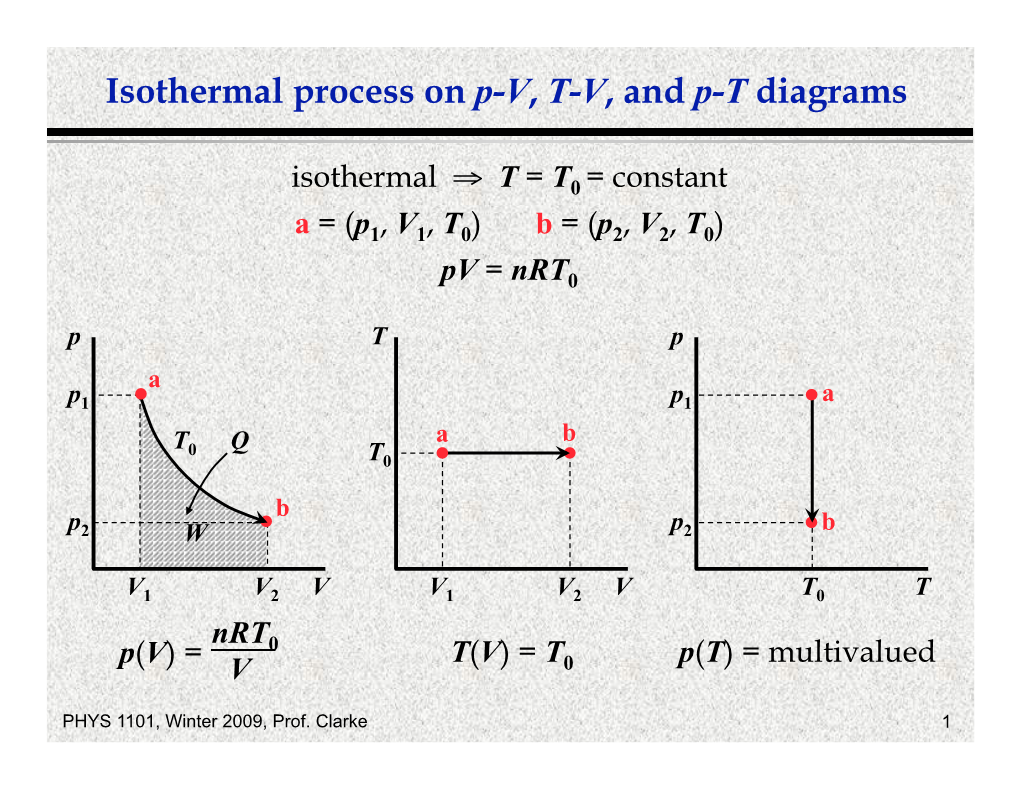
Isothermal Process on P-V, T-V, and P-T Diagrams
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
3-1 Adiabatic Compression
Solution Physics 213 Problem 1 Week 3 Adiabatic Compression a) Last week, we considered the problem of isothermal compression: 1.5 moles of an ideal diatomic gas at temperature 35oC were compressed isothermally from a volume of 0.015 m3 to a volume of 0.0015 m3. The pV-diagram for the isothermal process is shown below. Now we consider an adiabatic process, with the same starting conditions and the same final volume. Is the final temperature higher, lower, or the same as the in isothermal case? Sketch the adiabatic processes on the p-V diagram below and compute the final temperature. (Ignore vibrations of the molecules.) α α For an adiabatic process ViTi = VfTf , where α = 5/2 for the diatomic gas. In this case Ti = 273 o 1/α 2/5 K + 35 C = 308 K. We find Tf = Ti (Vi/Vf) = (308 K) (10) = 774 K. b) According to your diagram, is the final pressure greater, lesser, or the same as in the isothermal case? Explain why (i.e., what is the energy flow in each case?). Calculate the final pressure. We argued above that the final temperature is greater for the adiabatic process. Recall that p = nRT/ V for an ideal gas. We are given that the final volume is the same for the two processes. Since the final temperature is greater for the adiabatic process, the final pressure is also greater. We can do the problem numerically if we assume an idea gas with constant α. γ In an adiabatic processes, pV = constant, where γ = (α + 1) / α. -

