Chapter 3 the Fundamentals of Nuclear Physics Outline Natural
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
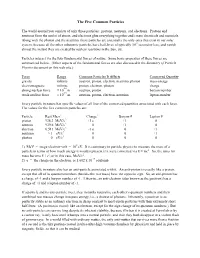
The Five Common Particles
The Five Common Particles The world around you consists of only three particles: protons, neutrons, and electrons. Protons and neutrons form the nuclei of atoms, and electrons glue everything together and create chemicals and materials. Along with the photon and the neutrino, these particles are essentially the only ones that exist in our solar system, because all the other subatomic particles have half-lives of typically 10-9 second or less, and vanish almost the instant they are created by nuclear reactions in the Sun, etc. Particles interact via the four fundamental forces of nature. Some basic properties of these forces are summarized below. (Other aspects of the fundamental forces are also discussed in the Summary of Particle Physics document on this web site.) Force Range Common Particles It Affects Conserved Quantity gravity infinite neutron, proton, electron, neutrino, photon mass-energy electromagnetic infinite proton, electron, photon charge -14 strong nuclear force ≈ 10 m neutron, proton baryon number -15 weak nuclear force ≈ 10 m neutron, proton, electron, neutrino lepton number Every particle in nature has specific values of all four of the conserved quantities associated with each force. The values for the five common particles are: Particle Rest Mass1 Charge2 Baryon # Lepton # proton 938.3 MeV/c2 +1 e +1 0 neutron 939.6 MeV/c2 0 +1 0 electron 0.511 MeV/c2 -1 e 0 +1 neutrino ≈ 1 eV/c2 0 0 +1 photon 0 eV/c2 0 0 0 1) MeV = mega-electron-volt = 106 eV. It is customary in particle physics to measure the mass of a particle in terms of how much energy it would represent if it were converted via E = mc2. -

Exotic Nuclear Decay Discovered
Exotic nuclear decay discovered The discovery, nearly a century after Becquerel, ofa novel mode of radioactive decay is a surprise, but one that confirms a decay as the chief means by which heavy nuclei shed mass. THOSE who decorate their offices with wall observations now reported, there are particle exists within the nucleus it will then charts showing the isotopes, stable and roughly 1,000 million times as many escape, or the rate of the corresponding otherwise, of the elements are in for a-particles and 14C nuclei in the decay of disintegration process, is thus a function of trouble. As things are, these elaborate mRa, perhaps as vivid a proof as there the height of the potential barrier, its width diagrams usually show by means of a could be of the dominance of the familiar and of the total decrease of the potential colour scheme of some kind which radio mechanisms of decay. energy of the system once the disinte actively unstable nuclei decay by which The rarity of these events is also the gration has taken place. means. Decays in which the a- or explanation why this novel form of radio This calculation, first made in simple {J-particles (electrons) are emitted activity has not previously been found. So form by Gamow, has more recently been predominate, but the diagrams must also much can be told from the account by Rose much refined and accounts for the make room for other less common and Jones of their observations, which "Gamow factors" used by Rose and Jones processes - positron emission, internal entailed more than half a year of running as part of their reason for believing that conversion (of an electron in an inner shell) time with detectors arranged so as to their disintegration products are 14C and and even fission. -

Neutrinoless Double Beta Decay
REPORT TO THE NUCLEAR SCIENCE ADVISORY COMMITTEE Neutrinoless Double Beta Decay NOVEMBER 18, 2015 NLDBD Report November 18, 2015 EXECUTIVE SUMMARY In March 2015, DOE and NSF charged NSAC Subcommittee on neutrinoless double beta decay (NLDBD) to provide additional guidance related to the development of next generation experimentation for this field. The new charge (Appendix A) requests a status report on the existing efforts in this subfield, along with an assessment of the necessary R&D required for each candidate technology before a future downselect. The Subcommittee membership was augmented to replace several members who were not able to continue in this phase (the present Subcommittee membership is attached as Appendix B). The Subcommittee solicited additional written input from the present worldwide collaborative efforts on double beta decay projects in order to collect the information necessary to address the new charge. An open meeting was held where these collaborations were invited to present material related to their current projects, conceptual designs for next generation experiments, and the critical R&D required before a potential down-select. We also heard presentations related to nuclear theory and the impact of future cosmological data on the subject of NLDBD. The Subcommittee presented its principal findings and comments in response to the March 2015 charge at the NSAC meeting in October 2015. The March 2015 charge requested the Subcommittee to: Assess the status of ongoing R&D for NLDBD candidate technology demonstrations for a possible future ton-scale NLDBD experiment. For each candidate technology demonstration, identify the major remaining R&D tasks needed ONLY to demonstrate downselect criteria, including the sensitivity goals, outlined in the NSAC report of May 2014. -

