A Guide for Introducing and Strengthening Hepatitis B Birth Dose Vaccination
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Immunogenicity and Efficacy Testing in Chimpanzees of an Oral Hepatitis B Vaccine Based on Live Recombinant Adenovirus
Proc. Natl. Acad. Sci. USA Vol. 86, pp. 6763-6767, September 1989 Medical Sciences Immunogenicity and efficacy testing in chimpanzees of an oral hepatitis B vaccine based on live recombinant adenovirus (adenovirus animal model/adenovirus-vectored vacdnes) MICHAEL D. LUBECK*, ALAN R. DAVIS*, MURTY CHENGALVALA*, ROBERT J. NATUK*, JOHN E. MORIN*, KATHERINE MOLNAR-KIMBER*, BRUCE B. MASON*, BHEEM M. BHAT*, SATOSHI MIZUTANI*, PAUL P. HUNG*, AND ROBERT H. PURCELLO *Wyeth-Ayerst Research, Biotechnology and Microbiology Division, P.O. Pox 8299, Philadelphia, PA 19101; and tLaboratory of Infectious Diseases, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD 20892 Contributed by Robert H. Purcell, May 30, 1989 ABSTRACT As a major cause of acute and chronic liver which, because oftheir wide prior usage, are good candidates disease as well as hepatocellular carcinoma, hepatitis B virus as vectors. Other less well-characterized small animal models (HBV) continues to pose significant health problems world- for human adenoviruses have been occasionally reported wide. Recombinant hepatitis B vaccines based on adenovirus (11-13) but are likewise nonpermissive for Ad4 and Ad7 vectors have been developed to address global needs for infections (unpublished data). An early study of animal effective control of hepatitits B infection. Although consider- species including nonhuman primates indicated that human able progress has been made in the construction ofrecombinant adenoviruses do not induce acute respiratory disease in adenoviruses that express large amounts of HBV gene prod- monkeys or chimpanzees following intranasal inoculations ucts, preclinical immunogenicity and efficacy testing of candi- (14). Serological data, however, indicated that such experi- date vaccines has remained difficult due to the lack ofa suitable mental infections occasionally induced anti-adenovirus anti- animal model. -

Food Handler Exclusion Guidelines
Food Handler Exclusion Guidelines A guide for determining suitable exclusion periods for ill food handlers November 2017 Food handler exclusion guidelines Published by the State of Queensland (Queensland Health), November 2017 This document is licensed under a Creative Commons Attribution 3.0 Australia licence. To view a copy of this licence, visit creativecommons.org/licenses/by/3.0/au © State of Queensland (Queensland Health) 2017 You are free to copy, communicate and adapt the work, as long as you attribute the State of Queensland (Queensland Health). For more information contact: Food Safety Standards and Regulation, Department of Health, GPO Box 48, Brisbane QLD 4001, email [email protected], phone 07 3328 9310. An electronic version of this document is available at www.health.qld.gov.au. Disclaimer: The content presented in this publication is distributed by the Queensland Government as an information source only. The State of Queensland makes no statements, representations or warranties about the accuracy, completeness or reliability of any information contained in this publication. The State of Queensland disclaims all responsibility and all liability (including without limitation for liability in negligence) for all expenses, losses, damages and costs you might incur as a result of the information being inaccurate or incomplete in any way, and for any reason reliance was placed on such information. Food Handler Exclusion Guidelines - ii - Contents 1. Introduction ............................................................................................... -

