New Insights Into the Earlier Evolutionary History of Epiphytic
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The 2014 Golden Gate National Parks Bioblitz - Data Management and the Event Species List Achieving a Quality Dataset from a Large Scale Event
National Park Service U.S. Department of the Interior Natural Resource Stewardship and Science The 2014 Golden Gate National Parks BioBlitz - Data Management and the Event Species List Achieving a Quality Dataset from a Large Scale Event Natural Resource Report NPS/GOGA/NRR—2016/1147 ON THIS PAGE Photograph of BioBlitz participants conducting data entry into iNaturalist. Photograph courtesy of the National Park Service. ON THE COVER Photograph of BioBlitz participants collecting aquatic species data in the Presidio of San Francisco. Photograph courtesy of National Park Service. The 2014 Golden Gate National Parks BioBlitz - Data Management and the Event Species List Achieving a Quality Dataset from a Large Scale Event Natural Resource Report NPS/GOGA/NRR—2016/1147 Elizabeth Edson1, Michelle O’Herron1, Alison Forrestel2, Daniel George3 1Golden Gate Parks Conservancy Building 201 Fort Mason San Francisco, CA 94129 2National Park Service. Golden Gate National Recreation Area Fort Cronkhite, Bldg. 1061 Sausalito, CA 94965 3National Park Service. San Francisco Bay Area Network Inventory & Monitoring Program Manager Fort Cronkhite, Bldg. 1063 Sausalito, CA 94965 March 2016 U.S. Department of the Interior National Park Service Natural Resource Stewardship and Science Fort Collins, Colorado The National Park Service, Natural Resource Stewardship and Science office in Fort Collins, Colorado, publishes a range of reports that address natural resource topics. These reports are of interest and applicability to a broad audience in the National Park Service and others in natural resource management, including scientists, conservation and environmental constituencies, and the public. The Natural Resource Report Series is used to disseminate comprehensive information and analysis about natural resources and related topics concerning lands managed by the National Park Service. -

Icmadophila Aversa and Piccolia Conspersa, Two Lichen Species New to Bolivia
Polish Botanical Journal 55(1): 217–221, 2010 ICMADOPHILA AVERSA AND PICCOLIA CONSPERSA, TWO LICHEN SPECIES NEW TO BOLIVIA KARINA WILK Abstract. The species Icmadophila aversa and Piccolia conspersa are reported as new to the lichen biota of Bolivia. The studied material was collected in Madidi National Park (NW Bolivia). The species are briefl y characterized and their ecology and distribution are discussed. Key words: lichenized fungi, new records, Madidi region, Andes, South America Karina Wilk, Laboratory of Lichenology, W. Szafer Institute of Botany, Polish Academy of Sciences, Lubicz 46, 31-512 Kraków, Poland; e-mail: [email protected] INTRODUCTION Bolivia is still one of the countries least studied While studying the material collected in the biologically, but the data already available indi- Madidi region I identifi ed two interesting lichen cate a potentially high level of biodiversity (Ibisch species – Icmadophila aversa and Piccolia con- & Mérida 2004). Knowledge of the cryptogams, spersa. The species are reported here as new to Bo- including lichens, is particularly defi cient (Feuerer livia. Brief descriptions and notes on their ecology et al. 1998). In the last decade, however, licheno- and worldwide distribution are provided. logical studies have progressed in Bolivia. The most recent works have provided many new dis- MATERIAL AND METHODS coveries: records new to the country, continent or Southern Hemisphere, and species new to The study is based on material collected in 2006–2007 in science (e.g., Ferraro 2002; Feuerer & Sipman Madidi National Park. The collection sites are located in 2005; Flakus & Wilk 2006; Flakus & Kukwa 2007; the Cordillera Apolobamba (Fig. -