Muddiest Point – Entropy and Reversible I Am Confused About Entropy and How It Is Different in a Reversible Versus Irreversible Case
1 Muddiest Point { Entropy and Reversible I am confused about entropy and how it is different in a reversible versus irreversible case. Note: Some of the discussion below follows from the previous muddiest points comment on the general idea of a reversible and an irreversible process. You may wish to have a look at that comment before reading this one. Let's talk about entropy first, and then we will consider how \reversible" gets involved. Generally we divide the universe into two parts, a system (what we are studying) and the surrounding (everything else). In the end the total change in the entropy will be the sum of the change in both, dStotal = dSsystem + dSsurrounding: This total change of entropy has only two possibilities: Either there is no spontaneous change (equilibrium) and dStotal = 0, or there is a spontaneous change because we are not at equilibrium, and dStotal > 0. Of course the entropy change of each piece, system or surroundings, can be positive or negative. However, the second law says the sum must be zero or positive. Let's start by thinking about the entropy change in the system and then we will add the entropy change in the surroundings. Entropy change in the system: When you consider the change in entropy for a process you should first consider whether or not you are looking at an isolated system. Start with an isolated system. An isolated system is not able to exchange energy with anything else (the surroundings) via heat or work. Think of surrounding the system with a perfect, rigid insulating blanket. -

Isothermal Process It Is the Process in Which Other Physical Quantities Might Change but the Temperature of the System Remains Or Is Forced to Remain Constant
Sajit Chandra Shakya Department of Physics Kathmandu Don Bosco College New Baneshwor, Kathmandu Isothermal process It is the process in which other physical quantities might change but the temperature of the system remains or is forced to remain constant. For example, the constant temperature of human body. Under constant temperature, the volume of a gas system is inversely proportional to the pressure applied, the phenomena being called Boyle's Law, written in symbols as 1 V ∝ P 1 Or, V = KB× where KB is a constant quantity. P Or, PV = KB …………….. (i) This means whatever be the values of volume and pressure, their product will be constant. So, P1V1 = KB, P2V2 = KB, P3V3 = KB, etc, Or, P1V1 = P2V2 = P3V3, etc. The requirements for an isothermal process are as follows: 1. The process should be carried very slowly so that there is an ample time for compensation of heat in case of any loss or addition. 2. The boundaries of the system should be highly conducting so that there is a path for heat to flow into or flow away from a closed space in case of any energy loss or oversupply. 3. The boundaries should be made very thin because the resistance of the substance for heat conduction will be less for thin boundaries. Since in an isothermal change, the temperature remains constant, the internal energy also does not change, i.e. dU = 0. So if dQ amount of heat is given to a system which undergoes isothermal change, the relation for the first law of thermodynamics would be dQ = dU + dW Or, dQ = 0 + PdV Or, dQ = PdV This means all the heat supplied will be utilized for performing external work and consequently its value will be very high compared to other processes. -

The First Law of Thermodynamics Continued Pre-Reading: §19.5 Where We Are
Lecture 7 The first law of thermodynamics continued Pre-reading: §19.5 Where we are The pressure p, volume V, and temperature T are related by an equation of state. For an ideal gas, pV = nRT = NkT For an ideal gas, the temperature T is is a direct measure of the average kinetic energy of its 3 3 molecules: KE = nRT = NkT tr 2 2 2 3kT 3RT and vrms = (v )av = = r m r M p Where we are We define the internal energy of a system: UKEPE=+∑∑ interaction Random chaotic between atoms motion & molecules For an ideal gas, f UNkT= 2 i.e. the internal energy depends only on its temperature Where we are By considering adding heat to a fixed volume of an ideal gas, we showed f f Q = Nk∆T = nR∆T 2 2 and so, from the definition of heat capacity Q = nC∆T f we have that C = R for any ideal gas. V 2 Change in internal energy: ∆U = nCV ∆T Heat capacity of an ideal gas Now consider adding heat to an ideal gas at constant pressure. By definition, Q = nCp∆T and W = p∆V = nR∆T So from ∆U = Q W − we get nCV ∆T = nCp∆T nR∆T − or Cp = CV + R It takes greater heat input to raise the temperature of a gas a given amount at constant pressure than constant volume YF §19.4 Ratio of heat capacities Look at the ratio of these heat capacities: we have f C = R V 2 and f + 2 C = C + R = R p V 2 so C p γ = > 1 CV 3 For a monatomic gas, CV = R 3 5 2 so Cp = R + R = R 2 2 C 5 R 5 and γ = p = 2 = =1.67 C 3 R 3 YF §19.4 V 2 Problem An ideal gas is enclosed in a cylinder which has a movable piston. -