PEDIARIX Is a Vaccine
HIGHLIGHTS OF PRESCRIBING INFORMATION • If Guillain-Barré syndrome occurs within 6 weeks of receipt of a prior These highlights do not include all the information needed to use vaccine containing tetanus toxoid, the decision to give PEDIARIX should PEDIARIX safely and effectively. See full prescribing information for be based on potential benefits and risks. (5.2) PEDIARIX. • The tip caps of the prefilled syringes contain natural rubber latex which may cause allergic reactions. (5.3) PEDIARIX [Diphtheria and Tetanus Toxoids and Acellular Pertussis • Syncope (fainting) can occur in association with administration of Adsorbed, Hepatitis B (Recombinant) and Inactivated Poliovirus injectable vaccines, including PEDIARIX. Procedures should be in place Vaccine], Suspension for Intramuscular Injection to avoid falling injury and to restore cerebral perfusion following Initial U.S. Approval: 2002 syncope. (5.4) • If temperature ≥105°F, collapse or shock-like state, or persistent, ----------------------------- INDICATIONS AND USAGE ---------------------------- PEDIARIX is a vaccine indicated for active immunization against diphtheria, inconsolable crying lasting ≥3 hours have occurred within 48 hours after tetanus, pertussis, infection caused by all known subtypes of hepatitis B virus, receipt of a pertussis-containing vaccine, or if seizures have occurred and poliomyelitis. PEDIARIX is approved for use as a 3-dose series in infants within 3 days after receipt of a pertussis-containing vaccine, the decision born of hepatitis B surface antigen (HBsAg)-negative mothers. PEDIARIX to give PEDIARIX should be based on potential benefits and risks. (5.5) may be given as early as 6 weeks of age through 6 years of age (prior to the • For children at higher risk for seizures, an antipyretic may be 7th birthday). -

Hepatitis B Fast Facts Everything You Need to Know in 2 Minutes Or Less!
Hepatitis B Foundation Cause for a Cure www.hepb.org Hepatitis B Fast Facts Everything you need to know in 2 minutes or less! Hepatitis B is the most common serious liver infection in the world. It is caused by the hepatitis B virus (HBV) that attacks liver cells and can lead to liver failure, cirrhosis (scarring) or cancer of the liver. The virus is transmitted through contact with blood and bodily fluids that contain blood. Most people are able to fight off the hepatitis B infection and clear the virus from their blood. This may take up to six months. While the virus is present in their blood, infected people can pass the virus on to others. Approximately 5-10% of adults, 30-50% of children, and 90% of babies will not get rid of the virus and will develop chronic infection. Chronically infected people can pass the virus on to others and are at increased risk for liver problems later in life. The hepatitis B virus is 100 times more infectious than the AIDS virus. Yet, hepatitis B can be pre- vented with a safe and effective vaccine. For the 400 million people worldwide who are chronically infected with hepatitis B, the vaccine is of no use. However, there are promising new treatments for those who live with chronic hepatitis B. In the World: • This year alone, 10 to 30 million people will become infected with the hepatitis B virus (HBV). • The World Health Organization estimates that 400 million people worldwide are already chronically infected with hepatitis B. -