The Puzzle of Lichen Symbiosis
Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 1503 The puzzle of lichen symbiosis Pieces from Thamnolia IOANA ONUT, -BRÄNNSTRÖM ACTA UNIVERSITATIS UPSALIENSIS ISSN 1651-6214 ISBN 978-91-554-9887-0 UPPSALA urn:nbn:se:uu:diva-319639 2017 Dissertation presented at Uppsala University to be publicly examined in Lindhalsalen, EBC, Norbyvägen 14, Uppsala, Thursday, 1 June 2017 at 09:15 for the degree of Doctor of Philosophy. The examination will be conducted in English. Faculty examiner: Associate Professor Anne Pringle (University of Wisconsin-Madison, Department of Botany). Abstract Onuț-Brännström, I. 2017. The puzzle of lichen symbiosis. Pieces from Thamnolia. Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 1503. 62 pp. Uppsala: Acta Universitatis Upsaliensis. ISBN 978-91-554-9887-0. Symbiosis brought important evolutionary novelties to life on Earth. Lichens, the symbiotic entities formed by fungi, photosynthetic organisms and bacteria, represent an example of a successful adaptation in surviving hostile environments. Yet many aspects of the lichen symbiosis remain unexplored. This thesis aims at bringing insights into lichen biology and the importance of symbiosis in adaptation. I am using as model system a successful colonizer of tundra and alpine environments, the worm lichens Thamnolia, which seem to only reproduce vegetatively through symbiotic propagules. When the genetic architecture of the mating locus of the symbiotic fungal partner was analyzed with genomic and transcriptomic data, a sexual self-incompatible life style was revealed. However, a screen of the mating types ratios across natural populations detected only one of the mating types, suggesting that Thamnolia has no potential for sexual reproduction because of lack of mating partners. -

Phytochemical Studies of Endolichenic Fungi Isolated from Hypotrachyna Infirma (Kurok.) Hale
International Journal of Pharmacy and Pharmaceutical Sciences Print ISSN: 2656-0097 | Online ISSN: 0975-1491 Vol 13, Issue 8, 2021 Original Article PHYTOCHEMICAL STUDIES OF ENDOLICHENIC FUNGI ISOLATED FROM HYPOTRACHYNA INFIRMA (KUROK.) HALE GOKILAVANI R.1, H. REHANA BANU2 1,2Department of Botany, PSGR Krishnammal College for Women, Peelamedu, Coimbatore 641004, Tamil Nadu, India Email: [email protected] Received: 08 Apr 2021, Revised and Accepted: 19 Jun 2021 ABSTRACT Objective: The aim of this study is to investigate of phytopharmaceutical importance of endolichenic fungi isolated from Hypotrachyna infirma (Kurok.) Hale. Methods: The lichen species were collected from Sholaiyar hills, Coimbatore and identified as Hypotrachyna infirma (Kurok.)Hale. From this lichen, 29 endolichenic fungi were isolated and 13 endolichenic fungi were identified. From the identified endolichenic fungi, 26 extracts were prepared by successive solvent extraction methods using Ethyl acetate and chloroform. Results: The phytochemical study revealed the presence of important constituents like Alkaloids, Tannins, Carbohydrates, Phenols, Protein, Terpenoids, Steroids, Glycosides Flavonoids and Saponins. From the 13 endolichenic fungi, only 5 endolichenic fungi (Nigrospora oryzae (Berkand Broome)Petch, Geotrichum candidum Link, Scytalidium lignicola pesante, Aspergillus oryzae(Ahlb.) cohn, Aspergillus niger Gr.) have more constitutents. These 5 endolichenic fungi have good results in Quantitative analysis also. Conclusion: Compared to ethyl acetate extracts Chloroform extracts showed very less concentration of the phytochemicals. From this study we conclutated Nigrospora oryzae (Berk and Broome) Petch gave the best results in both qualitative and quantitative compared to other endolichenic fungi. Keywords: Hypotrachyna infirma, Endolichenic fungus, Ethyl acetate, Chloroform and phytochemical © 2021 The Authors. Published by Innovare Academic Sciences Pvt Ltd. -

