T-Cell Clonality and Myelodysplasia Without Chromo- Somal Fragility in a Patient with Features of Seckel Syndrome
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Green Teeth Associated Hyperbilirubinemia in Primary Dentition
https://doi.org/10.5933/JKAPD.2017.44.3.378 J Korean Acad Pediatr Dent 44(3) 2017 ISSN (print) 1226-8496 ISSN (online) 2288-3819 Green Teeth Associated Hyperbilirubinemia in Primary Dentition Min Kyung Park1†, Yeji Sun1†, Chung-Min Kang1, Hyo-Seol Lee2, Je Seon Song1 1Department of Pediatric Dentistry, College of Dentistry, Yonsei University 2Department of Pediatric Dentistry, College of Dentistry, Kyung-Hee University Abstract There are many reasons for tooth discoloration. An increase in the bilirubin level may cause tooth discolorations. Such cases are rare, but most involve tooth discoloration with a greenish hue. The purpose of this case report is to describe green discoloration of the primary dentition in the presence of neonatal hyperbilirubinemia. 2 boys aged 16 and 22-months presented with chief complaints of erupting teeth of abnormal color. Their primary teeth exhibited a greenish discoloration along enamel hypoplasia. Both patients were born prematurely with a low birth weight and had been diagnosed with neonatal hyperbilirubinemia. Systematic diseases can affect the hard tissue of teeth during their formation and result in changes in tooth color. Periodic follow-ups are required for establishing a normal dental condition and meeting the esthetic needs of patients. A pediatric dentist may be the first person to observe patients with discoloration in their primary dentition. In such cases the dentist can deduce the systematic disease responsible for this discoloration. Key words : Green teeth, Intrinsic discoloration, Hyperbilirubinemia, Bilirubin Ⅰ. Introduction ative materials infiltrate into the tooth structure, their removal is impossible[5-8]. Tooth discoloration can be an esthetic prob- There are many reasons for tooth discoloration, which can lem, and it is one of main reasons why patients visit dentists. -
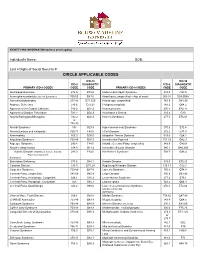
Circle Applicable Codes
IDENTIFYING INFORMATION (please print legibly) Individual’s Name: DOB: Last 4 Digits of Social Security #: CIRCLE APPLICABLE CODES ICD-10 ICD-10 ICD-9 DIAGNOSTIC ICD-9 DIAGNOSTIC PRIMARY ICD-9 CODES CODE CODE PRIMARY ICD-9 CODES CODE CODE Abetalipoproteinemia 272.5 E78.6 Hallervorden-Spatz Syndrome 333.0 G23.0 Acrocephalosyndactyly (Apert’s Syndrome) 755.55 Q87.0 Head Injury, unspecified – Age of onset: 959.01 S09.90XA Adrenaleukodystrophy 277.86 E71.529 Hemiplegia, unspecified 342.9 G81.90 Arginase Deficiency 270.6 E72.21 Holoprosencephaly 742.2 Q04.2 Agenesis of the Corpus Callosum 742.2 Q04.3 Homocystinuria 270.4 E72.11 Agenesis of Septum Pellucidum 742.2 Q04.3 Huntington’s Chorea 333.4 G10 Argyria/Pachygyria/Microgyria 742.2 Q04.3 Hurler’s Syndrome 277.5 E76.01 or 758.33 Aicardi Syndrome 333 G23.8 Hyperammonemia Syndrome 270.6 E72.4 Alcohol Embryo and Fetopathy 760.71 F84.5 I-Cell Disease 272.2 E77.0 Anencephaly 655.0 Q00.0 Idiopathic Torsion Dystonia 333.6 G24.1 Angelman Syndrome 759.89 Q93.5 Incontinentia Pigmenti 757.33 Q82.3 Asperger Syndrome 299.8 F84.5 Infantile Cerebral Palsy, unspecified 343.9 G80.9 Ataxia-Telangiectasia 334.8 G11.3 Intractable Seizure Disorder 345.1 G40.309 Autistic Disorder (Childhood Autism, Infantile 299.0 F84.0 Klinefelter’s Syndrome 758.7 Q98.4 Psychosis, Kanner’s Syndrome) Biotinidase Deficiency 277.6 D84.1 Krabbe Disease 333.0 E75.23 Canavan Disease 330.0 E75.29 Kugelberg-Welander Disease 335.11 G12.1 Carpenter Syndrome 759.89 Q87.0 Larsen’s Syndrome 755.8 Q74.8 Cerebral Palsy, unspecified 343.69 G80.9 -

