Pterional Transsylvian–Transinsular Approach in Three Cavernomas Of
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans Ten Donkelaar, Nathalie Tzourio-Mazoyer, Jürgen Mai
Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans ten Donkelaar, Nathalie Tzourio-Mazoyer, Jürgen Mai To cite this version: Hans ten Donkelaar, Nathalie Tzourio-Mazoyer, Jürgen Mai. Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex. Frontiers in Neuroanatomy, Frontiers, 2018, 12, pp.93. 10.3389/fnana.2018.00093. hal-01929541 HAL Id: hal-01929541 https://hal.archives-ouvertes.fr/hal-01929541 Submitted on 21 Nov 2018 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. REVIEW published: 19 November 2018 doi: 10.3389/fnana.2018.00093 Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans J. ten Donkelaar 1*†, Nathalie Tzourio-Mazoyer 2† and Jürgen K. Mai 3† 1 Department of Neurology, Donders Center for Medical Neuroscience, Radboud University Medical Center, Nijmegen, Netherlands, 2 IMN Institut des Maladies Neurodégénératives UMR 5293, Université de Bordeaux, Bordeaux, France, 3 Institute for Anatomy, Heinrich Heine University, Düsseldorf, Germany The gyri and sulci of the human brain were defined by pioneers such as Louis-Pierre Gratiolet and Alexander Ecker, and extensified by, among others, Dejerine (1895) and von Economo and Koskinas (1925). -

01 05 Lateral Surface of the Brain-NOTES.Pdf
Lateral Surface of the Brain Medical Neuroscience | Tutorial Notes Lateral Surface of the Brain 1 MAP TO NEUROSCIENCE CORE CONCEPTS NCC1. The brain is the body's most complex organ. LEARNING OBJECTIVES After study of the assigned learning materials, the student will: 1. Demonstrate the four paired lobes of the cerebral cortex and describe the boundaries of each. 2. Sketch the major features of each cerebral lobe, as seen from the lateral view, identifying major gyri and sulci that characterize each lobe. NARRATIVE by Leonard E. WHITE and Nell B. CANT Duke Institute for Brain Sciences Department of Neurobiology Duke University School of Medicine Overview When you view the lateral aspect of a human brain specimen (see Figures A3A and A102), three structures are usually visible: the cerebral hemispheres, the cerebellum, and part of the brainstem (although the brainstem is not visible in the specimen photographed in lateral view for Fig. 1 below). The spinal cord has usually been severed (but we’ll consider the spinal cord later), and the rest of the subdivisions are hidden from lateral view by the hemispheres. The diencephalon and the rest of the brainstem are visible on the medial surface of a brain that has been cut in the midsagittal plane. Parts of all of the subdivisions are also visible from the ventral surface of the whole brain. Over the next several tutorials, you will find video demonstrations (from the brain anatomy lab) and photographs (in the tutorial notes) of these brain surfaces, and sufficient detail in the narrative to appreciate the overall organization of the parts of the brain that are visible from each perspective. -

Acetylcholinesterase Staining in Human Auditory and Language
Acetylcholinesterase Staining in Human Jeffrey J. Hutsler and Michael S. Gazzaniga Auditory and Language Cortices: Center for Neuroscience, University of California, Davis Regional Variation of Structural Features California 95616 Cholinergic innovation of the cerebral neocortex arises from the basal 1974; Greenfield, 1984, 1991; Robertson, 1987; Taylor et al., forebrain and projects to all cortical regions. Acetylcholinesterase 1987; Krisst, 1989; Small, 1989, 1990). AChE-containing axons (AChE), the enzyme responsible for deactivating acetylcholine, is found of the cerebral cortex are also immunoreactive for choline within both cholinergic axons arising from the basal forebrain and a acetyltransferase (ChAT) and are therefore known to be cho- Downloaded from subgroup of pyramidal cells in layers III and V of the cerebral cortex. linergic (Mesulam and Geula, 1992). This pattern of staining varies with cortical location and may contrib- AChE-containing pyramidal cells of layers III and V are not ute uniquely to cortical microcircuhry within functionally distinct cholinergic (Mesulam and Geula, 1991), but it has been sug- regions. To explore this issue further, we examined the pattern of AChE gested that they are cholinoceptive (Krnjevic and Silver, 1965; staining within auditory, auditory association, and putative language Levey et al., 1984; Mesulam et al., 1984b). In support of this regions of whole, postmortem human brains. notion, layer in and V pyramidal cell excitability can be mod- http://cercor.oxfordjournals.org/ The density and distribution of acetylcholine-containing axons and ulated by the application of acetylcholine in the slice prepa- pyramidal cells vary systematically as a function of auditory process- ration (McCormick and Williamson, 1989). Additionally, mus- ing level. -