Leightoniella Zeylanensis Belongs to the Pannariaceae
doi: 10.1111/njb.01880 00 1–6 NORDIC JOURNAL OF BOTANY Research Leightoniella zeylanensis belongs to the Pannariaceae Gothamie Weerakoon, André Aptroot, Mats Wedin and Stefan Ekman G. Weerakoon (https://orcid.org/0000-0002-9550-2139), Algae, Fungi and Plant Division, Dept of Life Sciences, The Natural History Museum, Cromwell Road, London SW7 5BD, United Kingdom. – A. Aptroot (https://orcid.org/0000-0001-7949-2594), ABL Herbarium, XK Soest, the Netherlands. – M. Wedin (https://orcid.org/0000-0002-8295-5198), Swedish Museum of Natural History, Dept of Botany, Stockholm, Sweden. – S. Ekman (https://orcid. org/0000-0003-3021-1821) ([email protected]), Museum of Evolution, Uppsala Univ., Uppsala, Sweden. Nordic Journal of Botany Recent finds ofLeightoniella zeylanensis, classified variously in the Collemataceae and 2018: e01880 Pannariaceae, enabled us to generate DNA sequence data for investigating its phy- doi: 10.1111/njb.01880 logenetic affiliation. Newly generated sequence data from the internal transcribed spacer (ITS) region and the large subunit of the nuclear ribosomal DNA (nrLSU), Subject Editor and the small subunit of the mitochondrial ribosomal (mrSSU) DNA, and the largest sub- Editor-in-Chief: Torbjörn Tyler unit of the RNA polymerase II gene (RPB1) indicate that L. zeylanensis is a member Accepted 1 June 2018 of the Pannariaceae, belonging to a strongly supported clade together with Physma, Lepidocollema, and Gibbosporina (= the ‘Physma clade’). With the currently available data, however, relationships within this clade are largely impossible to reconstruct with confidence. Leightoniella zeylanensis was found to possess ellipsoid ascospores surrounded by a thick, gelatinous perispore with pointed ends, supporting a previously published hypothesis that such a perispore type is a synapomorphy for the Physma clade. -
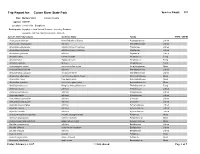
Cuivre Bryophytes
Trip Report for: Cuivre River State Park Species Count: 335 Date: Multiple Visits Lincoln County Agency: MODNR Location: Lincoln Hills - Bryophytes Participants: Bryophytes from Natural Resource Inventory Database Bryophyte List from NRIDS and Bruce Schuette Species Name (Synonym) Common Name Family COFC COFW Acarospora unknown Identified only to Genus Acarosporaceae Lichen Acrocordia megalospora a lichen Monoblastiaceae Lichen Amandinea dakotensis a button lichen (crustose) Physiaceae Lichen Amandinea polyspora a button lichen (crustose) Physiaceae Lichen Amandinea punctata a lichen Physiaceae Lichen Amanita citrina Citron Amanita Amanitaceae Fungi Amanita fulva Tawny Gresette Amanitaceae Fungi Amanita vaginata Grisette Amanitaceae Fungi Amblystegium varium common willow moss Amblystegiaceae Moss Anisomeridium biforme a lichen Monoblastiaceae Lichen Anisomeridium polypori a crustose lichen Monoblastiaceae Lichen Anomodon attenuatus common tree apron moss Anomodontaceae Moss Anomodon minor tree apron moss Anomodontaceae Moss Anomodon rostratus velvet tree apron moss Anomodontaceae Moss Armillaria tabescens Ringless Honey Mushroom Tricholomataceae Fungi Arthonia caesia a lichen Arthoniaceae Lichen Arthonia punctiformis a lichen Arthoniaceae Lichen Arthonia rubella a lichen Arthoniaceae Lichen Arthothelium spectabile a lichen Uncertain Lichen Arthothelium taediosum a lichen Uncertain Lichen Aspicilia caesiocinerea a lichen Hymeneliaceae Lichen Aspicilia cinerea a lichen Hymeneliaceae Lichen Aspicilia contorta a lichen Hymeneliaceae Lichen -

Global Biodiversity Patterns of the Photobionts Associated with the Genus Cladonia (Lecanorales, Ascomycota)