MECHANISMS in ENDOCRINOLOGY: Novel Genetic Causes of Short Stature
J M Wit and others Genetics of short stature 174:4 R145–R173 Review MECHANISMS IN ENDOCRINOLOGY Novel genetic causes of short stature 1 1 2 2 Jan M Wit , Wilma Oostdijk , Monique Losekoot , Hermine A van Duyvenvoorde , Correspondence Claudia A L Ruivenkamp2 and Sarina G Kant2 should be addressed to J M Wit Departments of 1Paediatrics and 2Clinical Genetics, Leiden University Medical Center, PO Box 9600, 2300 RC Leiden, Email The Netherlands [email protected] Abstract The fast technological development, particularly single nucleotide polymorphism array, array-comparative genomic hybridization, and whole exome sequencing, has led to the discovery of many novel genetic causes of growth failure. In this review we discuss a selection of these, according to a diagnostic classification centred on the epiphyseal growth plate. We successively discuss disorders in hormone signalling, paracrine factors, matrix molecules, intracellular pathways, and fundamental cellular processes, followed by chromosomal aberrations including copy number variants (CNVs) and imprinting disorders associated with short stature. Many novel causes of GH deficiency (GHD) as part of combined pituitary hormone deficiency have been uncovered. The most frequent genetic causes of isolated GHD are GH1 and GHRHR defects, but several novel causes have recently been found, such as GHSR, RNPC3, and IFT172 mutations. Besides well-defined causes of GH insensitivity (GHR, STAT5B, IGFALS, IGF1 defects), disorders of NFkB signalling, STAT3 and IGF2 have recently been discovered. Heterozygous IGF1R defects are a relatively frequent cause of prenatal and postnatal growth retardation. TRHA mutations cause a syndromic form of short stature with elevated T3/T4 ratio. Disorders of signalling of various paracrine factors (FGFs, BMPs, WNTs, PTHrP/IHH, and CNP/NPR2) or genetic defects affecting cartilage extracellular matrix usually cause disproportionate short stature. -

Genetic Short Stature ⁎ Michelle Grunauera, Alexander A.L
Growth Hormone & IGF Research xxx (xxxx) xxx–xxx Contents lists available at ScienceDirect Growth Hormone & IGF Research journal homepage: www.elsevier.com/locate/ghir Genetic short stature ⁎ Michelle Grunauera, Alexander A.L. Jorgeb, a Pediatric Intensive Care Unit, Hospital de los Valles, Escuela de Medicina, Universidad San Francisco de Quito (USFQ), Quito, Ecuador b Unidade de Endocrinologia Genetica (LIM25), Hospital das Clinicas da Faculdade de Medicina da Universidade de São Paulo (FMUSP), Sao Paulo, Brazil ARTICLE INFO ABSTRACT Keywords: Adult height and growth patterns are largely genetically programmed. Studies in twins have indicated that the Genetic heritability of height is high (> 80%), suggesting that genetic variation is the main determinant of stature. Short stature Height exhibits a normal (Gaussian) distribution according to sex, age, and ancestry. Short stature is usually Skeletal dysplasia defined as a height which is 2 standard deviations (S.D.) less than the mean height of a specific population. This Syndrome definition includes 2.3% of the population and usually includes healthy individuals. In this group of short stature Adult height non-syndromic conditions, the genetic influence occurs polygenically or oligogenically. As a rule, each common genetic variant accounts for a small effect (1 mm) on individual height variation. Recently, several studies de- monstrated that some rare variants can cause greater effect on height, without causing a syndromic condition. In more extreme cases, height SDS below 2.5 or 3 (which would comprise approximately 0.6 and 0.1% of the population, respectively) is frequently associated with syndromic conditions and are usually caused by a monogenic defect. -
