Benzyl Chloride)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Catalytic Oxidation of Benzyl Alcohol Using Nanosized Cu/Ni Schiff-Base Complexes and Their Metal Oxide Nanoparticles
catalysts Article Catalytic Oxidation of Benzyl Alcohol Using Nanosized Cu/Ni Schiff-Base Complexes and Their Metal Oxide Nanoparticles Sameerah I. Al-Saeedi 1, Laila H. Abdel-Rahman 2,*, Ahmed M. Abu-Dief 2, Shimaa M. Abdel-Fatah 2, Tawfiq M. Alotaibi 3, Ali M. Alsalme 4 and Ayman Nafady 4,* 1 Department of Chemistry, College of Science, Princess Nourah bint Abdulrahman University, Riyadh 11451, Saudi Arabia; [email protected] 2 Chemistry Department, Faculty of Science, Sohag University, Sohag 82534, Egypt; [email protected] (A.M.A.-D.); [email protected] (S.M.A.-F.) 3 King Abdullah City for Atomic and Renewable Energy, Riyadh 11451, Saudi Arabia; tawfi[email protected] 4 Chemistry Department, College of Science, King Saud University, Riyadh 11451, Saudi Arabia; [email protected] * Correspondence: [email protected] (L.H.A.R.); [email protected] (A.N.); Tel.: +966-569-407-110 (A.N.) Received: 26 September 2018; Accepted: 10 October 2018; Published: 13 October 2018 Abstract: In this work, nanosized Cu and Ni Schiff-base complexes, namely ahpvCu, ahpnbCu, and ahpvNi, incorporating imine ligands derived from the condensation of 2-amino-3-hydroxypyridine, with either 3-methoxysalicylaldehyde (ahpv) or 4-nitrobenzaldehyde (ahpnb), were synthesized using sonochemical approach. The structure and properties of the new ligands and their complexes with Ni(II) and Cu(II) were determined via infrared (IR), nuclear magnetic resonance (NMR), electronic spectra (UV-Vis), elemental analysis (CHN), thermal gravimetric analysis (TGA), molar conductivity (Lm), and magnetic moment (meff). The combined results revealed the formation of 1:1 (metal: ligand) complexes for ahpvCu and ahpvNi and 1:2 for ahpnbCu. -

Transport of Dangerous Goods
ST/SG/AC.10/1/Rev.16 (Vol.I) Recommendations on the TRANSPORT OF DANGEROUS GOODS Model Regulations Volume I Sixteenth revised edition UNITED NATIONS New York and Geneva, 2009 NOTE The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. ST/SG/AC.10/1/Rev.16 (Vol.I) Copyright © United Nations, 2009 All rights reserved. No part of this publication may, for sales purposes, be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, electrostatic, magnetic tape, mechanical, photocopying or otherwise, without prior permission in writing from the United Nations. UNITED NATIONS Sales No. E.09.VIII.2 ISBN 978-92-1-139136-7 (complete set of two volumes) ISSN 1014-5753 Volumes I and II not to be sold separately FOREWORD The Recommendations on the Transport of Dangerous Goods are addressed to governments and to the international organizations concerned with safety in the transport of dangerous goods. The first version, prepared by the United Nations Economic and Social Council's Committee of Experts on the Transport of Dangerous Goods, was published in 1956 (ST/ECA/43-E/CN.2/170). In response to developments in technology and the changing needs of users, they have been regularly amended and updated at succeeding sessions of the Committee of Experts pursuant to Resolution 645 G (XXIII) of 26 April 1957 of the Economic and Social Council and subsequent resolutions. -

Business Guidance on Phthalates
Business guidance on phthalates How to limit phthalates of concern in articles? November 2013 Danish branch of EPBA, Brussels This guidance has been prepared on the initiative of the Danish EPA and organisations, DI (the Confederation of Danish Industry), the Danish Chamber of Commerce, DI ITEK (the Danish ICT and electronics federation for it, telecommunications, electronics and communication enterprises), BFE (the Danish Consumer Electronics Association), Batteriforeningen (the Danish branch of the European battery association EPBA), ITB (the Danish IT Industry Association) and FEHA (the Danish Association for Suppliers of Electrical Domestic Appliances) in a collaboration between the Danish EPA and representatives from the organisations. The guidance applies to companies marketing articles to either private or industrial users in Denmark. The target group is buyers in Danish companies that import or act as commercial agents, intermediaries or retailers, as well as foreign companies that export to Denmark. 2 Content How to limit phthalates of concern in articles? ................................................................... 4 Key to identification of articles with phthalates .................................................................. 8 Facts on phthalates ............................................................................................................................. 10 What should you ask your supplier? .......................................................................................... 11 What are the -

