Chemical Hazards in the Artisanal Gold Sector
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Acute Toxicity of Cyanide in Aerobic Respiration: Theoretical and Experimental Support for Murburn Explanation
BioMol Concepts 2020; 11: 32–56 Research Article Open Access Kelath Murali Manoj*, Surjith Ramasamy, Abhinav Parashar, Daniel Andrew Gideon, Vidhu Soman, Vivian David Jacob, Kannan Pakshirajan Acute toxicity of cyanide in aerobic respiration: Theoretical and experimental support for murburn explanation https://doi.org/10.1515/bmc-2020-0004 received January 14, 2020; accepted February 19, 2020. of diffusible radicals and proton-equilibriums, explaining analogous outcomes in mOxPhos chemistry. Further, we Abstract: The inefficiency of cyanide/HCN (CN) binding demonstrate that superoxide (diffusible reactive oxygen with heme proteins (under physiological regimes) is species, DROS) enables in vitro ATP synthesis from demonstrated with an assessment of thermodynamics, ADP+phosphate, and show that this reaction is inhibited kinetics, and inhibition constants. The acute onset of by CN. Therefore, practically instantaneous CN ion-radical toxicity and CN’s mg/Kg LD50 (μM lethal concentration) interactions with DROS in matrix catalytically disrupt suggests that the classical hemeFe binding-based mOxPhos, explaining the acute lethal effect of CN. inhibition rationale is untenable to account for the toxicity of CN. In vitro mechanistic probing of Keywords: cyanide-poisoning; murburn concept; CN-mediated inhibition of hemeFe reductionist systems aerobic respiration; ATP-synthesis; hemoglobin; was explored as a murburn model for mitochondrial cytochrome oxidase; mitochondria; diffusible reactive oxidative phosphorylation (mOxPhos). The effect of CN oxygen species (DROS). in haloperoxidase catalyzed chlorine moiety transfer to small organics was considered as an analogous probe for phosphate group transfer in mOxPhos. Similarly, Introduction inclusion of CN in peroxidase-catalase mediated one- electron oxidation of small organics was used to explore Since the discovery of the dye Prussian blue (ferric electron transfer outcomes in mOxPhos, leading to water ferrocyanide, Fe7[CN]18) and the volatile prussic acid formation. -