Human Parietal Cortex in Action Jody C Culham and Kenneth F Valyear
Human parietal cortex in action Jody C Culham and Kenneth F Valyear Experiments using functional neuroimaging and transcranial owes, in large part, to the rapid growth of neuroimaging magnetic stimulation in humans have revealed regions of the studies, particularly experiments using functional mag- parietal lobes that are specialized for particular visuomotor netic resonance imaging (fMRI) and transcranial mag- actions, such as reaching, grasping and eye movements. In netic stimulation (TMS). addition, the human parietal cortex is recruited by processing and perception of action-related information, even when no In one popular view of the visual system [1], visual overt action occurs. Such information can include object shape information is segregated along two pathways: the ventral and orientation, knowledge about how tools are employed and stream (occipito-temporal cortex) computes vision for the understanding of actions made by other individuals. We perception, whereas the dorsal stream (occipito-parietal review the known subregions of the human posterior parietal cortex) computes vision for action. Here, we review cortex and the principles behind their organization. recent advances that address the organization of the posterior parietal cortex and the action-related subregions Addresses within it. We begin by focusing on the role of the dorsal Department of Psychology, Social Science Centre, University of stream in visually-guided real actions. However, we then Western Ontario, London, Ontario, Canada, N6A 5C2 discuss a topic that -

Neuroanatomy: Dissection of the Sheep Brain
Neuroanatomy: Dissection of the Sheep Brain The basic neuroanatomy of the mammalian brain is similar for all species. Instead of using a rodent brain (too small) or a human brain (no volunteer donors), we will study neuroanatomy by examining the brain of the sheep. We will be looking as several structures within the central nervous system (CNS), which is composed of the brain and spinal cord. You will learn individual structures now; later you will be looking at brain systems to see how different parts of the brain form functional units. Just as we differ with respect to individual characteristics, brains also differ slightly in the size, coloration, and shape of some structures. Carefully examine the brain you and your group are dissecting and try to look around the room at some other brains so you can see how the structures may vary. This is especially important when examining the cranial nerves, since rarely will all 12 nerve pairs be preserved on any one brain. This handout will guide you through the dissection and identification of structures. Refer to your text and the accompanying CD for additional information about comparative structures and functions. The last page of this handout lists all the structures you should be prepared to identify for the test. If the structure is marked with *, you should be prepared to identify the function of that structure. Check out the terrific tutorial of a sheep brain dissection on the web site at the University of Scranton (http://academic.uofs.edu/department/psych/sheep/). Lateral View of the Sheep Brain Dorsal Horizontal Section Anterior Posterior Ventral Olfactory bulbs Optic nerves Look for Medulla and chiasm pituitary here External and Midline Anatomy Examine the sheep brain with the membranes intact. -
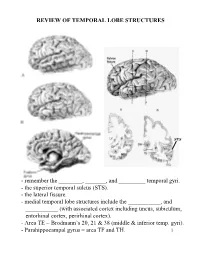
Review of Temporal Lobe Structures
REVIEW OF TEMPORAL LOBE STRUCTURES STS - remember the ________, _______, and _________ temporal gyri. - the superior temporal sulcus (STS). - the lateral fissure. - medial temporal lobe structures include the ___________, and ___________ (with associated cortex including uncus, subiculum, entorhinal cortex, perirhinal cortex). - Area TE = Brodmann’s 20, 21 & 38 (middle & inferior temp. gyri). - Parahippocampal gyrus = area TF and TH. 1 TEMPORAL LOBE FUNCTIONS Sensory Inputs to Temporal lobe: 1. ____________________________________________; 2. _________________________________________________. Temporal cortical regions and functional correlates: 1. Within lateral fissure (superior surface of lateral fissure): a) Heschel’s gyri (_____________________). b) posterior to Heschel’s gyri (_______________________). c) Planum temporale (secondary auditory cortex; Wernicke’s area - specialized in __________________________). 2 Temporal cortical regions and functional correlates (continued): 2. Superior temporal sulcus, middle and inferior temporal gyrus (area TE): _________________________________________ ____________. 3. Ventral/medial surface of temporal lobe (hippocampus and associated cortex): ______________________________. - the ventral/medial surface of the temporal lobe is also associated with the amygdala. Together with the surrounding ventral/medial temporal lobe, the amygdala is involved in __________________________________________. Hemispheric “specialization”: 1. Left hemisphere: a) ________________; b) ____________________________. -