Prevention & Control of Viral Hepatitis Infection
Prevention & Control of Viral Hepatitis Infection: A Strategy for Global Action © World Health Organization 2011. All rights reserved. The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement. The mention of specific companies or of certain manufacturers’ products does not imply that they are endorsed or recommended by the World Health Organization in preference to others of a similar nature that are not mentioned. Errors and omissions excepted, the names of proprietary products are distinguished by initial capital letters. All reasonable precautions have been taken by WHO to verify the information contained in this publication. However, the published material is being distributed without warranty of any kind, either express or implied. The responsibility for the interpretation and use of the material lies with the reader. In no event shall the World Health Organization be liable for damages arising from its use. Table of contents Disease burden 02 What is viral hepatitis? 05 Prevention & control: a tailored approach 06 Global Achievements 08 Remaining challenges 10 World Health Assembly: a mandate for comprehensive prevention & control 13 WHO goals and strategy -
Diphtheria, Tetanus, Pertussis, Polio, Hib and Hepatitis B Vaccine for Babies and Children
Diphtheria, Tetanus, Pertussis, Polio, Hib and Hepatitis B vaccine for babies and children This leaflet tells you about the DTaP/IPV/Hib/ HepB vaccine, also known as “6 in 1” as it protects against six diseases, diphtheria, tetanus, pertussis (whooping cough), polio, Haemophilus influenzae type b and hepatitis B (HepB) disease. What does the vaccine protect against? Diphtheria Diphtheria is a serious disease that usually begins with a sore throat and can quickly cause breathing problems. It can damage the heart and nervous system and, in severe cases, can kill. Before diphtheria vaccine was introduced in the UK, there were up to 70,000 cases of diphtheria and around 5,000 deaths a year. Diphtheria can be spread from person to person through close contact. Tetanus Tetanus is a disease affecting the nervous system which can cause muscle spasms and breathing problems and can kill. It is caused when germs found in soil and manure get into the body through wounds or burns. Tetanus cannot be passed from person to person. 2 Published June 2018 Pertussis (whooping cough) Whooping cough is a disease that can cause long bouts of coughing and choking, making it hard to breathe. It can last for up to 10 weeks and babies under one year of age are most at risk. The disease is very serious and can kill. Before the pertussis vaccine was introduced, the average number of cases reported each year in the UK was 120,000 and 92 children died in the year before the vaccine was introduced. Whooping cough is usually spread by coughs and sneezes. -

Hepatitis B? HEPATITIS B Hepatitis B Is a Contagious Liver Disease That Results from Infection with the Hepatitis B Virus
What is Hepatitis B? HEPATITIS B Hepatitis B is a contagious liver disease that results from infection with the Hepatitis B virus. When first infected, a person can develop Are you at risk? an “acute” infection, which can range in severity from a very mild illness with few or no symptoms to a serious condition requiring hospitalization. Acute Hepatitis B refers to the first 6 months after someone is exposed to the Hepatitis B virus. Some people are able to fight the infection and clear the virus. For others, the infection remains and leads to a “chronic,” or lifelong, illness. Chronic Hepatitis B refers to the illness that occurs when the Hepatitis B virus remains in a person’s body. Over time, the infection can cause serious health problems. How is Hepatitis B spread? Hepatitis B is usually spread when blood, semen, or other body fluids from a person infected with the Hepatitis B virus enter the body of someone who is not infected. This can happen through having sex with an infected partner; sharing needles, syringes, or other injection drug equipment; or from direct contact with the blood or open sores of an infected person. Hepatitis B can also be passed from an infected mother to her baby at birth. Who should be tested for Hepatitis B? Approximately 1.2 million people in the United States and 350 million people worldwide have Hepatitis B. Testing for Hepatitis B is recommended for certain groups of people, including: Most are unaware of their infection. ■ People born in Asia, Africa, and other regions with moderate or high rates Is Hepatitis B common? of Hepatitis B (see map) Yes. -
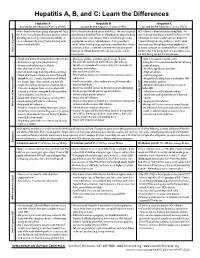
Hepatitis A, B, and C: Learn the Differences
Hepatitis A, B, and C: Learn the Differences Hepatitis A Hepatitis B Hepatitis C caused by the hepatitis A virus (HAV) caused by the hepatitis B virus (HBV) caused by the hepatitis C virus (HCV) HAV is found in the feces (poop) of people with hepa- HBV is found in blood and certain body fluids. The virus is spread HCV is found in blood and certain body fluids. The titis A and is usually spread by close personal contact when blood or body fluid from an infected person enters the body virus is spread when blood or body fluid from an HCV- (including sex or living in the same household). It of a person who is not immune. HBV is spread through having infected person enters another person’s body. HCV can also be spread by eating food or drinking water unprotected sex with an infected person, sharing needles or is spread through sharing needles or “works” when contaminated with HAV. “works” when shooting drugs, exposure to needlesticks or sharps shooting drugs, through exposure to needlesticks on the job, or from an infected mother to her baby during birth. or sharps on the job, or sometimes from an infected How is it spread? Exposure to infected blood in ANY situation can be a risk for mother to her baby during birth. It is possible to trans- transmission. mit HCV during sex, but it is not common. • People who wish to be protected from HAV infection • All infants, children, and teens ages 0 through 18 years There is no vaccine to prevent HCV. -