Hazardous Chemicals in Secondhand Marijuana Smoke
Hazardous Chemicals in Secondhand Marijuana Smoke “The following 33 marijuana smoke constituents included in Table 1 are listed under 33 Chemicals Proposition 65 as causing cancer: acetaldehyde, acetamide, acrylonitrile, 4- aminobiphenyl, arsenic, benz[a]anthracene, benzene, benzo[a]pyrene, That Can benzo[b]fluoranthene, benzo[j]fluoranthene, benzo[k]fluoranthene, benzofuran, 1,3- butadiene, cadmium, carbazole, catechol, chromium (hexavalent compounds), Cancer chrysene, dibenz[a,h]anthracene, dibenz[a,i]pyrene, dibenzo[a,e]pyrene, “Many of the chemical diethylnitrosamine, dimethylnitrosamine, formaldehyde, indeno[1,2,3,-c,d]pyrene, constituents that have been isoprene, lead, mercury, 5-methylchrysene, naphthalene, nickel, pyridine, and identified in marijuana smoke quinoline.” are carcinogens.” 2009 OEHHA document, Evidence on the Carcinogenicity of Marijuana Smoke Hydrogen Cyanide interferes with the normal use of oxygen by nearly every organ of Hydrogen the body. Exposure to hydrogen cyanide (AC) can be rapidly fatal. It has whole-body (systemic) effects, particularly affecting those organ systems most sensitive to low Cyanide oxygen levels: the central nervous system (brain), the cardiovascular system (heart Is the same chemical used for and blood vessels), and the pulmonary system (lungs). Hydrogen cyanide (AC) is a chemical weapons. chemical warfare agent (military designation, AC). Ammonia gas is a severe respiratory tract irritant. Can cause severe irritation of the Ammonia nose and throat. Can cause life-threatening accumulation of fluid in the lungs Household cleaner used on (pulmonary edema). Symptoms may include coughing, shortness of breath, difficult floors and toilets. There is 3 breathing and tightness in the chest. Symptoms may develop hours after exposure times more in secondhand and are made worse by physical effort. -
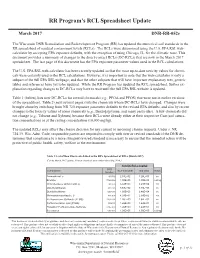
RR Program's RCL Spreadsheet Update
RR Program’s RCL Spreadsheet Update March 2017 RR Program RCL Spreadsheet Update DNR-RR-052e The Wisconsin DNR Remediation and Redevelopment Program (RR) has updated the numerical soil standards in the August 2015 DNR-RR- 052b RR spreadsheet of residual contaminant levels (RCLs). The RCLs were determined using the U.S. EPA RSL web- calculator by accepting EPA exposure defaults, with the exception of using Chicago, IL, for the climatic zone. This documentThe U.S. provides EPA updateda summary its Regionalof changes Screening to the direct-contact Level (RSL) RCLs website (DC-RCLs) in June that2015. are To now reflect in the that March 2017 spreadsheet.update, the The Wisconsin last page ofDNR this updated document the has numerical the EPA exposuresoil standards, parameter or residual values usedcontaminant in the RCL levels calculations. (RCLs), in the Remediation and Redevelopment program’s spreadsheet of RCLs. This document The providesU.S. EPA a RSL summary web-calculator of the updates has been incorporated recently updated in the Julyso that 2015 the spreadsheet.most up-to-date There toxicity were values no changes for chemi - cals madewere certainlyto the groundwater used in the RCLs,RCL calculations. but there are However, many changes it is important in the industrial to note that and the non-industrial web-calculator direct is only a subpartcontact of the (DC) full RCLsEPA RSL worksheets. webpage, Tables and that 1 andthe other 2 of thissubparts document that will summarize have important the DC-RCL explanatory changes text, generic tablesfrom and the references previous have spreadsheet yet to be (Januaryupdated. -

Evaluation of Scale-Up Model for Flotation with Kristineberg Ore
Evaluation of Scale-up Model for Flotation with Kristineberg Ore Adam Isaksson Chemical Engineering, master's level 2018 Luleå University of Technology Department of Civil, Environmental and Natural Resources Engineering Evaluation of Scale-up Model for Flotation with Kristineberg Ore Adam Isaksson 2018 For degree of MASTER OF SCIENCE Luleå University of Technology Department of Civil, Environmental and Natural Resources Engineering Division of Minerals and Metallurgical Engineering Printed by Luleå University of Technology, Graphic Production 2018 Luleå 2018 www.ltu.se Preface As you may have figured out by now, this thesis is all about mineral processing and the extraction of metals. It was written as part of my studies at Luleå University of Technology, for a master’s degree in Chemical Engineering with specialisation Mineral and Metal Winning. There are many people I would like to thank for helping me out during all these years. First of all, my thanks go to supervisors Bertil Pålsson and Lisa Malm for the guidance in this project. Iris Wunderlich had a paramount role during sampling and has kindly delivered me data to this report, which would not have been finished without her support. I would also like to thank Boliden Mineral AB as a company. Partly for giving me the chance to write this thesis in the first place, but also for supporting us students during our years at LTU. Speaking of which, thanks to Olle Bertilsson for reading the report and giving me feedback. The people at the TMP laboratory deserves another mention. I am also very grateful for the financial support and generous scholarships from Jernkontoret these five years. -

Qualitative Chemical Measures to Indirectly Assess Quality of Natural/Uncured, Organic Bacon Compared to Traditionally Cured Bacon
Iowa State University Capstones, Theses and Graduate Theses and Dissertations Dissertations 2009 Qualitative chemical measures to indirectly assess quality of natural/uncured, organic bacon compared to traditionally cured bacon. Charlwit Kulchaiyawat Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/etd Part of the Animal Sciences Commons Recommended Citation Kulchaiyawat, Charlwit, "Qualitative chemical measures to indirectly assess quality of natural/uncured, organic bacon compared to traditionally cured bacon." (2009). Graduate Theses and Dissertations. 10999. https://lib.dr.iastate.edu/etd/10999 This Thesis is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Graduate Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. Qualitative chemical measures to indirectly assess quality of natural/uncured, organic bacon compared to traditionally cured bacon by Charlwit Kulchaiyawat A thesis submitted to the graduate faculty in partial fulfillment of the requirements for the degree of MASTER OF SCIENCE Major: Meat Science Program of Study Committee: Joseph Sebranek, Major Professor James Dickson Mark Honeyman Lester Wilson Iowa State University Ames, Iowa 2009 Copyright © Charlwit Kulchaiyawat 2009. All rights reserved. ii TABLE OF CONTENTS LIST OF TABLES iii ABSTRACT -

