Chapter 3 Radioactivity
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
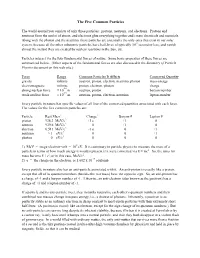
The Five Common Particles
The Five Common Particles The world around you consists of only three particles: protons, neutrons, and electrons. Protons and neutrons form the nuclei of atoms, and electrons glue everything together and create chemicals and materials. Along with the photon and the neutrino, these particles are essentially the only ones that exist in our solar system, because all the other subatomic particles have half-lives of typically 10-9 second or less, and vanish almost the instant they are created by nuclear reactions in the Sun, etc. Particles interact via the four fundamental forces of nature. Some basic properties of these forces are summarized below. (Other aspects of the fundamental forces are also discussed in the Summary of Particle Physics document on this web site.) Force Range Common Particles It Affects Conserved Quantity gravity infinite neutron, proton, electron, neutrino, photon mass-energy electromagnetic infinite proton, electron, photon charge -14 strong nuclear force ≈ 10 m neutron, proton baryon number -15 weak nuclear force ≈ 10 m neutron, proton, electron, neutrino lepton number Every particle in nature has specific values of all four of the conserved quantities associated with each force. The values for the five common particles are: Particle Rest Mass1 Charge2 Baryon # Lepton # proton 938.3 MeV/c2 +1 e +1 0 neutron 939.6 MeV/c2 0 +1 0 electron 0.511 MeV/c2 -1 e 0 +1 neutrino ≈ 1 eV/c2 0 0 +1 photon 0 eV/c2 0 0 0 1) MeV = mega-electron-volt = 106 eV. It is customary in particle physics to measure the mass of a particle in terms of how much energy it would represent if it were converted via E = mc2. -
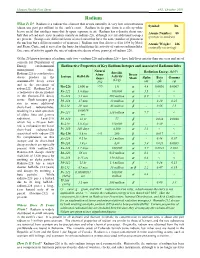
Radium What Is It? Radium Is a Radioactive Element That Occurs Naturally in Very Low Concentrations Symbol: Ra (About One Part Per Trillion) in the Earth’S Crust
Human Health Fact Sheet ANL, October 2001 Radium What Is It? Radium is a radioactive element that occurs naturally in very low concentrations Symbol: Ra (about one part per trillion) in the earth’s crust. Radium in its pure form is a silvery-white heavy metal that oxidizes immediately upon exposure to air. Radium has a density about one- Atomic Number: 88 half that of lead and exists in nature mainly as radium-226, although several additional isotopes (protons in nucleus) are present. (Isotopes are different forms of an element that have the same number of protons in the nucleus but a different number of neutrons.) Radium was first discovered in 1898 by Marie Atomic Weight: 226 and Pierre Curie, and it served as the basis for identifying the activity of various radionuclides. (naturally occurring) One curie of activity equals the rate of radioactive decay of one gram (g) of radium-226. Of the 25 known isotopes of radium, only two – radium-226 and radium-228 – have half-lives greater than one year and are of concern for Department of Energy environmental Radioactive Properties of Key Radium Isotopes and Associated Radionuclides management sites. Natural Specific Radiation Energy (MeV) Radium-226 is a radioactive Abun- Decay Isotope Half-Life Activity decay product in the dance Mode Alpha Beta Gamma (Ci/g) uranium-238 decay series (%) (α) (β) (γ) and is the precursor of Ra-226 1,600 yr >99 1.0 α 4.8 0.0036 0.0067 radon-222. Radium-228 is a radioactive decay product Rn-222 3.8 days 160,000 α 5.5 < < in the thorium-232 decay Po-218 3.1 min 290 million α 6.0 < < series. -