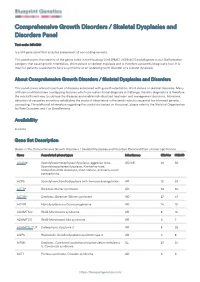
Blueprint Genetics Comprehensive Growth Disorders / Skeletal
Comprehensive Growth Disorders / Skeletal Dysplasias and Disorders Panel Test code: MA4301 Is a 374 gene panel that includes assessment of non-coding variants. This panel covers the majority of the genes listed in the Nosology 2015 (PMID: 26394607) and all genes in our Malformation category that cause growth retardation, short stature or skeletal dysplasia and is therefore a powerful diagnostic tool. It is ideal for patients suspected to have a syndromic or an isolated growth disorder or a skeletal dysplasia. About Comprehensive Growth Disorders / Skeletal Dysplasias and Disorders This panel covers a broad spectrum of diseases associated with growth retardation, short stature or skeletal dysplasia. Many of these conditions have overlapping features which can make clinical diagnosis a challenge. Genetic diagnostics is therefore the most efficient way to subtype the diseases and enable individualized treatment and management decisions. Moreover, detection of causative mutations establishes the mode of inheritance in the family which is essential for informed genetic counseling. For additional information regarding the conditions tested on this panel, please refer to the National Organization for Rare Disorders and / or GeneReviews. Availability 4 weeks Gene Set Description Genes in the Comprehensive Growth Disorders / Skeletal Dysplasias and Disorders Panel and their clinical significance Gene Associated phenotypes Inheritance ClinVar HGMD ACAN# Spondyloepimetaphyseal dysplasia, aggrecan type, AD/AR 20 56 Spondyloepiphyseal dysplasia, Kimberley -

Supplementary Information
Supplementary Information Structural Capacitance in Protein Evolution and Human Diseases Chen Li, Liah V T Clark, Rory Zhang, Benjamin T Porebski, Julia M. McCoey, Natalie A. Borg, Geoffrey I. Webb, Itamar Kass, Malcolm Buckle, Jiangning Song, Adrian Woolfson, and Ashley M. Buckle Supplementary tables Table S1. Disorder prediction using the human disease and polymorphisms dataseta OR DR OO OD DD DO mutations mutations 24,758 650 2,741 513 Disease 25,408 3,254 97.44% 2.56% 84.23% 15.77% 26,559 809 11,135 1,218 Non-disease 27,368 12,353 97.04% 2.96% 90.14% 9.86% ahttp://www.uniprot.org/docs/humsavar [1] (see Materials and Methdos). The numbers listed are the ones of unique mutations. ‘Unclassifiied’ mutations, according to the UniProt, were not counted. O = predicted as ordered; OR = Ordered regions D = predicted as disordered; DR = Disordered regions 1 Table S2. Mutations in long disordered regions (LDRs) of human proteins predicted to produce a DO transitiona Average # disorder # disorder # disorder # order UniProt/dbSNP Protein Mutation Disease length of predictors predictors predictorsb predictorsc LDRd in D2P2e for LDRf UHRF1-binding protein 1- A0JNW5/rs7296162 like S1147L - 4 2^ 101 6 3 A4D1E1/rs801841 Zinc finger protein 804B V1195I - 3* 2^ 37 6 1 A6NJV1/rs2272466 UPF0573 protein C2orf70 Q177L - 2* 4 34 3 1 Golgin subfamily A member A7E2F4/rs347880 8A K480N - 2* 2^ 91 N/A 2 Axonemal dynein light O14645/rs11749 intermediate polypeptide 1 A65V - 3* 3 43 N/A 2 Centrosomal protein of 290 O15078/rs374852145 kDa R2210C - 2 3 123 5 1 Fanconi -