Nitric Oxide.Vp
CHAPTER 5 Maple Leaf Foods, Toronto, Canada Brown Foundation Institute of Molecular Medicine, The University of Texas Health Science Center, Houston, TX 77030 ESPITE the many published reports on the beneficial properties of Dnitrite and nitrate in physiology, nitrite and nitrate in cured and pro- cessed meats continues to be perceived as harmful. The previous chapter revealed that certain foods, particularly green leafy vegetables are natu- rally enriched in nitrite and nitrate from growing in soil. However, en- riching meats with nitrite or nitrate during curing is perceived as harmful and advocated by some groups to be eliminated completely. The use of pure sodium nitrate in curing is now only a minor practice in the United States. The ingoing levels of sodium nitrite have been tightly controlled by the Food Safety and Inspection Service (FSIS) of the U.S. Depart- ment of Agriculture. To satisfy consumer demands for no added nitrite processed meats, efforts have recently been taken to creatively adjust the meat curing process by employing “nitrite free” organic vegetable pow- ders instead of directly adding sodium nitrite salts. Although the end re- sult is production of nitrite from the nitrate contained in the vegetable powders, a more “natural” or “organic” approach seems to appeal to con- sumers. This is primarily due to the public perception of nitrite and ni- trate. Reports about methemoglobinemia in infants (blue baby syndrome) caused by drinks or food prepared with nitrate-rich (and bac- terially contaminated) well water and vegetables, intentional and occu- pational intoxications in adults, increasing nitrate levels in soil and lakes as a result of fertilizer overuse, and the formation of potentially carcino- genic N-nitrosamines all contribute to the negative image that nitrite and nitrate have held in recent years. -

Mercury and Mercury Compounds
United States Office of Air Quality EPA-454/R-97-012 Environmental Protection Planning And Standards Agency Research Triangle Park, NC 27711 December 1997 AIR EPA LOCATING AND ESTIMATING AIR EMISSIONS FROM SOURCES OF MERCURY AND MERCURY COMPOUNDS L & E EPA-454/R-97-012 Locating And Estimating Air Emissions From Sources of Mercury and Mercury Compounds Office of Air Quality Planning and Standards Office of Air and Radiation U.S. Environmental Protection Agency Research Triangle Park, NC 27711 December 1997 This report has been reviewed by the Office of Air Quality Planning and Standards, U.S. Environmental Protection Agency, and has been approved for publication. Mention of trade names and commercial products does not constitute endorsement or recommendation for use. EPA-454/R-97-012 TABLE OF CONTENTS Section Page EXECUTIVE SUMMARY ................................................ xi 1.0 PURPOSE OF DOCUMENT .............................................. 1-1 2.0 OVERVIEW OF DOCUMENT CONTENTS ................................. 2-1 3.0 BACKGROUND ........................................................ 3-1 3.1 NATURE OF THE POLLUTANT ..................................... 3-1 3.2 OVERVIEW OF PRODUCTION, USE, AND EMISSIONS ................. 3-1 3.2.1 Production .................................................. 3-1 3.2.2 End-Use .................................................... 3-3 3.2.3 Emissions ................................................... 3-6 4.0 EMISSIONS FROM MERCURY PRODUCTION ............................. 4-1 4.1 PRIMARY MERCURY -