Hepatitis B Vaccine – Frequently Asked Questions (Information from the CDC)
AAMC Standardized Immunization Form 2020 Hepatitis B Vaccine – Frequently Asked Questions (Information from the CDC) 1. What are the hepatitis B vaccines licensed for use in the United States? Three single-antigen vaccines and two combination vaccines are currently licensed in the United States. Single-antigen hepatitis B vaccines: • ENGERIX-B® • RECOMBIVAX HB® • HEPLISAV-B™ Combination vaccines: • PEDIARIX®: Combined hepatitis B, diphtheria, tetanus, acellular pertussis (DTaP), and inactivated poliovirus (IPV) vaccine. Cannot be administered before age 6 weeks or after age 7 years. • TWINRIX®: Combined Hepatitis A and hepatitis B vaccine. Recommended for people aged ≥18 years who are at increased risk for both HAV and HBV infections. 2. What are the recommended schedules for hepatitis B vaccination? The vaccination schedule most often used for children and adults is three doses given at 0, 1, and 6 months. Alternate schedules have been approved for certain vaccines and/or populations. A new formulation, Heplisav-B (HepB-CpG), is approved to be given as two doses one month apart. 3. If there is an interruption between doses of hepatitis B vaccine, does the vaccine series need to be restarted? No. The series does not need to be restarted but the following should be considered: • If the vaccine series was interrupted after the first dose, the second dose should be administered as soon as possible. • The second and third doses should be separated by an interval of at least 8 weeks. • If only the third dose is delayed, it should be administered as soon as possible. 4. Is it harmful to administer an extra dose of hepatitis B vaccine or to repeat the entire vaccine series if documentation of the vaccination history is unavailable or the serology test is negative? No, administering extra doses of single-antigen hepatitis B vaccine is not harmful. -

Package Inserts
Individuals using assistive technology may not be able to fully access the information contained in this file. For assistance, please send an e-mail to: [email protected] and include 508 Accommodation and the title of the document in the subject line of your e-mail. HIGHLIGHTS OF PRESCRIBING INFORMATION -----------------------WARNINGS AND PRECAUTIONS------------------------ These highlights do not include all the information needed to use • Carefully consider benefits and risks before administering VAXELIS to VAXELIS safely and effectively. See full prescribing information for persons with a history of: VAXELIS. - fever ≥40.5°C (≥105°F), hypotonic-hyporesponsive episode (HHE) or VAXELISTM (Diphtheria and Tetanus Toxoids and Acellular Pertussis, persistent, inconsolable crying lasting ≥3 hours within 48 hours after a Inactivated Poliovirus, Haemophilus b Conjugate and Hepatitis B previous pertussis-containing vaccine. (5.2) Vaccine) - seizures within 3 days after a previous pertussis-containing vaccine. (5.2) Suspension for Intramuscular Injection Initial U.S. Approval: 2018 • If Guillain-Barré syndrome occurred within 6 weeks of receipt of a prior vaccine containing tetanus toxoid, the risk for Guillain-Barré syndrome ----------------------------RECENT MAJOR CHANGES ------------------------ may be increased following VAXELIS. (5.3) Dosage and Administration (2.2) 09/2020 • Apnea following intramuscular vaccination has been observed in some ----------------------------INDICATIONS AND USAGE--------------------------- infants born prematurely. The decision about when to administer an VAXELIS is a vaccine indicated for active immunization to prevent intramuscular vaccine, including VAXELIS, to an infant born prematurely diphtheria, tetanus, pertussis, poliomyelitis, hepatitis B, and invasive disease should be based on consideration of the individual infant’s medical status due to Haemophilus influenzae type b. -

