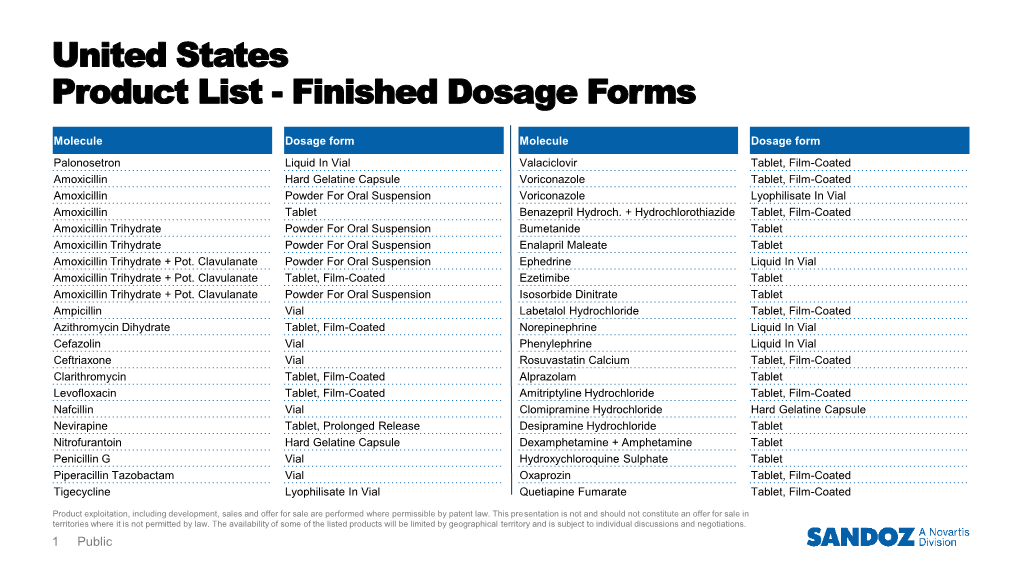
Finished Dosage Forms
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The National Drugs List
^ ^ ^ ^ ^[ ^ The National Drugs List Of Syrian Arab Republic Sexth Edition 2006 ! " # "$ % &'() " # * +$, -. / & 0 /+12 3 4" 5 "$ . "$ 67"5,) 0 " /! !2 4? @ % 88 9 3: " # "$ ;+<=2 – G# H H2 I) – 6( – 65 : A B C "5 : , D )* . J!* HK"3 H"$ T ) 4 B K<) +$ LMA N O 3 4P<B &Q / RS ) H< C4VH /430 / 1988 V W* < C A GQ ") 4V / 1000 / C4VH /820 / 2001 V XX K<# C ,V /500 / 1992 V "!X V /946 / 2004 V Z < C V /914 / 2003 V ) < ] +$, [2 / ,) @# @ S%Q2 J"= [ &<\ @ +$ LMA 1 O \ . S X '( ^ & M_ `AB @ &' 3 4" + @ V= 4 )\ " : N " # "$ 6 ) G" 3Q + a C G /<"B d3: C K7 e , fM 4 Q b"$ " < $\ c"7: 5) G . HHH3Q J # Hg ' V"h 6< G* H5 !" # $%" & $' ,* ( )* + 2 ا اوا ادو +% 5 j 2 i1 6 B J' 6<X " 6"[ i2 "$ "< * i3 10 6 i4 11 6! ^ i5 13 6<X "!# * i6 15 7 G!, 6 - k 24"$d dl ?K V *4V h 63[46 ' i8 19 Adl 20 "( 2 i9 20 G Q) 6 i10 20 a 6 m[, 6 i11 21 ?K V $n i12 21 "% * i13 23 b+ 6 i14 23 oe C * i15 24 !, 2 6\ i16 25 C V pq * i17 26 ( S 6) 1, ++ &"r i19 3 +% 27 G 6 ""% i19 28 ^ Ks 2 i20 31 % Ks 2 i21 32 s * i22 35 " " * i23 37 "$ * i24 38 6" i25 39 V t h Gu* v!* 2 i26 39 ( 2 i27 40 B w< Ks 2 i28 40 d C &"r i29 42 "' 6 i30 42 " * i31 42 ":< * i32 5 ./ 0" -33 4 : ANAESTHETICS $ 1 2 -1 :GENERAL ANAESTHETICS AND OXYGEN 4 $1 2 2- ATRACURIUM BESYLATE DROPERIDOL ETHER FENTANYL HALOTHANE ISOFLURANE KETAMINE HCL NITROUS OXIDE OXYGEN PROPOFOL REMIFENTANIL SEVOFLURANE SUFENTANIL THIOPENTAL :LOCAL ANAESTHETICS !67$1 2 -5 AMYLEINE HCL=AMYLOCAINE ARTICAINE BENZOCAINE BUPIVACAINE CINCHOCAINE LIDOCAINE MEPIVACAINE OXETHAZAINE PRAMOXINE PRILOCAINE PREOPERATIVE MEDICATION & SEDATION FOR 9*: ;< " 2 -8 : : SHORT -TERM PROCEDURES ATROPINE DIAZEPAM INJ. -

