Comprehensive Substrate Specificity Profiling of the Human Nek
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Targeted Kinase Inhibitor Profiling Using a Hybrid Quadrupole-Orbitrap Mass Spectrometer
Targeted Kinase Inhibitor Profiling Note Application Using a Hybrid Quadrupole-Orbitrap Mass Spectrometer 1 2 3 Scott Peterman , Ryan D. Bomgarden , Rosa Viner 574 1Thermo Fisher Scientific, Cambridge, MA;2 Thermo Fisher Scientific, Rockford, IL; 3Thermo Fisher Scientific, San Jose, CA Key Words An emerging technology for identifying kinase inhibitor Q Exactive, targeted peptide quantification, msxSIM, kinome profiling by targets is based on chemical proteomic profiling of kinase MS, desthiobiotin nucleotide probes inhibitor specificity and binding affinity. This technology combines mass spectrometry (MS)-based identification Goal and quantitation with small molecule probe binding and To identify and quantify kinase inhibition by staurosporine using kinase enrichment to determine kinase active site occupancy active sites probes in combination with targeted, multiplexed SIM (msxSIM). during inhibition. One of these methods uses novel biotinylated ATP and/or ADP probes that irreversibly react with conserved lysine residues of kinase ATP binding Introduction sites.1,2 Selective enrichment of active-site peptides from Protein kinases are key enzymes involved in a wide array labeled kinase digests dramatically reduces background of complex cellular functions and pathways. Misregulation matrix and increases signal for MS analysis of low- or mutation of protein kinases underlies numerous disease abundance kinase peptides. Using this method, more than states, including tumorigenesis, making them ideal candidates 400 different protein and lipid kinases from various for drug development. However, identifying specific mammalian tissues and cell lines have been identified and kinase inhibitors is challenging due to the high degree of functionally assayed using targeted acquisition on an ion homology among subfamily members of the 500+ human trap mass spectrometer.1 The assays are available kinases. -

AGC Kinases in Mtor Signaling, in Mike Hall and Fuyuhiko Tamanoi: the Enzymes, Vol
Provided for non-commercial research and educational use only. Not for reproduction, distribution or commercial use. This chapter was originally published in the book, The Enzymes, Vol .27, published by Elsevier, and the attached copy is provided by Elsevier for the author's benefit and for the benefit of the author's institution, for non-commercial research and educational use including without limitation use in instruction at your institution, sending it to specific colleagues who know you, and providing a copy to your institution’s administrator. All other uses, reproduction and distribution, including without limitation commercial reprints, selling or licensing copies or access, or posting on open internet sites, your personal or institution’s website or repository, are prohibited. For exceptions, permission may be sought for such use through Elsevier's permissions site at: http://www.elsevier.com/locate/permissionusematerial From: ESTELA JACINTO, AGC Kinases in mTOR Signaling, In Mike Hall and Fuyuhiko Tamanoi: The Enzymes, Vol. 27, Burlington: Academic Press, 2010, pp.101-128. ISBN: 978-0-12-381539-2, © Copyright 2010 Elsevier Inc, Academic Press. Author's personal copy 7 AGC Kinases in mTOR Signaling ESTELA JACINTO Department of Physiology and Biophysics UMDNJ-Robert Wood Johnson Medical School, Piscataway New Jersey, USA I. Abstract The mammalian target of rapamycin (mTOR), a protein kinase with homology to lipid kinases, orchestrates cellular responses to growth and stress signals. Various extracellular and intracellular inputs to mTOR are known. mTOR processes these inputs as part of two mTOR protein com- plexes, mTORC1 or mTORC2. Surprisingly, despite the many cellular functions that are linked to mTOR, there are very few direct mTOR substrates identified to date. -