R-Process Elements from Magnetorotational Hypernovae
r-Process elements from magnetorotational hypernovae D. Yong1,2*, C. Kobayashi3,2, G. S. Da Costa1,2, M. S. Bessell1, A. Chiti4, A. Frebel4, K. Lind5, A. D. Mackey1,2, T. Nordlander1,2, M. Asplund6, A. R. Casey7,2, A. F. Marino8, S. J. Murphy9,1 & B. P. Schmidt1 1Research School of Astronomy & Astrophysics, Australian National University, Canberra, ACT 2611, Australia 2ARC Centre of Excellence for All Sky Astrophysics in 3 Dimensions (ASTRO 3D), Australia 3Centre for Astrophysics Research, Department of Physics, Astronomy and Mathematics, University of Hertfordshire, Hatfield, AL10 9AB, UK 4Department of Physics and Kavli Institute for Astrophysics and Space Research, Massachusetts Institute of Technology, Cambridge, MA 02139, USA 5Department of Astronomy, Stockholm University, AlbaNova University Center, 106 91 Stockholm, Sweden 6Max Planck Institute for Astrophysics, Karl-Schwarzschild-Str. 1, D-85741 Garching, Germany 7School of Physics and Astronomy, Monash University, VIC 3800, Australia 8Istituto NaZionale di Astrofisica - Osservatorio Astronomico di Arcetri, Largo Enrico Fermi, 5, 50125, Firenze, Italy 9School of Science, The University of New South Wales, Canberra, ACT 2600, Australia Neutron-star mergers were recently confirmed as sites of rapid-neutron-capture (r-process) nucleosynthesis1–3. However, in Galactic chemical evolution models, neutron-star mergers alone cannot reproduce the observed element abundance patterns of extremely metal-poor stars, which indicates the existence of other sites of r-process nucleosynthesis4–6. These sites may be investigated by studying the element abundance patterns of chemically primitive stars in the halo of the Milky Way, because these objects retain the nucleosynthetic signatures of the earliest generation of stars7–13. -

Qcd in Heavy Quark Production and Decay
QCD IN HEAVY QUARK PRODUCTION AND DECAY Jim Wiss* University of Illinois Urbana, IL 61801 ABSTRACT I discuss how QCD is used to understand the physics of heavy quark production and decay dynamics. My discussion of production dynam- ics primarily concentrates on charm photoproduction data which is compared to perturbative QCD calculations which incorporate frag- mentation effects. We begin our discussion of heavy quark decay by reviewing data on charm and beauty lifetimes. Present data on fully leptonic and semileptonic charm decay is then reviewed. Mea- surements of the hadronic weak current form factors are compared to the nonperturbative QCD-based predictions of Lattice Gauge The- ories. We next discuss polarization phenomena present in charmed baryon decay. Heavy Quark Effective Theory predicts that the daugh- ter baryon will recoil from the charmed parent with nearly 100% left- handed polarization, which is in excellent agreement with present data. We conclude by discussing nonleptonic charm decay which are tradi- tionally analyzed in a factorization framework applicable to two-body and quasi-two-body nonleptonic decays. This discussion emphasizes the important role of final state interactions in influencing both the observed decay width of various two-body final states as well as mod- ifying the interference between Interfering resonance channels which contribute to specific multibody decays. "Supported by DOE Contract DE-FG0201ER40677. © 1996 by Jim Wiss. -251- 1 Introduction the direction of fixed-target experiments. Perhaps they serve as a sort of swan song since the future of fixed-target charm experiments in the United States is A vast amount of important data on heavy quark production and decay exists for very short. -