A Simple, Fast, and Reliable LC-MS/MS Method for Determination and Quantification of Phthalates in Distilled Beverages
A Simple, Fast, and Reliable LC-MS/MS Method for Determination and Quantification of Phthalates in Distilled Beverages Dimple Shah and Jennifer Burgess Waters Corporation, Milford, MA, USA APPLICATION BENEFITS INTRODUCTION ■■ Separation and detection of 7 phthalates Phthalates, esters of phthalic acid, are often used as plasticizers for polymers such in 11 minutes. as polyvinylchloride. They are widely applicable in various products including personal care goods, cosmetics, paints, printing inks, detergents, coatings, ■■ Simple, quick “dilute and shoot” sample preparation method for routine applications and food packaging. These phthalates have been found to leach readily into the where high sample throughput is required. environment and food as they are not chemically bound to plastics. As such they are known to be ubiquitously present in our environment. ■■ Elimination of major phthalate contamination using the ACQUITY UPLC® Isolator Column. Phthalates have been reported to show a variety of toxic effects related to reproduction in animal studies, which has resulted in these compounds being ■■ Selective mass spectra obtained for all seven considered as endocrine disruptors. Screening food and beverages for phthalates phthalates with dominant precursor ion. contamination is required by many legislative bodies, although regulations vary ■■ Low limits of quantification achieved using from country to country in regards to acceptable daily tolerances and specific Waters® ACQUITY UPLC H-Class System migration limits. and Xevo® TQD. Traditionally, phthalates have been analyzed by gas chromatography-mass ■■ Single and robust method for a variety spectrometry (GC-MS), where derivatization and/or extraction and sample of distilled beverages. preparation is often required to improve chromatographic separation.1 The ■■ Maintain consumer confidence and resulting GC-EI-MS spectra can lack selectivity, where the base ion, used for ensure compliance in market. -

Benzyl Alcohol Formulation
SAFETY DATA SHEET Benzyl Alcohol Formulation Version Revision Date: SDS Number: Date of last issue: 10.10.2020 1.2 09.04.2021 4702904-00003 Date of first issue: 29.07.2019 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1 Product identifier Trade name : Benzyl Alcohol Formulation 1.2 Relevant identified uses of the substance or mixture and uses advised against Use of the Sub- : Veterinary product stance/Mixture 1.3 Details of the supplier of the safety data sheet Company : MSD 20 Spartan Road 1619 Spartan, South Africa Telephone : +27119239300 E-mail address of person : [email protected] responsible for the SDS 1.4 Emergency telephone number +1-908-423-6000 SECTION 2: Hazards identification 2.1 Classification of the substance or mixture Classification (REGULATION (EC) No 1272/2008) Not a hazardous substance or mixture. 2.2 Label elements Labelling (REGULATION (EC) No 1272/2008) Not a hazardous substance or mixture. Additional Labelling EUH210 Safety data sheet available on request. 2.3 Other hazards This substance/mixture contains no components considered to be either persistent, bioaccumula- tive and toxic (PBT), or very persistent and very bioaccumulative (vPvB) at levels of 0.1% or higher. SECTION 3: Composition/information on ingredients 3.2 Mixtures Components Chemical name CAS-No. Classification Concentration 1 / 13 SAFETY DATA SHEET Benzyl Alcohol Formulation Version Revision Date: SDS Number: Date of last issue: 10.10.2020 1.2 09.04.2021 4702904-00003 Date of first issue: 29.07.2019 EC-No. (% w/w) Index-No. Registration number Benzyl alcohol 100-51-6 Acute Tox. -

Floor Polishing Composition
Europaisches Patentamt 0 358 984 J European Patent Office (p) Publication number: A2 Office europeen des brevets © EUROPEAN PATENT APPLICATION £) Application number: 89115478.3 (f) int. ci.5- C09G 1/16 © Date of filing: 22.08.89 ® Priority: 16.09.88 US 245427 © Applicant: STERLING DRUG INC. 90 Park Avenue © Date of publication of application: New York New York(US) 21.03.90 Bulletin 90/12 @ Inventor: Jethwa, Anil Parmanand © Designated Contracting States: 1363 Omara Drive AT BE CH DE ES FR GB GR IT LI NL SE Union New Jersey 07083(US) ™) Representative: Baillie, lain Cameron et al c/o Ladas & Parry Isartorplatz 5 D-8000 Munchen 2(DE) © Floor polishing composition. © An aqueous cleaning, coating and polishing composition consisting essentially of 1 to 20% by weight of a water insoluble alkali soluble addition polymer comprising at least one hydrophilic monomer selected from acrylic acid and methacrylic acid and at least one hydrophobic monomer selected from alkyl acrylate and alkyl methacrylate, where aikyl has from 1 to 6 carbon atoms, and at least one hydrophobic monomer selected from styrene and monoalkylstyrene where monoalkyl has from 1 to 6 carbon atoms, 1 to 13% by weight of an alkali soluble 2:1 to 3:1 styrene:acrylic acid resin, 5 to 15% by weight of a fugitive plasticizer, 1 to 3% by weight of a permanent plasticizer, 0.01 to 0.05% by weight of an anionic or nonionic fluorocarbon surfactant leveling agent, 0.0003 to 0.003% by weight of a dimethylpolysiloxane antifoaming agent; 0.0003 to 0.003% by weight of a 1 ,2- benzoisothiazol-3(2H)-one or 3(2H)-isothazolone preservative, ammonia to provide a pH of 8.0 to 9.6 and water q.s. -