A Calcium- and Calmodulin-Dependent Kinase I␣/ Microtubule Affinity Regulating Kinase 2 Signaling Cascade Mediates Calcium-Dependent Neurite Outgrowth
The Journal of Neuroscience, April 18, 2007 • 27(16):4413–4423 • 4413 Cellular/Molecular A Calcium- and Calmodulin-Dependent Kinase I␣/ Microtubule Affinity Regulating Kinase 2 Signaling Cascade Mediates Calcium-Dependent Neurite Outgrowth Nataliya V. Uboha,1 Marc Flajolet,2 Angus C. Nairn,1,2 and Marina R. Picciotto1 1Department of Psychiatry, Yale University School of Medicine, New Haven, Connecticut 06508, and 2Laboratory of Molecular and Cellular Neuroscience, The Rockefeller University, New York, New York 10021 Calcium is a critical regulator of neuronal differentiation and neurite outgrowth during development, as well as synaptic plasticity in adulthood. Calcium- and calmodulin-dependent kinase I (CaMKI) can regulate neurite outgrowth; however, the signal transduction cascades that lead to its physiological effects have not yet been elucidated. CaMKI␣ was therefore used as bait in a yeast two-hybrid assay and microtubule affinity regulating kinase 2 (MARK2)/Par-1b was identified as an interacting partner of CaMKI in three independent screens. The interaction between CaMKI and MARK2 was confirmed in vitro and in vivo by coimmunoprecipitation. CaMKI binds MARK2 within its kinase domain, but only if it is activated by calcium and calmodulin. Expression of CaMKI and MARK2 in Neuro-2A (N2a) cells and in primary hippocampal neurons promotes neurite outgrowth, an effect dependent on the catalytic activities of these enzymes. In addition, decreasing MARK2 activity blocks the ability of the calcium ionophore ionomycin to promote neurite outgrowth. Finally, CaMKI phosphorylates MARK2 on novel sites within its kinase domain. Mutation of these phosphorylation sites decreases both MARK2 kinase activity and its ability to promote neurite outgrowth. -

Kinome Profiling of Clinical Cancer Specimens
Published OnlineFirst March 23, 2010; DOI: 10.1158/0008-5472.CAN-09-3989 Review Cancer Research Kinome Profiling of Clinical Cancer Specimens Kaushal Parikh and Maikel P. Peppelenbosch Abstract Over the past years novel technologies have emerged to enable the determination of the transcriptome and proteome of clinical samples. These data sets will prove to be of significant value to our elucidation of the mechanisms that govern pathophysiology and may provide biological markers for future guidance in person- alized medicine. However, an equally important goal is to define those proteins that participate in signaling pathways during the disease manifestation itself or those pathways that are made active during successful clinical treatment of the disease: the main challenge now is the generation of large-scale data sets that will allow us to define kinome profiles with predictive properties on the outcome-of-disease and to obtain insight into tissue-specific analysis of kinase activity. This review describes the current techniques available to gen- erate kinome profiles of clinical tissue samples and discusses the future strategies necessary to achieve new insights into disease mechanisms and treatment targets. Cancer Res; 70(7); 2575–8. ©2010 AACR. Background the current state of the cell or tissue as characterized by pro- teomic and metabolic measurements (3). The past five years have seen an exponential increase in In this review we describe and evaluate the various tech- technological development to explore the genome and the niques that have recently become available to study the ki- proteome to understand the molecular basis of disease. How- nomes of clinical tissue samples. -
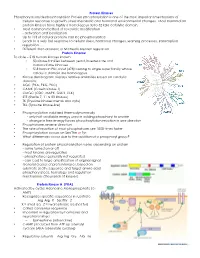
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