Neutrinoless Double Beta Decay
REPORT TO THE NUCLEAR SCIENCE ADVISORY COMMITTEE Neutrinoless Double Beta Decay NOVEMBER 18, 2015 NLDBD Report November 18, 2015 EXECUTIVE SUMMARY In March 2015, DOE and NSF charged NSAC Subcommittee on neutrinoless double beta decay (NLDBD) to provide additional guidance related to the development of next generation experimentation for this field. The new charge (Appendix A) requests a status report on the existing efforts in this subfield, along with an assessment of the necessary R&D required for each candidate technology before a future downselect. The Subcommittee membership was augmented to replace several members who were not able to continue in this phase (the present Subcommittee membership is attached as Appendix B). The Subcommittee solicited additional written input from the present worldwide collaborative efforts on double beta decay projects in order to collect the information necessary to address the new charge. An open meeting was held where these collaborations were invited to present material related to their current projects, conceptual designs for next generation experiments, and the critical R&D required before a potential down-select. We also heard presentations related to nuclear theory and the impact of future cosmological data on the subject of NLDBD. The Subcommittee presented its principal findings and comments in response to the March 2015 charge at the NSAC meeting in October 2015. The March 2015 charge requested the Subcommittee to: Assess the status of ongoing R&D for NLDBD candidate technology demonstrations for a possible future ton-scale NLDBD experiment. For each candidate technology demonstration, identify the major remaining R&D tasks needed ONLY to demonstrate downselect criteria, including the sensitivity goals, outlined in the NSAC report of May 2014. -

RIB Production by Photofission in the Framework of the ALTO Project
Available online at www.sciencedirect.com NIM B Beam Interactions with Materials & Atoms Nuclear Instruments and Methods in Physics Research B 266 (2008) 4092–4096 www.elsevier.com/locate/nimb RIB production by photofission in the framework of the ALTO project: First experimental measurements and Monte-Carlo simulations M. Cheikh Mhamed *, S. Essabaa, C. Lau, M. Lebois, B. Roussie`re, M. Ducourtieux, S. Franchoo, D. Guillemaud Mueller, F. Ibrahim, J.F. LeDu, J. Lesrel, A.C. Mueller, M. Raynaud, A. Said, D. Verney, S. Wurth Institut de Physique Nucle´aire, IN2P3-CNRS/Universite´ Paris-Sud, F-91406 Orsay Cedex, France Available online 11 June 2008 Abstract The ALTO facility (Acce´le´rateur Line´aire aupre`s du Tandem d’Orsay) has been built and is now under commissioning. The facility is intended for the production of low energy neutron-rich ion-beams by ISOL technique. This will open new perspectives in the study of nuclei very far from the valley of stability. Neutron-rich nuclei are produced by photofission in a thick uranium carbide target (UCx) using a 10 lA, 50 MeV electron beam. The target is the same as that already had been used on the previous deuteron based fission ISOL setup (PARRNE [F. Clapier et al., Phys. Rev. ST-AB (1998) 013501.]). The intended nominal fission rate is about 1011 fissions/s. We have studied the adequacy of a thick carbide uranium target to produce neutron-rich nuclei by photofission by means of Monte-Carlo simulations. We present the production rates in the target and after extraction and mass separation steps. -

Inertial Electrostatic Confinement Fusor Cody Boyd Virginia Commonwealth University
Virginia Commonwealth University VCU Scholars Compass Capstone Design Expo Posters College of Engineering 2015 Inertial Electrostatic Confinement Fusor Cody Boyd Virginia Commonwealth University Brian Hortelano Virginia Commonwealth University Yonathan Kassaye Virginia Commonwealth University See next page for additional authors Follow this and additional works at: https://scholarscompass.vcu.edu/capstone Part of the Engineering Commons © The Author(s) Downloaded from https://scholarscompass.vcu.edu/capstone/40 This Poster is brought to you for free and open access by the College of Engineering at VCU Scholars Compass. It has been accepted for inclusion in Capstone Design Expo Posters by an authorized administrator of VCU Scholars Compass. For more information, please contact [email protected]. Authors Cody Boyd, Brian Hortelano, Yonathan Kassaye, Dimitris Killinger, Adam Stanfield, Jordan Stark, Thomas Veilleux, and Nick Reuter This poster is available at VCU Scholars Compass: https://scholarscompass.vcu.edu/capstone/40 Team Members: Cody Boyd, Brian Hortelano, Yonathan Kassaye, Dimitris Killinger, Adam Stanfield, Jordan Stark, Thomas Veilleux Inertial Electrostatic Faculty Advisor: Dr. Sama Bilbao Y Leon, Mr. James G. Miller Sponsor: Confinement Fusor Dominion Virginia Power What is Fusion? Shielding Computational Modeling Because the D-D fusion reaction One of the potential uses of the fusor will be to results in the production of neutrons irradiate materials and see how they behave after and X-rays, shielding is necessary to certain levels of both fast and thermal neutron protect users from the radiation exposure. To reduce the amount of time and produced by the fusor. A Monte Carlo resources spent testing, a computational model n-Particle (MCNP) model was using XOOPIC, a particle interaction software, developed to calculate the necessary was developed to model the fusor. -