Corrosion and Mercury Release from Dental Amalgam
Corrosion and Mercury Release from Dental Amalgam 1 Jaro Pleva, Ph.D. Abstract been presented, amalgam is still by far the most Corrosion attacks on twenty-two dental extensively used material for dental restorations.2 amalgam restorations after in vivo service have Since the investigations and warnings of the been studied by Scanning Electron Microscopy outstanding German chemist Alfred Stock, 1920- together with the Energy Dispersive X-Ray 19453 4 5 little epidemiological studies of mercury Technique, and by optical microscopy. From the release and its effects have been made. One of the measured depth and type of corrosion attack, possible reasons may be the very interdisciplinary estimates of released mercury amounts are made. character of the problem. For correct answer, The amalgam fillings have been obtained from specialist competence in the following fields is members of a group of 250 individuals, who required: materials science, suspected their health troubles potentially to be corrosion/electrochemistry, toxicology, chronic mercury poisoning from amalgam and medicine/diagnostics, physical biology, analytical were to have all amalgam fillings removed. Three chemistry. typical patient cases are presented. The common type of dental amalgam is an Model calculations of released mercury, based alloy containing typically in weight-%; 50 Hg, 35 on previously published measurements of Ag, 10 Sn, Cu, Zn. corrosion currents with and without abrasion are Reported types of amalgam degradation are also given. crevice corrosion,2 6 7 8 selective corrosion,9 The investigations show, that the long-term galvanic corrosion in contact with dissimilar release of mercury from a few amalgam fillings alloys10 11 and mechanical wear.12 33 Besides will often reach or exceed the recommended selective attack, stress corrosion has been limits for daily intake of mercury. -

NATURE MAY 6, 1944, Vol
562 NATURE MAY 6, 1944, VoL. 153 If the number of young people leaving school and nineteenth century led to the emancipation of the college is much larger in recent years than at the lower classes and of women, and to increasing de beginning of the century, it would seem that this is mands for improved social status and better educa less marked in Switzerland than in certain other tion. Dr. Erb considers that, in Switzerland, the countries. For example, it is pointed out that, in exaggerated importance imputed to academic learn 1930-31, per 100,000 of population, Germany had 63 ing of the more showy or superficial type was par Abiturienten (holders of school-leaving certificates or ticularly marked, and was closely associated with equivalent) whereas Switzerland had only 34. Japan the growing soci»l prejudice for black-coated respect and Rumania in 1934 had more than six times as ability. Those who had not themselves attained many with academic training as in 1913. In the to this status were anxious and determined that their same period those of Holland increased by 146 per children should do so; and this delusion, says Dr. cent, of France 112 per cent, of Great Britain 83 per Erb, will continue until it is more generally realized cent, and of Switzerland 59 per cent. According to that the harder one studies the more certainly will Dr. Erb, the most disturbing aspect of this increase he miss the road to wealth. This may be often true, in quantity is the serious decline in quality : he but scarcely attains the dignity of a general thinks the standard, however measured, is definitely proposition ; and in any event strikes a slightly dis lower. -

Silver City Santa Rita Hurley
SCENIC TRIPS to the GEOLOGIC PAST SILVER CITY SANTA RITA HURLEY NEW MEXICO Scenic Trips to the Geologic Past Series: No. I - Santa Fe, New Mexico No. 2 - Taos-Red River - Eagle Nest, New Mexico, Circle Drive No. 3 - Roswell- Capitan - Ruidoso and Bottomless Lakes Park, New Mexico No. 4 - Southern Zuni Mountains, New Mexico No. 5 - Silver City-Santa Rita-Hurley, New Mexico Additional copies of these guidebooks are available, for 25 cents, from the New Mexico Bureau of Mines and Mineral Resources, Campus Station, Socorro, New Mexico. HO: FOR THE GOLD AND SILVER MINES OF NEW MEXICO Fortune hunters, capitalists, poor men, Sickly folks, all whose hearts are bowed down; And Ye who would live long, be rich, healthy, and Happy; Come to our sunny clime and see For Yourselves. Handbill -- 1883 Santa Rita Open Pit, 1959. Scenic Trips to the Geologic Past No. 5 SILVER CITY - SANTA RITA - HURLEY, NEW MEXICO by JOHN H. SCHILLING 1959 STATE BUREAU OF MINES AND MINERAL RESOURCES a division of NEW MEXICO INSTITUTE OF MINING AND TECHNOLOGY Socorro - New Mexico NEW MEXICO INSTITUTE OF MINING AND TECHNOLOGY E. J. Workman, President STATE BUREAU OF MINES AND MINERAL RESOURCES Alvin J. Thompson, Director THE REGENTS Members Ex Officio The Honorable John Burroughs ...............Governor of New Mexico Tom Wiley ……….…… ...........Superintendent of Public Instruction Appointed Members Holm 0. Bursum, Jr. ........................................................ Socorro Thomas M. Cramer.......................................................... Carlsbad Frank C. DiLuzio ...................................................... Albuquerque John N. Mathews, Jr. ....................................................... Socorro Richard A. Matuszeski .............................................. Albuquerque PREFACE Much of the work undertaken at the New Mexico Bureau of Mines and Mineral Resources is done to help the mineral industries -- the prospector, miner, geologist, oil man. -