Identification of Protein O-Glcnacylation Sites Using Electron Transfer Dissociation Mass Spectrometry on Native Peptides
Identification of protein O-GlcNAcylation sites using electron transfer dissociation mass spectrometry on native peptides Robert J. Chalkleya, Agnes Thalhammerb, Ralf Schoepfera,b, and A. L. Burlingamea,1 aDepartment of Pharmaceutical Chemistry, University of California, 600 16th Street, Genentech Hall, Suite N472, San Francisco, CA 94158; and bLaboratory for Molecular Pharmacology, and Department of Neuroscience, Physiology, and Pharmacology, University College London, Gower Street, London WC1E 6BT, United Kingdom Edited by James A. Wells, University of California, San Francisco, CA, and approved April 15, 2009 (received for review January 12, 2009) Protein O-GlcNAcylation occurs in all animals and plants and is and synaptic plasticity have been found O-GlcNAcylated (8). The implicated in modulation of a wide range of cytosolic and nuclear importance of O-GlcNAcylation is highlighted by the brain-specific protein functions, including gene silencing, nutrient and stress sens- OGT knockout mouse model that displays developmental defects ing, phosphorylation signaling, and diseases such as diabetes and that result in neonatal death (9). Most interestingly, in analogy to Alzheimer’s. The limiting factor impeding rapid progress in decipher- phosphorylation, synaptic activity leads to a dynamic modulation of ing the biological functions of protein O-GlcNAcylation has been the O-GlcNAc levels (10) and is also involved in long-term potentiation inability to easily identify exact residues of modification. We describe and memory (11). a robust, high-sensitivity strategy able to assign O-GlcNAcylation sites Phosphorylation is the most heavily studied regulatory PTM, and of native modified peptides using electron transfer dissociation mass proteomic approaches for global enrichment, detection, and char- spectrometry. -

Camk Iialpha) (C6974
Anti-CaM Kinase IIa (CaMK IIa) produced in rabbit, IgG fraction of antiserum Catalog Number C6974 Product Description residue in the autoinhibitory domain 7 (Thr286 in Anti-CaM Kinase IIa (CaMK IIa) is produced in rabbit CaMKIIa and Thr287 in CaMKIIb). Autophosphorylation using as immunogen a synthetic peptide of CaMKIIa at Thr286 has been shown to be required for (KWQIVHFHRSGAPSVLPH) corresponding to the LTP and learning.8 CaMKII activation results in C-terminal region of rat CaM Kinase IIa (amino acids switching of the kinase to a Ca2+/CaM- independent 461-478), conjugated to KLH. This sequence is state and its translocation to the PSD.9,10 identical in human, mouse and chicken CaM Kinase IIa PSD-associated CaMKII in turn phosphorylates and has limited homology (50-60%) with CaM Kinase ionotropic glutamate receptors (e.g. NMDAR, AMPA-R), IIb, g and d subunits. Whole antiserum is purified to thus providing a mechanism for increased synaptic 9-12 provide an IgG fraction of antiserum. signaling during LTP. Anti-CaM Kinase IIa recognizes rat CaM Kinase IIa Reagent (50 kDa). Applications include the detection and Supplied as a solution in 0.01 M phosphate buffered localization of CaM Kinase IIa (50 kDa) by saline, pH 7.4, containing 15 mM sodium azide. immunoblotting. Staining of CaM Kinase IIa in immunoblotting is specifically inhibited with the CaM Precautions and Disclaimer Kinase IIa immunizing peptide. This product is for R&D use only, not for drug, household, or other uses. Please consult the Material Ca2+/Calmodulin dependent protein kinase II (CaMKII) Safety Data Sheet for information regarding hazards belongs to the family of Ser/Thr protein kinases and safe handling practices. -

GSK3 and Its Interactions with the PI3K/AKT/Mtor Signalling Network
Heriot-Watt University Research Gateway GSK3 and its interactions with the PI3K/AKT/mTOR signalling network Citation for published version: Hermida, MA, Kumar, JD & Leslie, NR 2017, 'GSK3 and its interactions with the PI3K/AKT/mTOR signalling network', Advances in Biological Regulation, vol. 65, pp. 5-15. https://doi.org/10.1016/j.jbior.2017.06.003 Digital Object Identifier (DOI): 10.1016/j.jbior.2017.06.003 Link: Link to publication record in Heriot-Watt Research Portal Document Version: Peer reviewed version Published In: Advances in Biological Regulation Publisher Rights Statement: © 2017 Elsevier B.V. General rights Copyright for the publications made accessible via Heriot-Watt Research Portal is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy Heriot-Watt University has made every reasonable effort to ensure that the content in Heriot-Watt Research Portal complies with UK legislation. If you believe that the public display of this file breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 27. Sep. 2021 Accepted Manuscript GSK3 and its interactions with the PI3K/AKT/mTOR signalling network Miguel A. Hermida, J. Dinesh Kumar, Nick R. Leslie PII: S2212-4926(17)30124-0 DOI: 10.1016/j.jbior.2017.06.003 Reference: JBIOR 180 To appear in: Advances in Biological Regulation Received Date: 13 June 2017 Accepted Date: 23 June 2017 Please cite this article as: Hermida MA, Dinesh Kumar J, Leslie NR, GSK3 and its interactions with the PI3K/AKT/mTOR signalling network, Advances in Biological Regulation (2017), doi: 10.1016/ j.jbior.2017.06.003. -