Microbial Ecology https://doi.org/10.1007/s00248-020-01633-3 FUNGAL MICROBIOLOGY Global Biodiversity Patterns of the Photobionts Associated with the Genus Cladonia (Lecanorales, Ascomycota) Raquel Pino-Bodas1 & Soili Stenroos2 Received: 19 August 2020 /Accepted: 22 October 2020 # The Author(s) 2020 Abstract The diversity of lichen photobionts is not fully known. We studied here the diversity of the photobionts associated with Cladonia, a sub-cosmopolitan genus ecologically important, whose photobionts belong to the green algae genus Asterochloris. The genetic diversity of Asterochloris was screened by using the ITS rDNA and actin type I regions in 223 specimens and 135 species of Cladonia collected all over the world. These data, added to those available in GenBank, were compiled in a dataset of altogether 545 Asterochloris sequences occurring in 172 species of Cladonia. A high diversity of Asterochloris associated with Cladonia was found. The commonest photobiont lineages associated with this genus are A. glomerata, A. italiana,andA. mediterranea. Analyses of partitioned variation were carried out in order to elucidate the relative influence on the photobiont genetic variation of the following factors: mycobiont identity, geographic distribution, climate, and mycobiont phylogeny. The mycobiont identity and climate were found to be the main drivers for the genetic variation of Asterochloris. The geographical distribution of the different Asterochloris lineages was described. Some lineages showed a clear dominance in one or several climatic regions. In addition, the specificity and the selectivity were studied for 18 species of Cladonia. Potentially specialist and generalist species of Cladonia were identified. A correlation was found between the sexual reproduction frequency of the host and the frequency of certain Asterochloris OTUs. -

Aportes Al Conocimiento De La Biota Liquénica Del Oasis De Neblina De Alto Patache, Desierto De Atacama1
Revista de Geografía Norte Grande, 68: 49-64 (2017) Artículos Aportes al conocimiento de la biota liquénica del oasis de neblina de Alto Patache, Desierto de Atacama1 Reinaldo Vargas Castillo2, Daniel Stanton3 y Peter R. Nelson4 RESUMEN Los denominados oasis de neblina son áreas en las zonas costeras del Desierto de Ataca- ma donde el ingreso habitual de niebla permite el establecimiento y desarrollo de diver- sas poblaciones de plantas vasculares, generando verdaderos hotspots de diversidad. En estas áreas, la biota liquenológica ha sido poco explorada y representa uno de los ele- mentos perennes más importantes que conforman la comunidad. En un estudio previo de la biota del oasis de neblina de Alto Patache se reportaron siete especies. Con el fin de mejorar este conocimiento, se analizó la riqueza de especies presentes en el oasis si- guiendo dos transectos altitudinales en diferentes orientaciones del farellón. Aquí repor- tamos preliminarmente 77 especies de líquenes para el oasis de neblina de Alto Patache. De estas, 61 especies corresponden a nuevos registros para la región de Tarapacá, en tanto que las especies Amandinea eff lorescens, Diploicia canescens, Myriospora smarag- dula y Rhizocarpon simillimum corresponden a nuevos registros para el país. Asimismo, se destaca a Alto Patache como la única localidad conocida para Santessonia cervicornis, una especie endémica y en Peligro Crítico. Palabras clave: Oasis de neblina, Desierto de Atacama, líquenes. ABSTRACT Fog oases are zones along the Atacama Desert where the regular input of fog favors the development of rich communities of vascular plants, becoming biodiversity hotspots. In these areas, the lichen biota has been poorly explored and represents one of the most conspicuous elements among the perennials organisms that form the community. -