Role of Corticosteroids in Treatment of Vitiligo
Chapter 9 Role of Corticosteroids in Treatment of Vitiligo Nooshin Bagherani Additional information is available at the end of the chapter http://dx.doi.org/10.5772/48384 1. Introduction Vitiligo is the most frequent pigmentary disorder ( Bagherani et al., 2011; Nazer et al., 2011;Yaghoobi et al., 2011a; as cited in Wolff et al., 2007). It is an acquired, idiopathic and progressive skin disease (Bagherani et al., 2011; Shameer et al., 2005; Yaghoobi et al., 2011a), characterized by sharply demarcated depigmented lesions on any part of the body (Van Geel et al., 2004). This disease can also affect hair and mucosal areas such as mouth and genitalia (Gawkrodger et al., 2010). Vitiligo usually begins after birth (Gawkrodger et al., 2010). Regarding the studies retrieved from PubMed since 1995, it has been shown that approximately 50% of the vitiligo cases have its onset before the age of 20 years and 25% before the age of 14 years (Kakourou, 2009). The incidence rate of vitiligo is between 0.1-2% of the world population ( Bagherani et al., 20011; Yaghoobi et al., 2011a,b; as cited in Alkhateeb et al., 2003). Its incidence in those with racially pigmented skin is higher, although reliable figures are not available (Burns et al., 2004; Howitz et al., 1977) . The prevalence has been reported as high as 4% in some South Asian, Mexican and American populations ( Parsad et al., 2003; Sehgal & Srivastava, 2007; Szczurko & Boon, 2008). Both sexes are equally afflicted by vitiligo (Krϋger et al., 2011; Njoo & Westerhof, 2001; Wolf et al.,2007) . In some studies, a female preponderance has been reported ( Burns at al., 2004; Howitz et al. -

(CD-P-PH/PHO) Report Classification/Justifica
COMMITTEE OF EXPERTS ON THE CLASSIFICATION OF MEDICINES AS REGARDS THEIR SUPPLY (CD-P-PH/PHO) Report classification/justification of medicines belonging to the ATC group D07A (Corticosteroids, Plain) Table of Contents Page INTRODUCTION 4 DISCLAIMER 6 GLOSSARY OF TERMS USED IN THIS DOCUMENT 7 ACTIVE SUBSTANCES Methylprednisolone (ATC: D07AA01) 8 Hydrocortisone (ATC: D07AA02) 9 Prednisolone (ATC: D07AA03) 11 Clobetasone (ATC: D07AB01) 13 Hydrocortisone butyrate (ATC: D07AB02) 16 Flumetasone (ATC: D07AB03) 18 Fluocortin (ATC: D07AB04) 21 Fluperolone (ATC: D07AB05) 22 Fluorometholone (ATC: D07AB06) 23 Fluprednidene (ATC: D07AB07) 24 Desonide (ATC: D07AB08) 25 Triamcinolone (ATC: D07AB09) 27 Alclometasone (ATC: D07AB10) 29 Hydrocortisone buteprate (ATC: D07AB11) 31 Dexamethasone (ATC: D07AB19) 32 Clocortolone (ATC: D07AB21) 34 Combinations of Corticosteroids (ATC: D07AB30) 35 Betamethasone (ATC: D07AC01) 36 Fluclorolone (ATC: D07AC02) 39 Desoximetasone (ATC: D07AC03) 40 Fluocinolone Acetonide (ATC: D07AC04) 43 Fluocortolone (ATC: D07AC05) 46 2 Diflucortolone (ATC: D07AC06) 47 Fludroxycortide (ATC: D07AC07) 50 Fluocinonide (ATC: D07AC08) 51 Budesonide (ATC: D07AC09) 54 Diflorasone (ATC: D07AC10) 55 Amcinonide (ATC: D07AC11) 56 Halometasone (ATC: D07AC12) 57 Mometasone (ATC: D07AC13) 58 Methylprednisolone Aceponate (ATC: D07AC14) 62 Beclometasone (ATC: D07AC15) 65 Hydrocortisone Aceponate (ATC: D07AC16) 68 Fluticasone (ATC: D07AC17) 69 Prednicarbate (ATC: D07AC18) 73 Difluprednate (ATC: D07AC19) 76 Ulobetasol (ATC: D07AC21) 77 Clobetasol (ATC: D07AD01) 78 Halcinonide (ATC: D07AD02) 81 LIST OF AUTHORS 82 3 INTRODUCTION The availability of medicines with or without a medical prescription has implications on patient safety, accessibility of medicines to patients and responsible management of healthcare expenditure. The decision on prescription status and related supply conditions is a core competency of national health authorities. -