Nercc1, a Mammalian NIMA-Family Kinase, Binds the Ran Gtpase and Regulates Mitotic Progression
Downloaded from genesdev.cshlp.org on September 26, 2021 - Published by Cold Spring Harbor Laboratory Press Nercc1, a mammalian NIMA-family kinase, binds the Ran GTPase and regulates mitotic progression Joan Roig, Alexei Mikhailov, Christopher Belham, and Joseph Avruch1 Department of Molecular Biology and the Diabetes Unit and Medical Services, Massachusetts General Hospital, and the Department of Medicine, Harvard Medical School, Boston, Massachusetts 02114, USA The protein kinase NIMA is an indispensable pleiotropic regulator of mitotic progression in Aspergillus. Although several mammalian NIMA-like kinases (Neks) are known, none appears to have the broad importance for mitotic regulation attributed to NIMA. Nercc1 is a new NIMA-like kinase that regulates chromosome alignment and segregation in mitosis. Its NIMA-like catalytic domain is followed by a noncatalytic tail containing seven repeats homologous to those of the Ran GEF, RCC1, a Ser/Thr/Pro-rich segment, and a coiled-coil domain. Nercc1 binds to another NIMA-like kinase, Nek6, and also binds specifically to the Ran GTPase through both its catalytic and its RCC1-like domains, preferring RanGDP in vivo. Nercc1 exists as a homooligomer and can autoactivate in vitro by autophosphorylation. Nercc1 is a cytoplasmic protein that is activated during mitosis and is avidly phosphorylated by active p34Cdc2. Microinjection of anti-Nercc1 antibodies in prophase results in spindle abnormalities and/or chromosomal misalignment. In Ptk2 cells the outcome is prometaphase arrest or aberrant chromosome segregation and aneuploidy, whereas in CFPAC-1 cells prolonged arrest in prometaphase is the usual response. Nercc1 and its partner Nek6 represent a new signaling pathway that regulates mitotic progression. -

Intracellular Calcium Regulates Amp-Activated Protein Kinase Activity in an Oscillation-Dependent Manner
INTRACELLULAR CALCIUM REGULATES AMP-ACTIVATED PROTEIN KINASE ACTIVITY IN AN OSCILLATION-DEPENDENT MANNER Sungkwon Park, Eric M. England, Haibo Zhu, Jason M. Scheffler, Steve C. Kasten, Tracy L. Scheffler, and * David E. Gerrard Department of Animal and Poultry Sciences, Virginia Tech, Blacksburg, VA, 24061, USA *Corresponding author (phone: +1-540-231-9157; fax: +1-540-231-3010; e-mail: [email protected]) Abstract—Skeletal muscle calcium signaling is important for muscle contraction, as well as regulates many cellular processes. Calcium-regulated calmodulin dependent kinase kinase (CaMKK) has recently been identified as upstream regulator of AMP-activated protein kinase (AMPK), which is energy regulator in skeletal muscle. Although there is evidence that cytosolic calcium regulates AMPK through a series of pathways, the molecular mechanisms by which calcium regulates AMPK are poorly understood. The objective of this study is to understand the function of calcium oscillations on AMPK activity and define the specific calcium-regulated signaling molecules in this pathway. AMPK activity was increased by 2 folds in muscles from mice treated with AICAR (known AMPK activator). Administration of caffeine (calcium releasing agent) for 10 d decreased AICAR-induced AMPK activity to control level. This repressed AMPK activity was blocked by dantrolene, a ryanodine receptor stabilizer. Different calcium frequencies were simulated in C2C12 myotubes by alternating media containing caffeine and dantrolene. Changes in intracellular calcium levels were confirmed by fluorescent calcium indicator, Fura2. To define the function of calcium signaling, CaMKK or CaMK was knocked down. Low frequency calcium stimulations had a positive effect on AICAR-induced AMPK activity, whereas continuous high calcium level decreases AMPK activity suggesting a biphasic control of AMPK activity by calcium. -