Windows to the Brain: Introduction to Neuroanatomy
VA Mid-Atlantic Health Care Network Windows to the Brain: Introduction to Neuroanatomy Overview Planes of Section Radiographic Perspective Major Divisions Cortical Lobes, Gyri & Sulci General Functions Brodmann’s Areas Basal Forebrain Subcortical Structures Symptoms Cerebellum Structures Symptoms Katherine Taber, PhD, FANPA MIRECC Assistant Director - Education Research Health Scientist W.G. “Bill” Hefner VAMC, Salisbury NC Research Professor, Div Biomedical Sciences Edward Via College of Osteopathic Medicine Robin Hurley, MD, FANPA MIRECC Associate Director - Education ACOS/Research and Academic Affairs Service Line W.G. “Bill” Hefner VAMC, Salisbury NC Professor, Psychiatry and Radiology Wake Forest School of Medicine revised September 2019 Source: http://www.mirecc.va.gov/visn6/Tools-Tips.asp Use of text, images and other content are subject to the following terms and conditions: Fair Use Is Permitted Fair use of copyrighted material includes the use for non-commercial educational purposes, such as teaching, scholarship, research, criticism, commentary, news reporting, and other content. Unless otherwise noted, users who wish to download or print text and image files from this Web site for such uses may do so without the VISN 6 MIRECC’s express permission, provided that they comply with the following conditions: The content may only be used for personal, educational or non- commercial purposes; Users must always specifically cite the author(s) and source of the content every time the material is used, as they would for material from any printed work; None of the content may be altered or modified. Warranty By downloading, printing, or otherwise using text and image files from this website, users agree and warrant that they will limit their use of such files to fair use. -

1. Lateral View of Lobes in Left Hemisphere TOPOGRAPHY
TOPOGRAPHY T1 Division of Cerebral Cortex into Lobes 1. Lateral View of Lobes in Left Hemisphere 2. Medial View of Lobes in Right Hemisphere PARIETAL PARIETAL LIMBIC FRONTAL FRONTAL INSULAR: buried OCCIPITAL OCCIPITAL in lateral fissure TEMPORAL TEMPORAL 3. Dorsal View of Lobes 4. Ventral View of Lobes PARIETAL TEMPORAL LIMBIC FRONTAL OCCIPITAL FRONTAL OCCIPITAL Comment: The cerebral lobes are arbitrary divisions of the cerebrum, taking their names, for the most part, from overlying bones. They are not functional subdivisions of the brain, but serve as a reference for locating specific functions within them. The anterior (rostral) end of the frontal lobe is referred to as the frontal pole. Similarly, the anterior end of the temporal lobe is the temporal pole, and the posterior end of the occipital lobe the occipital pole. TOPOGRAPHY T2 central sulcus central sulcus parietal frontal occipital lateral temporal lateral sulcus sulcus SUMMARY CARTOON: LOBES SUMMARY CARTOON: GYRI Lateral View of Left Hemisphere central sulcus postcentral superior parietal superior precentral gyrus gyrus lobule frontal intraparietal sulcus gyrus inferior parietal lobule: supramarginal and angular gyri middle frontal parieto-occipital sulcus gyrus incision for close-up below OP T preoccipital O notch inferior frontal cerebellum gyrus: O-orbital lateral T-triangular sulcus superior, middle and inferior temporal gyri OP-opercular Lateral View of Insula central sulcus cut surface corresponding to incision in above figure insula superior temporal gyrus Comment: Insula (insular gyri) exposed by removal of overlying opercula (“lids” of frontal and parietal cortex). TOPOGRAPHY T3 Language sites and arcuate fasciculus. MRI reconstruction from a volunteer. central sulcus supramarginal site (posterior Wernicke’s) Language sites (squares) approximated from electrical stimulation sites in patients undergoing operations for epilepsy or tumor removal (Ojeman and Berger). -