Soonerstart Automatic Qualifying Syndromes and Conditions
SoonerStart Automatic Qualifying Syndromes and Conditions - Appendix O Abetalipoproteinemia Acanthocytosis (see Abetalipoproteinemia) Accutane, Fetal Effects of (see Fetal Retinoid Syndrome) Acidemia, 2-Oxoglutaric Acidemia, Glutaric I Acidemia, Isovaleric Acidemia, Methylmalonic Acidemia, Propionic Aciduria, 3-Methylglutaconic Type II Aciduria, Argininosuccinic Acoustic-Cervico-Oculo Syndrome (see Cervico-Oculo-Acoustic Syndrome) Acrocephalopolysyndactyly Type II Acrocephalosyndactyly Type I Acrodysostosis Acrofacial Dysostosis, Nager Type Adams-Oliver Syndrome (see Limb and Scalp Defects, Adams-Oliver Type) Adrenoleukodystrophy, Neonatal (see Cerebro-Hepato-Renal Syndrome) Aglossia Congenita (see Hypoglossia-Hypodactylia) Aicardi Syndrome AIDS Infection (see Fetal Acquired Immune Deficiency Syndrome) Alaninuria (see Pyruvate Dehydrogenase Deficiency) Albers-Schonberg Disease (see Osteopetrosis, Malignant Recessive) Albinism, Ocular (includes Autosomal Recessive Type) Albinism, Oculocutaneous, Brown Type (Type IV) Albinism, Oculocutaneous, Tyrosinase Negative (Type IA) Albinism, Oculocutaneous, Tyrosinase Positive (Type II) Albinism, Oculocutaneous, Yellow Mutant (Type IB) Albinism-Black Locks-Deafness Albright Hereditary Osteodystrophy (see Parathyroid Hormone Resistance) Alexander Disease Alopecia - Mental Retardation Alpers Disease Alpha 1,4 - Glucosidase Deficiency (see Glycogenosis, Type IIA) Alpha-L-Fucosidase Deficiency (see Fucosidosis) Alport Syndrome (see Nephritis-Deafness, Hereditary Type) Amaurosis (see Blindness) Amaurosis -

Clinical and Etiological Profile of Thrombocytopenia in Adults
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by UKM Journal Article Repository Research Article Clinical and etiological profile of thrombocytopenia in adults: A tertiary-care hospital-based cross-sectional study Shruti K Bhalara, Smita Shah, Hansa Goswami, RN Gonsai Department of Pathology, B.J. Medical College, Ahmedabad, Gujarat, India. Correspondence to: Shruti K Bhalara, E-mail: [email protected] Received September 6, 2014. Accepted September 13, 2014 Abstract Background: The etiologies of thrombocytopenia are diverse. Various studies on thrombocytopenia are done in the past have related to specific etiologies. Objectives: This study attempts to determine the common etiologies of thrombocytopenia in adult patients admitted to Civil Hospital, Ahmedabad. Material and Methods: A cross-sectional study was carried out at Civil Hospital, Ahmedabad. Patients with thrombo- cytopenia more than 18 years of age at admission between 1 October and 31 October 2013 were followed up during their stay in hospital, diagnosis were made, and bleeding manifestations and requirement of platelet transfusion were recorded. Results: From the 412 patients studied, dengue was diagnosed in 28.6% of patients followed by malaria in 22.8%, chronic liver disease in 15.2%, hypersplenism in 12.3%, septicemia in 6.3%, gestational thrombocytopenia and disseminated intravascular coagulation in 5.5%, immune thrombocytopenic purpura (ITP) in 3.1%, megaloblastic anemia in 1.9%, human immunodeficiency virus in 1.4%, drug-induced thrombocytopenia in 1.2%, leukemia in 0.7%, and aplastic anemia in 0.48%. Bleeding secondary to thrombocytopenia was seen in 46 (11.2%) patients; of them, 28 were diagnosed with dengue fever, 4 with chronic liver disease, 3 with sepsis, 2 with hematological malignancies, and 9 with ITP. -