2018 Medicines in Development for Skin Diseases
2018 Medicines in Development for Skin Diseases Acne Drug Name Sponsor Indication Development Phase ADPS topical Taro Pharmaceuticals USA acne vulgaris Phase II completed Hawthorne, NY www.taro.com AOB101 AOBiome acne vulgaris Phase II (topical ammonia oxidizing bacteria) Cambridge, MA www.aobiome.com ASC-J9 AndroScience acne vulgaris Phase II (androgen receptor degradation Solana Beach, CA www.androscience.com enhancer) BLI1100 Braintree Laboratories acne vulgaris Phase II completed Braintree, MA www.braintreelabs.com BPX-01 BioPharmX acne vulgaris Phase II (minocycline topical) Menlo Park, CA www.biopharmx.com BTX1503 Botanix Pharmaceuticals moderate to severe acne vulgaris Phase II (cannabidiol) Plymouth Meeting, PA www.botanixpharma.com CJM112 Novartis Pharmaceuticals acne vulgaris Phase II (IL-17A protein inhibitor) East Hanover, NJ www.novartis.com clascoterone Cassiopea acne vulgaris Phase III (androgen receptor antagonist) Lainate, Italy www.cassiopea.com Medicines in Development: Skin Diseases ǀ 2018 Update 1 Acne Drug Name Sponsor Indication Development Phase CLS001 Cutanea acne vulgaris Phase II (omiganan) Wayne, PA www.cutanea.com DFD-03 Promius Pharma acne vulgaris Phase III (tazarotene topical) Princeton, NJ www.promiuspharma.com DMT310 Dermata Therapeutics moderate to severe acne vulgaris Phase II (freshwater sponge-derived) San Diego, CA www.dermatarx.com finasteride Elorac severe nodulocystic acne Phase II (cholestenone 5-alpha Vernon Hills, IL www.eloracpharma.com reductase inhibitor) FMX101 Foamix moderate to severe -

(12) Patent Application Publication (10) Pub. No.: US 2009/0099225A1 Freund Et Al
US 20090099225A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2009/0099225A1 Freund et al. (43) Pub. Date: Apr. 16, 2009 (54) METHOD FOR THE PRODUCTION OF Related U.S. Application Data PROPELLANT GAS-FREE AEROSOLS FROM (63) Continuation of application No. 1 1/506,128, filed on AQUEOUSMEDICAMENT PREPARATIONS Aug. 17, 2006, now Pat. No. 7,470,422, which is a continuation of application No. 10/417.766, filed on (75) Inventors: Bernhard Freund, Gau-Algesheim Apr. 17, 2003, now abandoned, which is a continuation (DE); Bernd Zierenberg, Bingen of application No. 09/331,023, filed on Sep. 15, 1999, am Rhein (DE) now abandoned. (30) Foreign Application Priority Data Correspondence Address: MICHAEL P. MORRIS Dec. 20, 1996 (DE) ............................... 19653969.2 BOEHRINGERINGELHEMI USA CORPORA Dec. 16, 1997 (EP) ......................... PCT/EP97/07062 TION Publication Classification 900 RIDGEBURY RD, P. O. BOX 368 RIDGEFIELD, CT 06877-0368 (US) (51) Int. Cl. A63L/46 (2006.01) (73) Assignee: Boehringer Ingelheim Pharma A63L/437 (2006.01) KG, Ingelheim (DE) (52) U.S. Cl. ......................................... 514/291; 514/299 (57) ABSTRACT (21) Appl. No.: 12/338,812 The present invention relates to pharmaceutical preparations in the form of aqueous solutions for the production of propel (22) Filed: Dec. 18, 2008 lant-free aerosols. US 2009/0099225A1 Apr. 16, 2009 METHOD FOR THE PRODUCTION OF 0008 All substances which are suitable for application by PROPELLANT GAS-FREE AEROSOLS FROM inhalation and which are soluble in the specified solvent can AQUEOUSMEDICAMENT PREPARATIONS be used as pharmaceuticals in the new preparations. Pharma ceuticals for the treatment of diseases of the respiratory pas RELATED APPLICATIONS sages are of especial interest. -