Gymnoderma Insulare
The IUCN Red List of Threatened Species™ ISSN 2307-8235 (online) IUCN 2008: T58520980A58520984 Gymnoderma insulare Assessment by: Ohmura, Y., Nadyeina, O. & Scheidegger, C. View on www.iucnredlist.org Citation: Ohmura, Y., Nadyeina, O. & Scheidegger, C. 2014. Gymnoderma insulare. The IUCN Red List of Threatened Species 2014: e.T58520980A58520984. http://dx.doi.org/10.2305/IUCN.UK.2014- 3.RLTS.T58520980A58520984.en Copyright: © 2015 International Union for Conservation of Nature and Natural Resources Reproduction of this publication for educational or other non-commercial purposes is authorized without prior written permission from the copyright holder provided the source is fully acknowledged. Reproduction of this publication for resale, reposting or other commercial purposes is prohibited without prior written permission from the copyright holder. For further details see Terms of Use. The IUCN Red List of Threatened Species™ is produced and managed by the IUCN Global Species Programme, the IUCN Species Survival Commission (SSC) and The IUCN Red List Partnership. The IUCN Red List Partners are: BirdLife International; Botanic Gardens Conservation International; Conservation International; Microsoft; NatureServe; Royal Botanic Gardens, Kew; Sapienza University of Rome; Texas A&M University; Wildscreen; and Zoological Society of London. If you see any errors or have any questions or suggestions on what is shown in this document, please provide us with feedback so that we can correct or extend the information provided. THE IUCN RED LIST OF THREATENED SPECIES™ Taxonomy Kingdom Phylum Class Order Family Fungi Ascomycota Lecanoromycetes Lecanorales Cladoniaceae Taxon Name: Gymnoderma insulare Yoshim. & Sharp Taxonomic Notes: This attractive macrolichen is easy to recognize by lichen specialists. -

1307 Fungi Representing 1139 Infrageneric Taxa, 317 Genera and 66 Families ⇑ Jolanta Miadlikowska A, , Frank Kauff B,1, Filip Högnabba C, Jeffrey C
Molecular Phylogenetics and Evolution 79 (2014) 132–168 Contents lists available at ScienceDirect Molecular Phylogenetics and Evolution journal homepage: www.elsevier.com/locate/ympev A multigene phylogenetic synthesis for the class Lecanoromycetes (Ascomycota): 1307 fungi representing 1139 infrageneric taxa, 317 genera and 66 families ⇑ Jolanta Miadlikowska a, , Frank Kauff b,1, Filip Högnabba c, Jeffrey C. Oliver d,2, Katalin Molnár a,3, Emily Fraker a,4, Ester Gaya a,5, Josef Hafellner e, Valérie Hofstetter a,6, Cécile Gueidan a,7, Mónica A.G. Otálora a,8, Brendan Hodkinson a,9, Martin Kukwa f, Robert Lücking g, Curtis Björk h, Harrie J.M. Sipman i, Ana Rosa Burgaz j, Arne Thell k, Alfredo Passo l, Leena Myllys c, Trevor Goward h, Samantha Fernández-Brime m, Geir Hestmark n, James Lendemer o, H. Thorsten Lumbsch g, Michaela Schmull p, Conrad L. Schoch q, Emmanuël Sérusiaux r, David R. Maddison s, A. Elizabeth Arnold t, François Lutzoni a,10, Soili Stenroos c,10 a Department of Biology, Duke University, Durham, NC 27708-0338, USA b FB Biologie, Molecular Phylogenetics, 13/276, TU Kaiserslautern, Postfach 3049, 67653 Kaiserslautern, Germany c Botanical Museum, Finnish Museum of Natural History, FI-00014 University of Helsinki, Finland d Department of Ecology and Evolutionary Biology, Yale University, 358 ESC, 21 Sachem Street, New Haven, CT 06511, USA e Institut für Botanik, Karl-Franzens-Universität, Holteigasse 6, A-8010 Graz, Austria f Department of Plant Taxonomy and Nature Conservation, University of Gdan´sk, ul. Wita Stwosza 59, 80-308 Gdan´sk, Poland g Science and Education, The Field Museum, 1400 S. -

Three Challenges to Contemporaneous Taxonomy from a Licheno-Mycological Perspective