Ch 19. the First Law of Thermodynamics
Ch 19. The First Law of Thermodynamics Liu UCD Phy9B 07 1 19-1. Thermodynamic Systems Thermodynamic system: A system that can interact (and exchange energy) with its surroundings Thermodynamic process: A process in which there are changes in the state of a thermodynamic system Heat Q added to the system Q>0 taken away from the system Q<0 (through conduction, convection, radiation) Work done by the system onto its surroundings W>0 done by the surrounding onto the system W<0 Energy change of the system is Q + (-W) or Q-W Gaining energy: +; Losing energy: - Liu UCD Phy9B 07 2 19-2. Work Done During Volume Changes Area: A Pressure: p Force exerted on the piston: F=pA Infinitesimal work done by system dW=Fdx=pAdx=pdV V Work done in a finite volume change W = final pdV ∫V initial Liu UCD Phy9B 07 3 Graphical View of Work Gas expands Gas compresses Constant p dV>0, W>0 dV<0, W<0 W=p(V2-V1) Liu UCD Phy9B 07 4 19-3. Paths Between Thermodynamic States Path: a series of intermediate states between initial state (p1, V1) and a final state (p2, V2) The path between two states is NOT unique. V2 W= p1(V2-V1) +0 W=0+ p2(V2-V1) W = pdV ∫V 1 Work done by the system is path-dependent. Liu UCD Phy9B 07 5 Path Dependence of Heat Transfer Isotherml: Keep temperature const. Insulation + Free expansion (uncontrolled expansion of a gas into vacuum) Heat transfer depends on the initial & final states, also on the path. -

Thermodynamic Propertiesproperties
ThermodynamicThermodynamic PropertiesProperties PropertyProperty TableTable -- from direct measurement EquationEquation ofof StateState -- any equation that relates P,v, and T of a substance jump ExerciseExercise 33--1212 A bucket containing 2 liters of R-12 is left outside in the atmosphere (0.1 MPa) a) What is the R-12 temperature assuming it is in the saturated state. b) the surrounding transfer heat at the rate of 1KW to the liquid. How long will take for all R-12 vaporize? See R-12 (diclorindifluormethane) on Table A-2 SolutionSolution -- pagepage 11 Part a) From table A-2, at the saturation pressure of 0.1 MPa one finds: • Tsaturation = - 30oC 3 • vliq = 0.000672 m /kg 3 • vvap = 0.159375 m /kg • hlv = 165KJ/kg (vaporization heat) SolutionSolution -- pagepage 22 Part b) The mass of R-12 is m = Volume/vL, m = 0.002/0.000672 = 2.98 kg The vaporization energy: Evap = vap energy * mass = 165*2.98 = 492 KJ Time = Heat/Power = 492 sec or 8.2 min GASGAS PROPERTIESPROPERTIES Ideal -Gas Equation of State M PV = nR T; n = u mol Universal gas constant is given on Ru = 8.31434 kJ/kmol-K = 8.31434 kPa-m3/kmol-k = 0.0831434 bar-m3/kmol-K = 82.05 L-atm/kmol-K = 1.9858 Btu/lbmol-R = 1545.35 ft-lbf/lbmol-R = 10.73 psia-ft3/lbmol-R EExamplexample Determine the particular gas constant for air (28.97 kg/kmol) and hydrogen (2.016 kg/kmol). kJ 8.1417 kJ = Ru = kmol − K = 0.287 Rair kg M 28.97 kg − K kmol kJ 8.1417 kJ = kmol − K = 4.124 Rhydrogen kg 2.016 kg − K kmol IdealIdeal GasGas “Law”“Law” isis aa simplesimple EquationEquation ofof StateState PV = MRT Pv = RT PV = NRuT P V P V 1 1 = 2 2 T1 T2 QuestionQuestion …...….. -