A New Locus for Seckel Syndrome on Chromosome 18P11.31-Q11.2
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by University of Southern Denmark Research Output European Journal of Human Genetics (2001) 9, 753 ± 757 ã 2001 Nature Publishing Group All rights reserved 1018-4813/01 $15.00 www.nature.com/ejhg ARTICLE A new locus for Seckel syndrome on chromosome 18p11.31-q11.2 Anders D Bùrglum*,1, Thomas Balslev2, Annette Haagerup1, Niels Birkebñk2, Helle Binderup1, Torben A Kruse3 and Jens Michael Hertz4 1Institute of Human Genetics, University of Aarhus, Denmark; 2Department of Paediatrics, Aarhus University Hospital, Denmark; 3Department of Clinical Biochemistry and Genetics, KKA, Odense University Hospital, Denmark; 4Department of Clinical Genetics, Aarhus University Hospital, Denmark Seckel syndrome (MIM 210600) is a rare autosomal recessive disorder with a heterogeneous appearance. Key features are growth retardation, microcephaly with mental retardation, and a characteristic `bird-headed' facial appearance. We have performed a genome-wide linkage scan in a consanguineous family of Iraqi descent. By homozygosity mapping a new locus for the syndrome was assigned to a *30 cM interval between markers D18S78 and D18S866 with a maximum multipoint lod score of 3.1, corresponding to a trans-centromeric region on chromosome 18p11.31-q11.2. This second locus for Seckel syndrome demonstrates genetic heterogeneity and brings us a step further towards molecular genetic delineation of this heterogeneous condition. European Journal of Human Genetics (2001) 9, 753 ± 757. Keywords: Seckel syndrome; homozygosity mapping; genome scan; linkage analysis Introduction neous family from Iraq with four siblings fulfilling the Seckel syndrome (MIM 210600, http://www.ncbi.nlm.nih. -

Complications of Laparoscopic Splenectomy
ORIGINAL ARTICLE Complications of Laparoscopic Splenectomy Eduardo M. Targarona, MD, PhD; Juan Jose´ Espert, MD; Ernest Bombuy, MD; Oscar Vidal, MD; Gemma Cerda´n, MD; Vicente Artigas, MD, PhD; Manuel Trı´as, MD, PhD Hypothesis: Analysis of the type and characteristics of Results: One hundred thirteen laparoscopic splenectomies complications after laparoscopic splenectomy may per- were completed (conversion rate, 7.4%). Twenty patients mit the identification of clinical factors with predictive (18%) developed 23 complications. All were Clavien type value for the development of complications. I or II, without mortality. One complication was intraop- erative (diaphragmatic perforation), and 22 were postop- Design: Univariate and multivariate analysis of factors erative: 6 pulmonary (26%), 3 fever (13%), 8 hemorrhagic related to complications in a prospective series of lapa- (35%) (5 episodes of postoperative bleeding and 3 abdomi- roscopic splenectomies. nal wall hematomas), and 6 others (26%). Ten (43%) of the 23 were technically related. Univariate analysis showed Setting: A large tertiary referral university–teaching gen- that complications were only related to age (mean±SD, eral hospital. 55±15 vs 39±17 years; P,.008) or transfusion (50% vs 11%; P,.001). Multivariate analysis showed that the learn- Patients: One hundred twenty-two nonselected con- ing curve (P,.005; 95% confidence interval, 2.46), age secutive patients, in whom laparoscopic splenectomy was (P,.001; 95% confidence interval, 1.04), spleen weight attempted between February 1993 and July 1999. (P,.009; 95% confidence interval, 1.00), and malignant neoplasmdiagnosis(P,.007;95%confidenceinterval,3.82) Intervention: Laparoscopic splenectomy. were independent predictors of complications. Main Outcome Measures: Immediate complica- Conclusions: Laparoscopic splenectomy is feasible, and tions classified according to the Clavien score. -