Megataxa 001 (1): 078–103 ISSN 2703-3082 (print edition) https://www.mapress.com/j/mt/ MEGATAXA Copyright © 2020 Magnolia Press Review ISSN 2703-3090 (online edition) https://doi.org/10.11646/megataxa.1.1.16 Three challenges to contemporaneous taxonomy from a licheno-mycological perspective ROBERT LÜCKING Botanischer Garten und Botanisches Museum, Freie Universität Berlin, Königin-Luise-Straße 6–8, 14195 Berlin, Germany �[email protected]; https://orcid.org/0000-0002-3431-4636 Abstract Nagoya Protocol, and does not need additional “policing”. Indeed, the Nagoya Protocol puts the heaviest burden on This paper discusses three issues that challenge contempora- taxonomy and researchers cataloguing biodiversity, whereas neous taxonomy, with examples from the fields of mycology for the intended target group, namely those seeking revenue and lichenology, formulated as three questions: (1) What is gain from nature, the protocol may not actually work effec- the importance of taxonomy in contemporaneous and future tively. The notion of currently freely accessible digital se- science and society? (2) An increasing methodological gap in quence information (DSI) to become subject to the protocol, alpha taxonomy: challenge or opportunity? (3) The Nagoya even after previous publication, is misguided and conflicts Protocol: improvement or impediment to the science of tax- with the guidelines for ethical scientific conduct. Through onomy? The importance of taxonomy in society is illustrated its implementation of the Nagoya Protocol, Colombia has using the example of popular field guides and digital me- set a welcome precedence how to exempt taxonomic and dia, a billion-dollar business, arguing that the desire to name systematic research from “access to genetic resources”, and species is an intrinsic feature of the cognitive component of hopefully other biodiversity-rich countries will follow this nature connectedness of humans. -

An Evolving Phylogenetically Based Taxonomy of Lichens and Allied Fungi
Opuscula Philolichenum, 11: 4-10. 2012. *pdf available online 3January2012 via (http://sweetgum.nybg.org/philolichenum/) An evolving phylogenetically based taxonomy of lichens and allied fungi 1 BRENDAN P. HODKINSON ABSTRACT. – A taxonomic scheme for lichens and allied fungi that synthesizes scientific knowledge from a variety of sources is presented. The system put forth here is intended both (1) to provide a skeletal outline of the lichens and allied fungi that can be used as a provisional filing and databasing scheme by lichen herbarium/data managers and (2) to announce the online presence of an official taxonomy that will define the scope of the newly formed International Committee for the Nomenclature of Lichens and Allied Fungi (ICNLAF). The online version of the taxonomy presented here will continue to evolve along with our understanding of the organisms. Additionally, the subfamily Fissurinoideae Rivas Plata, Lücking and Lumbsch is elevated to the rank of family as Fissurinaceae. KEYWORDS. – higher-level taxonomy, lichen-forming fungi, lichenized fungi, phylogeny INTRODUCTION Traditionally, lichen herbaria have been arranged alphabetically, a scheme that stands in stark contrast to the phylogenetic scheme used by nearly all vascular plant herbaria. The justification typically given for this practice is that lichen taxonomy is too unstable to establish a reasonable system of classification. However, recent leaps forward in our understanding of the higher-level classification of fungi, driven primarily by the NSF-funded Assembling the Fungal Tree of Life (AFToL) project (Lutzoni et al. 2004), have caused the taxonomy of lichen-forming and allied fungi to increase significantly in stability. This is especially true within the class Lecanoromycetes, the main group of lichen-forming fungi (Miadlikowska et al.