-7) o Diphosgene or surpalite (CAS number: 503-38-8) o Chloropicrin (CAS number: 76-06-2) o Fluorine (CAS number: 7782-41-4) o Perfluoroisobutylene (CAS number: 382-21-8) o Fumigants o Marketed industrial and domestic products - Phosphine or PH3 or Hydrogen Phosphide (CAS number: 7803-51-2). ! Key points not to forget The 1st emergency step is the extraction of victims from the hazard area: rescuers must possess suitable breathing and eye protection. At ambient temperature (20°C) most suffocating agents are gases that penetrate the body via the respiratory route. They are thus poorly or non-persistent, frequently limiting victim decontamination needs to simple undressing. Most suffocating agents are heavier than air. They affect the respiratory system: glottis, bronchi, alveoli and cause eye damage. In pre-hospital settings, avoid any physical exertion that could promote the onset of pulmonary oedema. In general, the shorter the symptom onset time, the more serious the intoxication and the more severe the symptoms. Treatment is symptomatic only. All symptomatic choking gas victims must encouraged to rest, in a sitting position, under oxygen. The duration of surveillance for symptomatic subjects is of at least 12 to 24 hours. For Phosphine (PH3): o victims must be undressed and showered; o toxicity is respiratory, cardiac, renal and neurological; o subjects having inhaled PH3 and presenting with significant initial manifestations shall be monitored in the hospital for 48 to 72 hours, due to the risk of delayed acute pulmonary oedema. -

Benzyl Chlorid Final
Survey of benzyl chloride (CAS no. 100-44-7) Part of the LOUS-review [Series Title and year] Consultation draft Title: Editing: Survey of benzyl chloride (CAS no. 100-44-7) Pia Brunn Poulsen, FORCE Technology Maria Strandesen, FORCE Technology Anders Schmidt, FORCE Technology Published by: Photography: The Danish Environmental Protection Agency [Name] Strandgade 29 1401 Copenhagen K Denmark Illustration: www.mst.dk/english [Name] Year: Map: [xxxx] [Name] ISBN no. [xxxxxx] Disclaimer: When the occasion arises, the Danish Environmental Protection Agency will publish reports and papers concerning research and development projects within the environmental sector, financed by study grants provided by the Danish Environmental Protection Agency. It should be noted that such publications do not necessarily reflect the position or opinion of the Danish Environmental Protection Agency. However, publication does indicate that, in the opinion of the Danish Environmental Protection Agency, the content represents an important contribution to the debate surrounding Danish environmental policy. While the information provided in this report is believed to be accurate, The Danish Environmental Protection Agency disclaims any responsibility for possible inaccuracies or omissions and consequences that may flow from them. Neither the Danish Environmental Protection Agency nor FORCE Technology or any individual involved in the preparation of this publication shall be liable for any injury, loss, damage or prejudice of any kind that may be caused by persons who have acted based on their understanding of the information contained in this publication. Sources must be acknowledged. 2 Survey of benzyl chloride (CAS no. 100-44-7) Contents Preface ...................................................................................................................... 6 Summary and conclusions ......................................................................................... 8 Sammenfatning og konklusion ............................................................................... -

The Preparation of Certain Organic Chloroformates and Carbonates
Brigham Young University BYU ScholarsArchive Theses and Dissertations 1947-04-01 The preparation of certain organic chloroformates and carbonates Robert E. Brailsford Brigham Young University - Provo Follow this and additional works at: https://scholarsarchive.byu.edu/etd BYU ScholarsArchive Citation Brailsford, Robert E., "The preparation of certain organic chloroformates and carbonates" (1947). Theses and Dissertations. 8175. https://scholarsarchive.byu.edu/etd/8175 This Thesis is brought to you for free and open access by BYU ScholarsArchive. It has been accepted for inclusion in Theses and Dissertations by an authorized administrator of BYU ScholarsArchive. For more information, please contact [email protected], [email protected]. THE PREPARATI OH OF CERTAI N 0RG.1NIC CHLOROFORJ.'\Ll.TES AND CARBON T Thesis ubmitted to the Department of Chemistry Brigham Young University ~ .. ... "') .~ . ~ . "'). ... .. .. .. .. , ... .. ... : : ....: . ..-. ~ ..·.: : ..: ...• : ·.. ~ . : ,.. .~ : :. : ·: : ··.... ; ~ : ·. : : .: . : . : : : ••• .... •." •,.r_·: -••• ~ .... In Parti a l Fulfillment of the Re~uirements for the Degree Master of cienoe 147143 by Robert E. Brailsford .tipril 1947 This Thesis by Robert E. Brailsford is accepted in its present farm by the Department of Chemistry of Brigham Young University as satisfying the ·rhesis requirement for the degree of Master of Science. PREFACE flhile working for the Hooker Electrochemical Company of Niagara Falls , New York , from April 3 , 1943 , to January 30 , 1946 , the writer became interested in organic chloroformates and. carbonates , an interest instigated by requests from B. F . Goodrich Company for a number of samples . fter returning to Brigham Young University that preliminary interest was revived and the experi - mental work of this thesis was performed. under the direction of Dr . Charles ' . :Maw and Professor Joseph K. -