Basics of Radiation Radiation Safety Orientation Open Source Booklet 1 (June 1, 2018)
Basics of Radiation Radiation Safety Orientation Open Source Booklet 1 (June 1, 2018) Before working with radioactive material, it is helpful to recall… Radiation is energy released from a source. • Light is a familiar example of energy traveling some distance from its source. We understand that a light bulb can remain in one place and the light can move toward us to be detected by our eyes. • The Electromagnetic Spectrum is the entire range of wavelengths or frequencies of electromagnetic radiation extending from gamma rays to the longest radio waves and includes visible light. Radioactive materials release energy with enough power to cause ionizations and are on the high end of the electromagnetic spectrum. • Although our bodies cannot sense ionizing radiation, it is helpful to think ionizing radiation behaves similarly to light. o Travels in straight lines with decreasing intensity farther away from the source o May be reflected off certain surfaces (but not all) o Absorbed when interacting with materials You will be using radioactive material that releases energy in the form of ionizing radiation. Knowing about the basics of radiation will help you understand how to work safely with radioactive material. What is “ionizing radiation”? • Ionizing radiation is energy with enough power to remove tightly bound electrons from the orbit of an atom, causing the atom to become charged or ionized. • The charged atoms can damage the internal structures of living cells. The material near the charged atom absorbs the energy causing chemical bonds to break. Are all radioactive materials the same? No, not all radioactive materials are the same. -

The Use of Cosmic-Rays in Detecting Illicit Nuclear Materials
The use of Cosmic-Rays in Detecting Illicit Nuclear Materials Timothy Benjamin Blackwell Department of Physics and Astronomy University of Sheffield This dissertation is submitted for the degree of Doctor of Philosophy 05/05/2015 Declaration I hereby declare that except where specific reference is made to the work of others, the contents of this dissertation are original and have not been submitted in whole or in part for consideration for any other degree or qualification in this, or any other Univer- sity. This dissertation is the result of my own work and includes nothing which is the outcome of work done in collaboration, except where specifically indicated in the text. Timothy Benjamin Blackwell 05/05/2015 Acknowledgements This thesis could not have been completed without the tremendous support of many people. Firstly I would like to express special appreciation and thanks to my academic supervisor, Dr Vitaly A. Kudryavtsev for his expertise, understanding and encourage- ment. I have enjoyed our many discussions concerning my research topic throughout the PhD journey. I would also like to thank my viva examiners, Professor Lee Thomp- son and Dr Chris Steer, for the time you have taken out of your schedules, so that I may take this next step in my career. Thanks must also be given to Professor Francis Liven, Professor Neil Hyatt and the rest of the Nuclear FiRST DTC team, for the initial oppor- tunity to pursue postgraduate research. Appreciation is also given to the University of Sheffield, the HEP group and EPRSC for providing me with the facilities and funding, during this work. -

Experimental Γ Ray Spectroscopy and Investigations of Environmental Radioactivity
Experimental γ Ray Spectroscopy and Investigations of Environmental Radioactivity BY RANDOLPH S. PETERSON 216 α Po 84 10.64h. 212 Pb 1- 415 82 0- 239 β- 01- 0 60.6m 212 1+ 1630 Bi 2+ 1513 83 α β- 2+ 787 304ns 0+ 0 212 α Po 84 Experimental γ Ray Spectroscopy and Investigations of Environmental Radioactivity Randolph S. Peterson Physics Department The University of the South Sewanee, Tennessee Published by Spectrum Techniques All Rights Reserved Copyright 1996 TABLE OF CONTENTS Page Introduction ....................................................................................................................4 Basic Gamma Spectroscopy 1. Energy Calibration ................................................................................................... 7 2. Gamma Spectra from Common Commercial Sources ........................................ 10 3. Detector Energy Resolution .................................................................................. 12 Interaction of Radiation with Matter 4. Compton Scattering............................................................................................... 14 5. Pair Production and Annihilation ........................................................................ 17 6. Absorption of Gammas by Materials ..................................................................... 19 7. X Rays ..................................................................................................................... 21 Radioactive Decay 8. Multichannel Scaling and Half-life ..................................................................... -