BMF 32 - Plasticizers: Phthalates and Adipates
BMF 32 - Plasticizers: Phthalates and Adipates Phthalate and adipate esters are widely used as surfactants and in food and per- sonal care products as plasticizers. Phthalates and adipates are also found in food as a result of leakage into food containers. Chiron offers a wide range of analytically pure deuterated and native phthalates and adipates, as well as mixes. Custom made mixes are available on request. Metabolites: Phthalate biomarkers It has been shown that dialkylphthalates are metabolized in the body to endocrine-active monoalkyl- phthalates, and appears to be linked with the prevalence of abdominal obesity and insulin resistance in men. General information Neat Compounds: Most of the compounds are also available in neat form. Please inquire for an offer. Custom solutions: Please inquire for custom solution of the plasticizers. Please inquire if you have need for others. Other labelled plasticizers are available upon request (deuterium or 13 C). Description of pr. no.: iso- octane (IO), isopropanol (IP), n-hexane (HX), mixture of solvents (MX). Phthalates, adipate and similar plasticizers Phthalates Deuterium labelled phthalates 2478.10-100-IO Dimethyl phthalate-3,4,5,6-d4 2892.12-K-IO Diethyl phthalate-3,4,5,6-d4 9365.14-100-IO Di-n-propyl phthalate-3,4,5,6-d4 9926.16-100-IO n-Butyl-2-methylpropyl phthalate-3,4,5,6-d4 2479.16-100-IO Di-n-butyl phthalate-3,4,5,6-d4 9925.18-100-IO n-Pentyl-3-methylbutyl phthalate-3,4,5,6-d4 2893.18-100-IO Di-n-pentyl phthalate-3,4,5,6-d4 2893.18-100-HX Di-n-pentyl phthalate-3,4,5,6-d4 2893.18-K-IO Di-n-pentyl phthalate-3,4,5,6-d4 3125.18-100-IO Benzyl-n-butyl phthalate-3,4,5,6-d4 9366.20-100-IO Dicyclohexyl phthalate-3,4,5,6-d4 9367.20-100-IO Di-n-hexyl phthalate-3,4,5,6-d4 2361.24-100-IO Di-n-octyl phthalate-3,4,5,6-d4 2361.24-100-IP Di-n-octyl phthalate-3,4,5,6-d4 2894.24-K-IO Bis(2-ethylhexyl) phthalate-3,4,5,6-d4 Monophthalate esters (metabolites) 3805. -

RAFT Memorabilia : Living Radical Polymerization in Homogeneous and Heterogeneous Media / by Hans De Brouwer
RAFT memorabilia ❉ living ❉ radical polymerization in homogeneous and heterogeneous media Hans de Brouwer CIP-DATA LIBRARY TECHNISCHE UNIVERSITEIT EINDHOVEN Brouwer, Hans de RAFT memorabilia : living radical polymerization in homogeneous and heterogeneous media / by Hans de Brouwer. - Eindhoven : Technische Universiteit Eindhoven, 2001. - Proefschrift. - ISBN 90-386-2802-1 NUGI 813 Trefwoorden: polymerisatie ; radikaalreacties / emulsiepolymerisatie / reactiekinetiek / ketenoverdracht ; RAFT Subject headings: polymerization ; radical reactions / emulsion polymerisation / reaction kinetics / chain transfer ; RAFT © 2001, Hans de Brouwer Druk: Universiteitsdrukkerij, Technische Universiteit Eindhoven Omslagontwerp: Hans de Brouwer RAFT memorabilia living radical polymerization in homogeneous and heterogeneous media PROEFSCHRIFT ter verkrijging van de graad van doctor aan de Technische Universiteit Eindhoven, op gezag van de Rector Magnificus, prof.dr. M. Rem, voor een commissie aangewezen door het College voor Promoties in het openbaar te verdedigen op woensdag 30 mei 2001 om 16.00 uur door Johannes A. M. de Brouwer geboren te Goirle Dit proefschrift is goedgekeurd door de promotoren: prof.dr.ir. A. L. German en prof.dr. J. F. Schork Copromotor: dr. M. J. Monteiro Het werk in dit proefschrift is financieel ondersteund door de Stichting Emulsiepolymerisatie (SEP) / The work in this thesis was financially supported by the Foundation Emulsion polymerization (SEP) look around, wonder why we can live a life that's never satisfied lonely hearts, troubled minds looking for a way that we can never find many roads are ahead of us with choices to be made but life's just one of the games we play there is no special way from Winter in July on the album Unknown Territory by Bomb the Bass © 1991, Rhythm King Records table of contents Table of Contents Chapter 1. -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