Different Strategies for Using Topical Corticosteroids in People with Eczema (Protocol)
Cochrane Database of Systematic Reviews Different strategies for using topical corticosteroids in people with eczema (Protocol) Chalmers JR, Axon E, Harvey J, Santer M, Ridd MJ, Lawton S, Langan S, Roberts A, Ahmed A, Muller I, Long CM, Panda S, Chernyshov P, Carter B, Williams HC, Thomas KS Chalmers JR, Axon E, Harvey J, Santer M, Ridd MJ, Lawton S, Langan S, Roberts A, Ahmed A, Muller I, Long CM, Panda S, Chernyshov P, Carter B, Williams HC, Thomas KS. Different strategies for using topical corticosteroids in people with eczema. Cochrane Database of Systematic Reviews 2019, Issue 6. Art. No.: CD013356. DOI: 10.1002/14651858.CD013356. www.cochranelibrary.com Different strategies for using topical corticosteroids in people with eczema (Protocol) Copyright © 2019 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd. TABLE OF CONTENTS HEADER....................................... 1 ABSTRACT ...................................... 1 BACKGROUND .................................... 1 OBJECTIVES ..................................... 4 METHODS ...................................... 4 ACKNOWLEDGEMENTS . 8 REFERENCES ..................................... 9 APPENDICES ..................................... 12 CONTRIBUTIONSOFAUTHORS . 13 DECLARATIONSOFINTEREST . 14 SOURCESOFSUPPORT . 15 Different strategies for using topical corticosteroids in people with eczema (Protocol) i Copyright © 2019 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd. [Intervention Protocol] Different strategies for using topical corticosteroids in -

Effect of Different Skin Penetration Promoters in Halobetasol Propionate Permeation and Retention in Human Skin
International Journal of Molecular Sciences Article Effect of Different Skin Penetration Promoters in Halobetasol Propionate Permeation and Retention in Human Skin Paulina Carvajal-Vidal 1,2,* ID , Mireia Mallandrich 1 ID , María Luisa García 1,2 and Ana Cristina Calpena 1,2 ID 1 Department of Pharmacy, Pharmaceutical Technology and Physical Chemistry, Faculty of Pharmacy and Food Sciences, University of Barcelona, 08028 Barcelona, Spain; [email protected] (M.M.); [email protected] or [email protected] (M.L.G.); [email protected] (A.C.C.) 2 Institute of Nanoscience and Nanotechnology (IN2UB), University of Barcelona, 08028 Barcelona, Spain * Correspondence: [email protected]; Tel.: +34-657-133227 Received: 29 October 2017; Accepted: 17 November 2017; Published: 21 November 2017 Abstract: Halobetasol propionate (HB) is a potent synthetic corticosteroid used against inflammatory skin diseases, such as dermatitis, eczema, and psoriasis, among others. The aim of this study is to define how the presence of different skin penetration enhancers (nonane, menthone, limonene, azone, carene, decanol, linoleic acid and cetiol) affects the penetration and retention in skin of HB. To determine drug penetration through skin, 5% of each promoter was used in an ex vivo system with human skin on Franz cells. The results showed that the highest permeation occurs in the presence of menthone, followed by nonane. Permeation parameters were determined. The in vivo test was assessed, and the formulation containing HB-menthone presented better anti-inflammatory efficacy. These results are useful to generate a specific treatment according to each patient’s needs, and the inflammatory characteristics of the disease. Keywords: halobetasol propionate; permeation enhancers; skin permeation; skin inflammation; topical corticosteroid 1. -