Carbonate and Benzyl Benzotriazol-L-Yl Carbonate. Now
70 Bull. Korean Chem. Soc., Vol. 7, No. 1, 1986 Sunggak Kim and Heung Chang t-Butyl Benzotriazol-l-yl Carbonate and Benzyl Benzotriazol-l-yl Carbonate. Now Reactive Amino Protective Reagents for t-Butoxy carbonylation and Benzyloxycarbonylation of Amines and Amino Acids Sunggak Kim* and Heung Chang Department of Chemistry, Korea Advanced Institute of Science and Technology, Seoul 131, Received September24,1985 New amino protective reagents, /-butyl benzotriazol-l-yl carbonate and benzyl benzotriazol-l-yl carbonate, for t~ butoxycarbonylation and benzyloxycarbonylation of amines and amino acids have been developed. f-Butyl benzotriazol-1 -yl carbonate reacts rapidly and cleanly with various amines and amino acids to afford N-Boc amines and N-Boc amino acids in high yields and benzyl benzotriazol-l-yl carbonate is also found to be very effective in the benzyloxycarbonylation of amino acids. Introduction a solution of an equimolar amount of 1-hydroxybenzotriazole and pyridine in methylene chloride to the solution of an ex The Z-butoxycarbonyl (Boc) group is one of the most im cess amount of phosgene in toluene under cooling portant amino protective groups along with benzyloxycarbonyl (-20~ 一 10°C). The resulting product was relatively unstable (Cbz) group in peptide synthesis.1 Since /-butyl chloroformate and was mainly decomposed to 1-hydroxybenzotriazole dur is only fairly stable above 10°C,2 it is difficult to is이 ate t- ing workup with cold water. Thus; benzotriazol-l-yl chlorofor butyl chloroformate as a pure form in high yields. Thus, con mate was used in a crude form. BBC could be prepared readily siderable efforts have been devoted to the development of a by the reaction of benzotriazol-l-yl chloroformate with variety of useful and reliable reagents for the preparation of equimolar amounts of f나 mtyl alcohol and pyridine in N-Boc amino acids during last 30 years.1 methylene chloride at room temperature for 2 h (eq. -

Combustion and Pyrolysis Kinetics of Chloropicrin J.-C
Combustion and Pyrolysis Kinetics of Chloropicrin J.-C. Lizardo-Huerta, B. Sirjean, L. Verdier, R. Fournet, Pierre-Alexandre Glaude To cite this version: J.-C. Lizardo-Huerta, B. Sirjean, L. Verdier, R. Fournet, Pierre-Alexandre Glaude. Combustion and Pyrolysis Kinetics of Chloropicrin. Journal of Physical Chemistry A, American Chemical Society, 2018, 122 (26), pp.5735 - 5741. 10.1021/acs.jpca.8b04007. hal-01921757 HAL Id: hal-01921757 https://hal.univ-lorraine.fr/hal-01921757 Submitted on 14 Nov 2018 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Combustion and Pyrolysis Kinetics of Chloropicrin J.-C. Lizardo-Huerta1, B. Sirjean1, L. Verdier2, R. Fournet1, P.-A. Glaude1* 1 Laboratoire Réactions et Génie des Procédés, CNRS, Université de Lorraine 1 rue Grandville BP 20451 54001 Nancy Cedex, France 2 DGA Maîtrise NRBC, Site du Bouchet, 5 rue Lavoisier, BP n°3, 91710 Vert le Petit, France Corresponding author : Pierre-Alexandre Glaude Laboratoire Réactions et Génie des Procédés 1 rue Grandville BP 20451 54001 Nancy Cedex, France Email:[email protected] 1 Abstract Chloropicrin (CCl3NO2) is widely used in agriculture as a pesticide, weed-killer, fungicide or nematicide.