Understanding Human Astrovirus from Pathogenesis to Treatment
University of Tennessee Health Science Center UTHSC Digital Commons Theses and Dissertations (ETD) College of Graduate Health Sciences 6-2020 Understanding Human Astrovirus from Pathogenesis to Treatment Virginia Hargest University of Tennessee Health Science Center Follow this and additional works at: https://dc.uthsc.edu/dissertations Part of the Diseases Commons, Medical Sciences Commons, and the Viruses Commons Recommended Citation Hargest, Virginia (0000-0003-3883-1232), "Understanding Human Astrovirus from Pathogenesis to Treatment" (2020). Theses and Dissertations (ETD). Paper 523. http://dx.doi.org/10.21007/ etd.cghs.2020.0507. This Dissertation is brought to you for free and open access by the College of Graduate Health Sciences at UTHSC Digital Commons. It has been accepted for inclusion in Theses and Dissertations (ETD) by an authorized administrator of UTHSC Digital Commons. For more information, please contact [email protected]. Understanding Human Astrovirus from Pathogenesis to Treatment Abstract While human astroviruses (HAstV) were discovered nearly 45 years ago, these small positive-sense RNA viruses remain critically understudied. These studies provide fundamental new research on astrovirus pathogenesis and disruption of the gut epithelium by induction of epithelial-mesenchymal transition (EMT) following astrovirus infection. Here we characterize HAstV-induced EMT as an upregulation of SNAI1 and VIM with a down regulation of CDH1 and OCLN, loss of cell-cell junctions most notably at 18 hours post-infection (hpi), and loss of cellular polarity by 24 hpi. While active transforming growth factor- (TGF-) increases during HAstV infection, inhibition of TGF- signaling does not hinder EMT induction. However, HAstV-induced EMT does require active viral replication. -

Recommended Adult Immunization Schedule
Recommended Adult Immunization Schedule UNITED STATES for ages 19 years or older 2021 Recommended by the Advisory Committee on Immunization Practices How to use the adult immunization schedule (www.cdc.gov/vaccines/acip) and approved by the Centers for Disease Determine recommended Assess need for additional Review vaccine types, Control and Prevention (www.cdc.gov), American College of Physicians 1 vaccinations by age 2 recommended vaccinations 3 frequencies, and intervals (www.acponline.org), American Academy of Family Physicians (www.aafp. (Table 1) by medical condition and and considerations for org), American College of Obstetricians and Gynecologists (www.acog.org), other indications (Table 2) special situations (Notes) American College of Nurse-Midwives (www.midwife.org), and American Academy of Physician Assistants (www.aapa.org). Vaccines in the Adult Immunization Schedule* Report y Vaccines Abbreviations Trade names Suspected cases of reportable vaccine-preventable diseases or outbreaks to the local or state health department Haemophilus influenzae type b vaccine Hib ActHIB® y Clinically significant postvaccination reactions to the Vaccine Adverse Event Hiberix® Reporting System at www.vaers.hhs.gov or 800-822-7967 PedvaxHIB® Hepatitis A vaccine HepA Havrix® Injury claims Vaqta® All vaccines included in the adult immunization schedule except pneumococcal 23-valent polysaccharide (PPSV23) and zoster (RZV) vaccines are covered by the Hepatitis A and hepatitis B vaccine HepA-HepB Twinrix® Vaccine Injury Compensation Program. Information on how to file a vaccine injury Hepatitis B vaccine HepB Engerix-B® claim is available at www.hrsa.gov/vaccinecompensation. Recombivax HB® Heplisav-B® Questions or comments Contact www.cdc.gov/cdc-info or 800-CDC-INFO (800-232-4636), in English or Human papillomavirus vaccine HPV Gardasil 9® Spanish, 8 a.m.–8 p.m.