Neurosurgical Anatomy of the Insular Cortex
2019/2020 Diogo da Cunha Cabral Neurosurgical anatomy of the insular cortex dezembro, 2019 Diogo da Cunha Cabral Neurosurgical anatomy of the insular cortex Mestrado Integrado em Medicina Área: Medicina Básica Tipologia: Dissertação Trabalho efetuado sob a Orientação de: Professor Doutor José Paulo Andrade E sob a Coorientação de: Professora Doutora Susana Maria Silva Trabalho organizado de acordo com as normas da revista: Clinical Neurology and Neurosurgery dezembro, 2019 1 OIPOJ1TO uc Dissertação/Projeto (6º Ano) - DECLARAÇÃO DE REPRODUÇÃO FMUP FACOU>Aõf.: 1)( Mf:DICJNA \JNIV(ll'SIOAO{PORTO DO NOME j Diogo da Cunha cabral NÚMERO DE ESTUDANTE E-MAIL 1201406119 j [email protected] DESIGNAÇÃO DAÁREA DO PROJECTO j Medicina Básica TÍTULODISSERTAÇÃO Neurosurgical anatomy of the insular cortex ORIENTADOR Professor Doutor José Paulo Alves Vieira de Andrade COORIENTADOR (se aplicável) Professora Doutora Susana Maria de Sousa da Silva Ferreira ASSINALE APENAS UMA DAS OPÇÕES: É AUTORIZADA A REPRODUÇÃOINTEGRAL DESTE TRABALHO APENAS PARA EFEITOSDE INVESTIGAÇÃO, MEDIANTE DECLARAÇÃO ESCRITA DO INTERESSADO, QUE A TAL SE COMPROMETE. D É AUTORIZADAA REPRODUÇÃO PARCIALDESTE TRABALHO (INDICAR, CASOTAL SEJA NECESSÁRIO, Nº MÁXIMODE PÁGINAS,ILUSTRAÇÕES, GRÁFICOS; ETC.)APENAS PARAEFEITOS DE INVESTIGAÇÃO, MEDIANTE D DECLARAÇÃO ESCRITA DO INTERESSADO, QUE A TAL SE COMPROMETE. DE ACORDO COM A LEGISLAÇÃO EM VIGOR, (INDICAR, CASO TAL SEJANECESSÁRIO, Nº MÁXIMO DE PÁGINAS, ILUSTRAÇÕES,GRÁFICOS, ETC.) NÃO É PERMillDAA REPRODUÇÃO DE QUALQUER PARTE DESTETRABALHO. � Faculdade de Medicina da Universidade do Porto, m�J lQ f '1 Assinatura conforme cartão de identificação: Y'7'4' Ú,.tti� /4À4if 2 “As cousas árduas e lustrosas Se alcançam com trabalho e com fadiga” Luís Vaz de Camões, in Os Lusíadas, Canto IV Este pequeno espaço reservado aos agradecimentos nunca será o suficiente para enaltecer o contributo dado por todos aqueles que me acompanharam neste pedaço de vida e me ajudaram a ultrapassar as dificuldades que no seu decurso se me depararam. -

Differential Projection from the Motor and Limbic Cortical Regions to the Mediodorsal Thalamic Nucleus in the Dog
ACTA NEUROBIOL. EXP. 1989. 49: 23-37 DIFFERENTIAL PROJECTION FROM THE MOTOR AND LIMBIC CORTICAL REGIONS TO THE MEDIODORSAL THALAMIC NUCLEUS IN THE DOG Iwona STEPNIEWSKA and Anna KOSMAL Department of Neurophysiology, Nencki Institute of Experimental Biology 3 Pasteur Str., 02-093 Warsaw, Poland Key words: mediodorsal thalamic nucleus, motor and limbic cortices, horseradish peroxidase Abstract. The cortical afferents to the mediodorsal thalamic nucleus in the dog were studied by using horseradish peroxidase. Small injec- tions allowed to establish two specific projection zones connected sepa- rately with the lateral and medial segments of the nucleus. The lateral segment received the major projection from the dorsal half of the hemi- sphere. It included premotor and part of the motor cortices in the ante- rior sigmoid gyrus and precruciate areas as well as the presylvian cor- tex. The medial segment of the nucleus was innervated by the limbic areas of the ventral half of the hemisphere. These areas included the medloventrally located genual, subcallosal and plriform cortices, as well as the cortex of the ventral bank of the anterior rhinal sulcus and the caudal part of the orbital gyrus. The cortical fields situated between these two main cortical zones, both on the lateral and medial surfaces (rhinal and sylvian sulci and anterior cingular gyrus, respectively) sent projec- tions to both medial and lateral segments of the nucleus. These results indicate that in the mediodorsal thalamic nucleus may take place the integration of information from two functionally defined systems, the motor and limbic ones. INTRODUCTIOPJ The numerous studies of the cortical afferents of the mediodorsal thalamic nucleus (MD) showed, that although the prefrontal cortex (PFC) gives rise to the heaviest projection to MD, it is not the only cortical region projecting to thls nucleus. -