Biallelic Loss of Function NEK3 Mutations Deacetylate Α-Tubulin and Downregulate NUP205 That Predispose Individuals to Cilia-Re
Zhang et al. Cell Death and Disease (2020) 11:1005 https://doi.org/10.1038/s41419-020-03214-1 Cell Death & Disease ARTICLE Open Access Biallelic loss of function NEK3 mutations deacetylate α-tubulin and downregulate NUP205 that predispose individuals to cilia-related abnormal cardiac left–right patterning Yuan Zhang1, Weicheng Chen2, Weijia Zeng3, Zhouping Lu4 and Xiangyu Zhou4 Abstract Defective left–right (LR) organization involving abnormalities in cilia ultrastructure causes laterality disorders including situs inversus (SI) and heterotaxy (Htx) with the prevalence approximately 1/10,000 births. In this study, we describe two unrelated family trios with abnormal cardiac LR patterning. Through whole-exome sequencing (WES), we identified compound heterozygous mutations (c.805-1G >C; p. Ile269GlnfsTer8/c.1117dupA; p.Thr373AsnfsTer19) (c.29T>C; p.Ile10Thr/c.356A>G; p.His119Arg) of NEK3, encoding a NIMA (never in mitosis A)-related kinase, in two affected individuals, respectively. Protein levels of NEK3 were abrogated in Patient-1 with biallelic loss-of function (LoF) NEK3 mutations that causes premature stop codon. Subsequence transcriptome analysis revealed that NNMT (nicotinamide N-methyltransferase) and SIRT2 (sirtuin2) was upregulated by NEK3 knockdown in human retinal pigment epithelial (RPE) cells in vitro, which associates α-tubulin deacetylation by western blot and immunofluorescence. Transmission electron microscopy (TEM) analysis further identified defective ciliary ultrastructure 1234567890():,; 1234567890():,; 1234567890():,; 1234567890():,; in Patient-1. Furthermore, inner ring components of nuclear pore complex (NPC) including nucleoporin (NUP)205, NUP188, and NUP155 were significantly downregulated in NEK3-silenced cells. In conclusion, we identified biallelic mutations of NEK3 predispose individual to abnormal cardiac left–right patterning via SIRT2-mediated α-tubulin deacetylation and downregulation of inner ring nucleoporins. -

Modified Recipe to Inhibit GSK-3 for the Living Fungal Biomaterial Manufacture
bioRxiv preprint doi: https://doi.org/10.1101/496265; this version posted December 13, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 Modified recipe to inhibit GSK-3 for the living fungal 2 biomaterial manufacture 1¶ 1¶ 1 1 1 3 Jinhui Chang , Po Lam Chan , Yichun Xie , Man Kit Cheung , Ka Lee Ma , Hoi Shan 1* 4 Kwan 1 5 School of Life Sciences, The Chinese University of Hong Kong, Shatin, New Territories, 6 Hong Kong 7 *Corresponding author: E-mail: [email protected] ¶ 8 These authors contributed equally to this work. 9 10 11 bioRxiv preprint doi: https://doi.org/10.1101/496265; this version posted December 13, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 12 Abstract 13 Living fungal mycelium with suppressed or abolished fruit-forming ability is a self-healing 14 substance particularly valuable biomaterial for further engineering and development in 15 applications such as monitoring/sensing environmental changes and secreting signals. The 16 ability to suppress fungal fruiting is also a useful tool for maintaining stability (e.g., shape, 17 form) of a mycelium-based biomaterial with ease and lower cost. 18 The objective of this present study is to provide a biochemical solution to regulate the fruiting 19 body formation to replace heat killing of mycelium during production.