Chemistry As a Tool for Historical Research: Identifying Paths of Historical Mercury Pollution in the Hispanic New World
Bull. Hist. Chem., VOLUME 37, Number 2 (2012) 61 CHEMISTRY AS A TOOL FOR HISTORICAL RESEARCH: IDENTIFYING PATHS OF HISTORICAL MERCURY POLLUTION IN THE HISPANIC NEW WORLD Saúl Guerrero, History Department, McGill University, Montreal, QC H3A 2T7, Canada, [email protected] Introduction silver ores to identify and quantify the different mercury loss vectors that resulted from the amalgamation process This article is the first of a series that explore the as practiced in the Hispanic New World. potential of chemistry as an efficient tool for historical research. Basic chemical principles such as the The Scale of Anthropogenic Emissions of stoichiometry of chemical reactions provide the historian Mercury in the New World with a powerful tool to judge the reliability of archival records and interpret better the historiography of events From 1521 to 1810 Spain produced nearly 69% of that relate directly to processes of production based the total world output of silver from its mines in New on chemical reactions. Chemical mass balances have Spain (present day Mexico) and in the Vice-Royalty of determined both revenue streams and environmental Peru (present day Peru and Bolivia). During this period consequences in the past. there was no other non-Hispanic major silver produc- A very appropriate case study to apply this ap- tion in the New World (2). The global economic impact proach is the first industrial scale chemical process to of these exports of silver to Europe and China during have caused a global economic impact. The application the Early Modern Era has received wide coverage in of mercury amalgamation to extract silver from the ores the historiography of this period (3). -

The Use of Sodium Cyanide in Wildlife Damage Management
Human Health and Ecological Risk Assessment for the Use of Wildlife Damage Management Methods by USDA-APHIS-Wildlife Services Chapter VII THE USE OF SODIUM CYANIDE IN WILDLIFE DAMAGE MANAGEMENT May 2017 Peer Reviewed Final October 2019 THE USE OF SODIUM CYANIDE IN WILDLIFE DAMAGE MANAGEMENT EXECUTIVE SUMMARY USDA-APHIS Wildlife Services (WS) uses sodium cyanide (NaCN) to manage coyotes, red foxes, gray foxes, arctic foxes, and wild dogs that prey upon livestock, poultry, and federally designated threatened and endangered species or animals that are vectors of disease. This human health and ecological risk assessment is an evaluation of the risks to human health, nontarget animals, and the environment from NaCN use by WS. WS uses the M-44, the name for the ejector device that delivers a single dose NaCN from a capsule, to target canids. The M-44 is spring-activated and is actuated when an animal pulls up on the capsule holder; a plunger propelled by the spring breaks through a capsule with dry NaCN to deliver the contents into the mouth of an animal. The WS applicator baits the M-44 capsule holder sides to attract target canids. Sodium cyanide reacts rapidly with moisture in the mouth or mucus membranes of the nose and eyes to form hydrogen cyanide (HCN), a toxicant. One NaCN capsule contains enough cyanide to be lethal to animals through oral contact, inhalation contact, and moist dermal pathway contact. WS annually averaged the known take of 13,959 target canids and 362 nontarget species with NaCN between FY11 and FY15, recording 1,548,000 Method Nights with M-44s in 17 States.