Dissection Technique for the Study of the Cerebral Sulci, Gyri and Ventricles
Arq Neuropsiquiatr 2008;66(2-A):282-287 Dissection technique for the stuDy of the cerebral sulci, gyri anD ventricles João Paulo Mattos1, Marcos Juliano dos Santos2, João Flavio Daniel Zullo2, Andrei Fernandes Joaquim2, Feres Chaddad-Neto1, Evandro de Oliveira3 Abstract – Neuroanatomy in addition to neurophysiology, are the basic areas for the proper formation from health students to specialized professionals in neuroscience. A step by step guide for practical studies of neuroanatomy is required for this kind of knowledge to become more acceptable among medical students, neurosurgeons, neurologists, neuropediatricians and psychiatric physicians. Based on the well known courses of sulci, gyri and ventricles offered by Beneficência Portuguesa Hospital in São Paulo, Brazil, two times a year, since 1994, totalizing more than 20 complete courses, and answering the request of many neuroscience students and professionals whose asked for a practical guide to the neuroanatomy study, the authors suggest a protocol for the study of superficial and deep brain structures showing how to approach the more structures as possible with minimum damage to the anatomic piece and with the smaller number of brains. Key wordS: neuroanatomy, brain, dissection technique. técnica de dissecação para o estudo dos sulcos, giros e ventriculos cerebrais Resumo – Neuroanatomia e a neurofisiologia são as áreas básicas para a adequada formação desde estudantes na área da saúde a profissionais especializados em neurociências. Um guia prático, passo a passo, para o estudo -

AN EXPERIMENTAL INVESTIGATION of the CONNEXIONS of the OLFACTORY TRACTS in the MONKEY by MARGARET MEYER and A
J Neurol Neurosurg Psychiatry: first published as 10.1136/jnnp.12.4.274 on 1 November 1949. Downloaded from J. Neurol. Neurosurg. Psychiat., 1949, 12, 274. AN EXPERIMENTAL INVESTIGATION OF THE CONNEXIONS OF THE OLFACTORY TRACTS IN THE MONKEY BY MARGARET MEYER and A. C. ALLISON From the Department ofAnatomy, University of Oxford The great expansion of the-cerebral cortex which bilateral degeneration of olfactory terminals appar- has taken place in higher primates has brought ently passing through the anterior limb of the about a considerable displacement of structures on anterior commissure. The present study has been the base of the telencephalon, and the precise undertaken to map out the connexions of the comparison of certain areas in this part of the olfactory bulb in the monkey's brain as precisely brain with those in lower mammals has been a as possible with the same silver technique. matter of some difficulty. This is true particularly Material and Methods of the olfactory areas which lie on the orbital aspect guest. Protected by copyright. of the frontal lobe and the adjacent part of the Three macaque monkeys (Macaca mulatta) and two this immature Guinea baboons (Papio papio) were used. temporal lobe. Although part of the brain The operative technique was similar in all cases: under in primates has been subjected to detailed cyto- nembutal anesthesia and with the usual aseptic pre- architectural and myelo-architectural examinations cautions a large right frontal bone flap was reflected; (Rose, 1927b, 1928; Beck, 1934, and others), the the frontal lobe of the hemisphere was carefully retraced, areas directly related to olfaction have never been and the olfactory peduncle, lying on the ventral surface, clearly defined.