12/8 and 12/10/2010
PY105 C1 1. Help for Final exam has been posted on WebAssign. 2. The Final exam will be on Wednesday December 15th from 6-8 pm. First Law of Thermodynamics 3. You will take the exam in multiple rooms, divided as follows: SCI 107: Abbasi to Fasullo, as well as Khajah PHO 203: Flynn to Okuda, except for Khajah SCI B58: Ordonez to Zhang 1 2 Heat and Work done by a Gas Thermodynamics Initial: Consider a cylinder of ideal Thermodynamics is the study of systems involving gas at room temperature. Suppose the piston on top of energy in the form of heat and work. the cylinder is free to move vertically without friction. When the cylinder is placed in a container of hot water, heat Equilibrium: is transferred into the cylinder. Where does the heat energy go? Why does the volume increase? 3 4 The First Law of Thermodynamics The First Law of Thermodynamics The First Law is often written as: Some of the heat energy goes into raising the temperature of the gas (which is equivalent to raising the internal energy of the gas). The rest of it does work by raising the piston. ΔEQWint =− Conservation of energy leads to: QEW=Δ + int (the first law of thermodynamics) This form of the First Law says that the change in internal energy of a system is Q is the heat added to a system (or removed if it is negative) equal to the heat supplied to the system Eint is the internal energy of the system (the energy minus the work done by the system (usually associated with the motion of the atoms and/or molecules). -

17.Examples of the 1St Law Rev2.Nb
17. Examples of the First Law Introduction and Summary The First Law of Thermodynamics is basically a statement of Conservation of Energy. The total energy of a thermodynamic system is called the Internal Energy U. A system can have its Internal Energy changed (DU) in two major ways: (1) Heat Q > 0 can flow into the system from the surroundings, in which case the change in internal energy of the system increases: DU = Q. (2) The system can do work W > 0 on the surroundings, in which case the internal energy of the system decreases: DU = - W. Generally, the system interacts with the surroundings by exchanging both heat Q and doing work W, and the internal energy changes DU = Q - W This is the first law of thermodynamics and in it, W is the thermodynamic work done by the system on the surroundings. If the definition of mechanical work W were used instead of W, then the 1st Law would appear DU = Q + W because doing work on the system by the surroundings would increase the internal energy of the system DU = W. The 1st Law is simply the statement that the internal energy DU of a system can change in two ways: through heat Q absorption where, for example, the temperature changes, or by the system doing work -W, where a macroscopic parameter like volume changes. Below are some examples of using the 1st Law. The Work Done and the Heat Input During an Isobaric Expansion Suppose a gas is inside a cylinder that has a movable piston. The gas is in contact with a heat reservoir that is at a higher temperature than the gas so heat Q flows into the gas from the reservoir. -

BASIC THERMODYNAMICS REFERENCES: ENGINEERING THERMODYNAMICS by P.K.NAG 3RD EDITION LAWS of THERMODYNAMICS
Module I BASIC THERMODYNAMICS REFERENCES: ENGINEERING THERMODYNAMICS by P.K.NAG 3RD EDITION LAWS OF THERMODYNAMICS • 0 th law – when a body A is in thermal equilibrium with a body B, and also separately with a body C, then B and C will be in thermal equilibrium with each other. • Significance- measurement of property called temperature. A B C Evacuated tube 100o C Steam point Thermometric property 50o C (physical characteristics of reference body that changes with temperature) – rise of mercury in the evacuated tube 0o C bulb Steam at P =1ice atm T= 30oC REASONS FOR NOT TAKING ICE POINT AND STEAM POINT AS REFERENCE TEMPERATURES • Ice melts fast so there is a difficulty in maintaining equilibrium between pure ice and air saturated water. Pure ice Air saturated water • Extreme sensitiveness of steam point with pressure TRIPLE POINT OF WATER AS NEW REFERENCE TEMPERATURE • State at which ice liquid water and water vapor co-exist in equilibrium and is an easily reproducible state. This point is arbitrarily assigned a value 273.16 K • i.e. T in K = 273.16 X / Xtriple point • X- is any thermomertic property like P,V,R,rise of mercury, thermo emf etc. OTHER TYPES OF THERMOMETERS AND THERMOMETRIC PROPERTIES • Constant volume gas thermometers- pressure of the gas • Constant pressure gas thermometers- volume of the gas • Electrical resistance thermometer- resistance of the wire • Thermocouple- thermo emf CELCIUS AND KELVIN(ABSOLUTE) SCALE H Thermometer 2 Ar T in oC N Pg 2 O2 gas -273 oC (0 K) Absolute pressure P This absolute 0K cannot be obtatined (Pg+Patm) since it violates third law. -