ULTRAVATE® Product Monograph Page 2 of 27
PRODUCT MONOGRAPH Tri-halogenated PrULTRAVATE® Halobetasol Propionate Topical Cream 0.05 % w/w Halobetasol Propionate Topical Ointment 0.05 % w/w Topical Corticosteroid Bausch Health, Canada Inc. Date of Revision: 2150 St-Elzear Blvd., West October 15, 2019 Laval, Quebec, H7L 4A8 Canada Submission Control No: 230873 NAME OF DRUG PrULTRAVATE® Halobetasol Propionate Topical Cream 0.05 % w/w Halobetasol Propionate Topical Ointment 0.05 % w/w THERAPEUTIC CLASSIFICATION Topical Corticosteroid INDICATIONS AND CLINICAL UES ULTRAVATE® (halobetasol propionate) Cream and Ointment are high to super-high potency topical corticosteroids indicated for the relief of inflammatory manifestations of resistant or severe psoriasis and corticosteroid-responsive dermatoses. These products are not recommended for use in children. CONTRAINDICATIONS ULTRAVATE® (halobetasol propionate) Cream and Ointment are contraindicated in patients who are hypersensitive to halobetasol propionate, to other corticosteroids, or to any of the ingredients in these products. ULTRAVATE® (halobetasol propionate) Cream and Ointment are contraindicated in viral diseases of the skin including herpes simplex, vaccinia and varicella. They are also contraindicated in untreated bacterial, tubercular and fungal infections involving the skin. WARNINGS Use in Pregnancy There are no clinical trials of ULTRAVATE® in pregnant women. Therefore, this product should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Corticosteroids have been shown to be teratogenic and embryotoxic in laboratory animals at low doses when administered systemically. Some corticosteroids have been shown to be teratogenic after topical application. Halobetasol propionate has been shown to be teratogenic in rats and rabbits at low doses. The human topical dose of ULTRAVATE ® was embryotoxic in rabbits. -

Stembook 2018.Pdf
The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 WHO/EMP/RHT/TSN/2018.1 © World Health Organization 2018 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances. Geneva: World Health Organization; 2018 (WHO/EMP/RHT/TSN/2018.1). Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. -

Method and Use for Increasing Efficacy of Anti-Adhesive Compositions in Controlling Inflammation and Pain
(19) & (11) EP 2 465 513 A2 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 20.06.2012 Bulletin 2012/25 A61K 31/77 (2006.01) A61K 33/06 (2006.01) A61K 45/06 (2006.01) A61P 29/00 (2006.01) (21) Application number: 11195175.2 (22) Date of filing: 12.11.2007 (84) Designated Contracting States: (72) Inventor: Chamness, Kathy L. AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Memphis, TN 38104-5305 (US) HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE SI SK TR (74) Representative: O’Connell, Maura et al FRKelly (30) Priority: 13.11.2006 US 598397 27 Clyde Road Ballsbridge (62) Document number(s) of the earlier application(s) in Dublin 4 (IE) accordance with Art. 76 EPC: 07864257.6 / 2 104 505 Remarks: •This application was filed on 22-12-2011 as a (71) Applicant: Warsaw Orthopedic, Inc. divisional application to the application mentioned Warsaw, IN 46581 (US) under INID code 62. •Claims filed after the date of filing of the application (Rule 68(4) EPC). (54) Method and use for increasing efficacy of anti-adhesive compositions in controlling inflammation and pain (57) The invention discloses a method and kit thereof prising an effective amount of at least one pharmaceuti- for increasing the efficiency of anti-adhesive composi- cally-acceptable anti-adhesive non-ionic polymer to a tions by parenterally administering a composition com- site of injury, controlling inflammation at the site of injury, and reducing pain. EP 2 465 513 A2 Printed by Jouve, 75001 PARIS (FR) EP 2 465 513 A2 Description FIELD OF THE INVENTION 5 [0001] The present invention relates to methods of increasing efficacy of anti-adhesive compositions by parental administration of compositions containing anti-adhesive polymers and magnesium salts. -