Carbon-Carbon Bond Formation by Reductive Coupling with Titanium(II) Chloride Bis(Tetrahydrofuran)* John J
Carbon-Carbon Bond Formation by Reductive Coupling with Titanium(II) Chloride Bis(tetrahydrofuran)* John J. Eisch**, Xian Shi, Jacek Lasota Department of Chemistry, The State University of New York at Binghamton, Binghamton, New York 13902-6000, U.S.A. Dedicated to Professor Dr. Dr. h. c. mult. Günther Wilke on the occasion of his 70th birthday Z. Naturforsch. 50b, 342-350 (1995), received September 20, 1994 Carbon-Carbon Bond Formation, Reductive Coupling, Titanium(II) Chloride, Oxidative Addition, Carbonyl and Benzylic Halide Substrates Titanium(II) bis(tetrahydrofuran) 1, generated by the treatment of TiCl4 in THF with two equivalents of n-butyllithium at -78 °C, has been found to form carbon-carbon bonds with a variety of organic substrates by reductive coupling. Diphenylacetylene is dimerized to ex clusively (E,E)-1,2,3,4-tetraphenyl-l,3-butadiene; benzyl bromide and 9-bromofluorene give their coupled products, bibenzyl and 9,9'-bifluorenyl, as do benzal chloride and benzotrichlo- ride yield the l,2-dichloro-l,2-diphenylethanes and l,l,2,2-tetrachloro-l,2-diphenylethane, respectively. Styrene oxide and and ris-stilbene oxide undergo deoxygenation to styrene and fra/«-stilbene, while benzyl alcohol and benzopinacol are coupled to bibenzyl and to a mix ture of tetraphenylethylene and 1,1,2,2-tetraphenylethane. Both aliphatic and aromatic ke tones are smoothly reductively coupled to a mixture of pinacols and/or olefins in varying proportions. By a choice of experimental conditions either the pinacol or the olefin could be made the predominant product in certain cases. The reaction has been carried out with heptanal, cyclohexanone, benzonitrile, benzaldehyde, furfural, acetophenone, benzophenone and 9-fluorenone. -
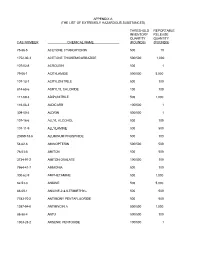
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

United States Patent O Fice Patented Dec
2,816,144 United States Patent O fice Patented Dec. 10, 1957 2. conventional manner and the hydrolysis mass is heated until completely free of any side chain chlorine deriva 2,816,144 tives. This can be determined by the use of alcoholic silver nitrate, for example, which will form a cloudy pre PROBEDUCTION OF RBENZALDEHYDE cipitate upon contact with compounds containing a side Robert W. Harris, Silsborough Township, Somerset chain chlorine group. County, N. S. The benzaldehyde which is obtained upon hydrolysis Separates from the aqueous layer and upon washing and No Drawing. Application August 4, 1955, fractionating yields a substantially pure compound which Seria No. 526,574. 0 is totally free from chlorine. 8 Claims. (C. 260-599) The chlorination of toluene is essentially a step-wise reaction and thus the chlorination can be terminated be fore more than two or three percent is converted to the The present invention relates to a process for the pro 5 tri-chloro derivative. This will correspond to a benzal duction of benzaldehyde from toluene. More particu chloride content of no more than about 35%, depending larly, it relates to a process wherein toluene is reacted upon the particular conditions of the reaction, the balance with chlorine and hydrolyzed to produce benzaldehyde being benzyl chloride with a minor amount of unreacted free from chlorine. toluene. Consequently, the reaction is terminated far The chlorination of toluene will result in the introduc 20 short of the maximum formation of benzal chloride so tion of one, two or three chlorine atoms in the methyl that substantially none of the toluene is lost through the side chain.