NATO and NATO-Russia Nuclear Terms and Definitions
NATO/RUSSIA UNCLASSIFIED PART 1 PART 1 Nuclear Terms and Definitions in English APPENDIX 1 NATO and NATO-Russia Nuclear Terms and Definitions APPENDIX 2 Non-NATO Nuclear Terms and Definitions APPENDIX 3 Definitions of Nuclear Forces NATO/RUSSIA UNCLASSIFIED 1-1 2007 NATO/RUSSIA UNCLASSIFIED PART 1 NATO and NATO-Russia Nuclear Terms and Definitions APPENDIX 1 Source References: AAP-6 : NATO Glossary of Terms and Definitions AAP-21 : NATO Glossary of NBC Terms and Definitions CP&MT : NATO-Russia Glossary of Contemporary Political and Military Terms A active decontamination alpha particle A nuclear particle emitted by heavy radionuclides in the process of The employment of chemical, biological or mechanical processes decay. Alpha particles have a range of a few centimetres in air and to remove or neutralise chemical, biological or radioactive will not penetrate clothing or the unbroken skin but inhalation or materials. (AAP-21). ingestion will result in an enduring hazard to health (AAP-21). décontamination active активное обеззараживание particule alpha альфа-частицы active material antimissile system Material, such as plutonium and certain isotopes of uranium, The basic armament of missile defence systems, designed to which is capable of supporting a fission chain reaction (AAP-6). destroy ballistic and cruise missiles and their warheads. It includes See also fissile material. antimissile missiles, launchers, automated detection and matière fissile радиоактивное вещество identification, antimissile missile tracking and guidance, and main command posts with a range of computer and communications acute radiation dose equipment. They can be subdivided into short, medium and long- The total ionising radiation dose received at one time and over a range missile defence systems (CP&MT). -

A Measurement of the 2 Neutrino Double Beta Decay Rate of 130Te in the CUORICINO Experiment by Laura Katherine Kogler
A measurement of the 2 neutrino double beta decay rate of 130Te in the CUORICINO experiment by Laura Katherine Kogler A dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Physics in the Graduate Division of the University of California, Berkeley Committee in charge: Professor Stuart J. Freedman, Chair Professor Yury G. Kolomensky Professor Eric B. Norman Fall 2011 A measurement of the 2 neutrino double beta decay rate of 130Te in the CUORICINO experiment Copyright 2011 by Laura Katherine Kogler 1 Abstract A measurement of the 2 neutrino double beta decay rate of 130Te in the CUORICINO experiment by Laura Katherine Kogler Doctor of Philosophy in Physics University of California, Berkeley Professor Stuart J. Freedman, Chair CUORICINO was a cryogenic bolometer experiment designed to search for neutrinoless double beta decay and other rare processes, including double beta decay with two neutrinos (2νββ). The experiment was located at Laboratori Nazionali del Gran Sasso and ran for a period of about 5 years, from 2003 to 2008. The detector consisted of an array of 62 TeO2 crystals arranged in a tower and operated at a temperature of ∼10 mK. Events depositing energy in the detectors, such as radioactive decays or impinging particles, produced thermal pulses in the crystals which were read out using sensitive thermistors. The experiment included 4 enriched crystals, 2 enriched with 130Te and 2 with 128Te, in order to aid in the measurement of the 2νββ rate. The enriched crystals contained a total of ∼350 g 130Te. The 128-enriched (130-depleted) crystals were used as background monitors, so that the shared backgrounds could be subtracted from the energy spectrum of the 130- enriched crystals.