Stability of Betamethasone Esters in Some Topical Dosage Forms and Its Impact on Their Biological Potential
STABILITY OF BETAMETHASONE ESTERS IN SOME TOPICAL DOSAGE FORMS AND ITS IMPACT ON THEIR BIOLOGICAL POTENTIAL Thesis submitted in partial fulfillment of the requirement for the degree of DOCTOR OF PHILOSOPHY by Saif-ur-Rehman Khattak B.Pharm, M.Pharm SUPERVISOR: PROF. DR. DILNAWAZ SHEIKH CO-SUPERVISOR: PROF. DR. USMAN GHANI KHAN Faculty of Pharmacy HAMDARD UNIVERSITY Karachi – 74600 March 2010 iii ABSTRACT The present work involves an investigation of the thermal and photochemical degradation of betamethasone esters i.e. betamethasone valerate and betamethasone dipropionate under various conditions and the evaluation of the photoxicity of these compounds. The thermal degradation (40 oC) of betamethasone-17-valerate leads to the formation of betamethasone-21-valerate and betamethasone alcohol whereas betamethasone dipropionate gives rise to betamethasone-17-propionate, betamethasone-21-propionate and betamethasone alcohol at pH 2.5-7.5, betamethasone-21-propionate being an intermediate in this reaction. The betamethasone esters on photodegradation, using a UV radiation source (300-400nm), yield two major unknown products in aqueous and organic solvents. The detection of the photodegradation products of betamethasone valerate and betamethasone dipropionate has been carried out by HPLC and the t R values of the unknown products have been reported. The USP HPLC method, after proper validation, has been used for the assay of betamethasone esters and their thermal and photodegradation products. The analytical data have been used to evaluate the kinetics of thermal and photochemical reactions. In both reactions the betamethasone esters have been found to follow the first-order kinetics under the conditions employed. The apparent first-order rate constants for the thermal degradation of betamethasone valerate and betamethasone dipropionate in various media lie in the range of 0.339-9.07x10 -3 hr -1 and 0.239-1.87x10 -3 hr -1, respectively. -

(12) United States Patent (10) Patent No.: US 8,859,774 B2 Hunt Et Al
US008859.774B2 (12) United States Patent (10) Patent No.: US 8,859,774 B2 Hunt et al. (45) Date of Patent: Oct. 14, 2014 (54) HETEROARYL-KETONE FUSED (56) References Cited AZADECALN GLUCOCORTICOD RECEPTORMODULATORS U.S. PATENT DOCUMENTS 7,678,813 B2 3/2010 Clark et al. (71) Applicant: Corcept Therapeutics, Inc., Menlo 7,790,745 B2 9/2010 Yang et al. Park, CA (US) 7,928,237 B2 4/2011 Clark et al. 8.461,172 B2 6, 2013 Clark et al. (72) Inventors: Hazel Hunt, West Sussex (GB); Tony 8,598,154 B2 * 12/2013 Clark et al. .............. 514,210.21 2007/0281928 A1 12/2007 Clark et al. Johnson, Essex (GB); Nicholas Ray, 2008.OO70950 A1 3/2008 Benjamin et al. Essex (GB); Iain Walters, Nottingham 2010, O292.477 A1 11, 2010 Clark et al. (GB) 2012fO220565 A1 8, 2012 Clarket al. 2013,0225633 A1 8, 2013 Hunt et al. (73) Assignee: Corcept Therapeutics, Inc., Menlo Park, CA (US) FOREIGN PATENT DOCUMENTS (*) Notice: Subject to any disclaimer, the term of this EP O145121 A2 6, 1985 EP O37521.0 A1 6, 1990 patent is extended or adjusted under 35 JP 9-505030 A 5, 1997 U.S.C. 154(b) by 0 days. JP 2002-506032. A 2, 2002 JP 2002-544271 A 12/2002 (21) Appl. No.: 13/901.946 WO 95,04734 A1 2, 1995 WO 99.45925 A1 9, 1999 (22) Filed: May 24, 2013 WO OO6984.6 A1 11, 2000 WO O3,O15692 A2 2, 2003 WO O3,061651 A1 T 2003 (65) Prior Publication Data WO 2005/087769 A1 9, 2005 US 2014/OO38926A1 Feb.