3. the First Law of Thermodynamics and Related Definitions
09/30/08 1 3. THE FIRST LAW OF THERMODYNAMICS AND RELATED DEFINITIONS 3.1 General statement of the law Simply stated, the First Law states that the energy of the universe is constant. This is an empirical conservation principle (conservation of energy) and defines the term "energy." We will also see that "internal energy" is defined by the First Law. [As is always the case in atmospheric science, we will ignore the Einstein mass-energy equivalence relation.] Thus, the First Law states the following: 1. Heat is a form of energy. 2. Energy is conserved. The ways in which energy is transformed is of interest to us. The First Law is the second fundamental principle in (atmospheric) thermodynamics, and is used extensively. (The first was the equation of state.) One form of the First Law defines the relationship among work, internal energy, and heat input. In this chapter (and in subsequent chapters), we will explore many applications derived from the First Law and Equation of State. 3.2 Work of expansion If a system (parcel) is not in mechanical (pressure) equilibrium with its surroundings, it will expand or contract. Consider the example of a piston/cylinder system, in which the cylinder is filled with a gas. The cylinder undergoes an expansion or compression as shown below. Also shown in Fig. 3.1 is a p-V thermodynamic diagram, in which the physical state of the gas is represented by two thermodynamic variables: p,V in this case. [We will consider this and other types of thermodynamic diagrams in more detail later. -

Exercise 1 Q.5 State the Postulates of Kinetic Theory of Gases
Physics | 13.25 Solved Examples 10−3 × 250 N× 300 JEE Main/Boards = × 23 760 22400 6×× 10 273 Example 1: An electric bulb of volume 250cm³ was sealed off during manufacture at the pressure of −3 23 10× 250 ×× 6 10 × 273 15 10-3mm of Hg at 27°C. Find the number of air molecules N = = 8.02x10 mole 760×× 22400 300 in the bulb. NR Sol: PV= RT = N T = NkT AA Example 2: One gram-mole of oxygen at 27°C and one atmospheric pressure is enclosed in a vessel. Let N be the number of air molecules in the bulb. (a) Assuming the molecules to be moving with V , find V =250cm³, P = 10-3mm of Hg, rms 1 1 the number of collisions per second which molecules T1 = 273 + 27 = 300°K make with one square meter area of the vessel wall. As P11 V= Nk T where k is constant, then (b) The vessel is next thermally insulated and moved with a constant speed ν . It is then suddenly stopped. 10-3x 250 = N.k.300 … (i) 0 The process results in a rise of the temperature of the At N.T.P., one mole of air occupies a volume of 22.4 litre, gas by 1°C. Calculate the speed ν0 . 3 V00= 22400cm ,P= = 760mmofHg,760 mm of Hg, P 3N kT × 23 n = V = A T = 273° K and N0 = 6 10 molecules Sol: Formula based: & rms . Recall kT Mn ∴760 × 22400 = 6x1023 × k × 273 … (ii) the assumption of KTG. -

W 15 “Isotherms of Real Gases”
Fakultät für Physik und Geowissenschaften Physikalisches Grundpraktikum W 15 “Isotherms of Real Gases” Tasks 1. Measure the isotherms of a substance for eight temperatures. Describe the processes that occur close to the critical point. 2. Plot the isotherms and determine the saturation vapour pressure ps in the region of the Maxwell line (coexistence of fluid and vapour). 3. Plot ln(ps) as a function of (1/T). Fit the vapour-pressure equation to the data and determine the average molar latent heat of vaporization of the substance under study. 4. Determine the amount of substance of the substance under study. 5. Use the Clausius-Clapeyron equation to determine the molar heat of vaporization as a function of temperature. Plot the latent heat as a function of the reduced temperature T/TK and fit a power law to the data. Literature Physikalisches Praktikum, Hrsg. W. Schenk, F. Kremer, 13. Auflage, Wärmelehre 2.0.1, 2.0.3 Physics, M. Alonso and E. J. Finn, 15.6 Accessories Instrument for investigation of the critical point, thermostat Keywords for preparation: - Ideal and real gas, kinetic theory, molar volume - Isothermal and adiabatic state transformation - Isothermal, isobaric, isochoric diagrams in p,V,T-space - Isotherms of an ideal and real gas in the p-V diagram - Equation of state after van der Waals, description in the p-V diagram - Maxwell construction, critical point - Compressibility factor z (ideal gas, real gas) - Vapour-pressure curve, Clausius-Clapeyron equation Remarks The “instrument for the investigation of the critical point” (company PHYWE) is already prepared for the measurement of a p-V diagram.