CASE REPORT Sensorineural Hearing Loss and Tinnitus As Presenting Symptoms of Polycythemia Vera
Int. Adv. Otol. 2012; 8:(2) 317-320 CASE REPORT Sensorineural Hearing Loss and Tinnitus as Presenting Symptoms of Polycythemia Vera Ki-Hong Chang, Eun-Ju Jeon Department of Otolaryngology-HNS, The Catholic University of Korea, Seoul, Korea (KHC, EJ) Abstract: Polycythemia vera (PV) is a blood disorder in which the bone marrow makes too many red blood cells. Commonly encountered neurologic symptoms include transient ischemic attack, dementia, dizziness, tinnitus, and visual disturbances which are thought to result from thrombosis and hemorrhage. Although hearing impairment is one of the neurologic symptoms of PV, tinnitus or hearing impairment as a presenting symptom of PV is very rare. We report a patient with polycythemia vera whose first sign and symptom were bilateral tinnitus and hearing loss. Unfortunately, the hearing was not recovered after treatment including aspirin and phlebotomy while blood level returned to near normal range. Submitted : 16 June 2011 Accepted : 7 November 2011 Introduction hearing loss and tinnitus had developed in his right ear. Polycythemia vera (PV) is characterized by an At that time, he visited another hospital for the hearing erythrocytosis variably associated with leukocytosis loss and was treated with some medication on the and thrombocytosis. PV is a rare disease with an impression of idiopathic sudden hearing loss. However, incidence rate of approximately 1.9-2.3 new cases per as his hearing was not recovered and his tinnitus was 100,000/year in Europe and the United States. The aggravated, he quit taking the medication. After 6 incidence of PV is slightly higher in men than women months, tinnitus gradually developed in his left ear and (2.8 vs. -

Early ACCESS Diagnosed Conditions List
Iowa Early ACCESS Diagnosed Conditions Eligibility List List adapted with permission from Early Intervention Colorado To search for a specific word type "Ctrl F" to use the "Find" function. Is this diagnosis automatically eligible for Early Medical Diagnosis Name Other Names for the Diagnosis and Additional Diagnosis Information ACCESS? 6q terminal deletion syndrome Yes Achondrogenesis I Parenti-Fraccaro Yes Achondrogenesis II Langer-Saldino Yes Schinzel Acrocallosal syndrome; ACLS; ACS; Hallux duplication, postaxial polydactyly, and absence of the corpus Acrocallosal syndrome, Schinzel Type callosum Yes Acrodysplasia; Arkless-Graham syndrome; Maroteaux-Malamut syndrome; Nasal hypoplasia-peripheral dysostosis-intellectual disability syndrome; Peripheral dysostosis-nasal hypoplasia-intellectual disability (PNM) Acrodysostosis syndrome Yes ALD; AMN; X-ALD; Addison disease and cerebral sclerosis; Adrenomyeloneuropathy; Siemerling-creutzfeldt disease; Bronze schilder disease; Schilder disease; Melanodermic Leukodystrophy; sudanophilic leukodystrophy; Adrenoleukodystrophy Pelizaeus-Merzbacher disease Yes Agenesis of Corpus Callosum Absence of the corpus callosum; Hypogenesis of the corpus callosum; Dysplastic corpus callosum Yes Agenesis of Corpus Callosum and Chorioretinal Abnormality; Agenesis of Corpus Callosum With Chorioretinitis Abnormality; Agenesis of Corpus Callosum With Infantile Spasms And Ocular Anomalies; Chorioretinal Anomalies Aicardi syndrome with Agenesis Yes Alexander Disease Yes Allan Herndon syndrome Allan-Herndon-Dudley