RIB Production by Photofission in the Framework of the ALTO Project
Available online at www.sciencedirect.com NIM B Beam Interactions with Materials & Atoms Nuclear Instruments and Methods in Physics Research B 266 (2008) 4092–4096 www.elsevier.com/locate/nimb RIB production by photofission in the framework of the ALTO project: First experimental measurements and Monte-Carlo simulations M. Cheikh Mhamed *, S. Essabaa, C. Lau, M. Lebois, B. Roussie`re, M. Ducourtieux, S. Franchoo, D. Guillemaud Mueller, F. Ibrahim, J.F. LeDu, J. Lesrel, A.C. Mueller, M. Raynaud, A. Said, D. Verney, S. Wurth Institut de Physique Nucle´aire, IN2P3-CNRS/Universite´ Paris-Sud, F-91406 Orsay Cedex, France Available online 11 June 2008 Abstract The ALTO facility (Acce´le´rateur Line´aire aupre`s du Tandem d’Orsay) has been built and is now under commissioning. The facility is intended for the production of low energy neutron-rich ion-beams by ISOL technique. This will open new perspectives in the study of nuclei very far from the valley of stability. Neutron-rich nuclei are produced by photofission in a thick uranium carbide target (UCx) using a 10 lA, 50 MeV electron beam. The target is the same as that already had been used on the previous deuteron based fission ISOL setup (PARRNE [F. Clapier et al., Phys. Rev. ST-AB (1998) 013501.]). The intended nominal fission rate is about 1011 fissions/s. We have studied the adequacy of a thick carbide uranium target to produce neutron-rich nuclei by photofission by means of Monte-Carlo simulations. We present the production rates in the target and after extraction and mass separation steps. -

Cyclotron Produced Lead-203 P
Postgrad Med J: first published as 10.1136/pgmj.51.601.751 on 1 November 1975. Downloaded from Postgraduate Medical Journal (November 1975) 51, 751-754. Cyclotron produced lead-203 P. L. HORLOCK M. L. THAKUR L.R.I.C. M.Sc., Ph.D. I. A. WATSON M.Sc. MRC Cyclotron Unit, Hammersmith Hospital, Du Cane Road, London W12 OHS Introduction TABLE 1. Isotopes of lead Radioactive isotopes of lead occur in the natural Isotope Half-life Principal y energy radioactive series and have been used as decay 194Pb m * tracers since the early days of radiochemistry. These 195Pb 17m * are 210Pb (ti--204 y, radium D), 212Pb (t--10. 6 h, 96Pb 37 m * thorium B) and 214Pb (t--26.8 m, radium B) all of 197Pb 42 m * which are from occurring decay 197pbm 42 m * separated naturally 198pb 24 h * products ofradium or thorium. There are in addition 19spbm 25 m * Protected by copyright. to these a number of other radioactive isotopes of 199Pb 90 m * lead which can be produced artificially with the aid 19spbm 122 m * of a nuclear reactor or a accelerator 20OPb 21-5 h ** 0-109 to 0-605 (complex) charge particle (daughter radiations) (Table 1). 2Pb 9-4 h * There are at least three criteria in choosing the 0oipbm 61 s * radioactive isotope for in vitro tracer studies. First, 20pb 3 x 105y *** TI X-rays, (daughter it should emit radiation which is easily detected. radiations) have a half-life to 202pbm 3-62 h * Secondly, it should long enough aPb 52-1 h ** 0-279 (81%) 0-401 (5%) 0-680 permit studies without excessive radioactive decay 203pbm 6-1 s * (0-9%) (no daughter occurring but not so long that waste disposal radiations) creates a problem. -

Investigational New Drug Applications for Positron Emission Tomography (PET) Drugs
Guidance Investigational New Drug Applications for Positron Emission Tomography (PET) Drugs GUIDANCE U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) December 2012 Clinical/Medical Guidance Investigational New Drug Applications for Positron Emission Tomography (PET) Drugs Additional copies are available from: Office of Communications Division of Drug Information, WO51, Room 2201 Center for Drug Evaluation and Research Food and Drug Administration 10903 New Hampshire Ave. Silver Spring, MD 20993-0002 Phone: 301-796-3400; Fax: 301-847-8714 [email protected] http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/default.htm U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) December 2012 Clinical/Medical Contains Nonbinding Recommendations TABLE OF CONTENTS I. INTRODUCTION..............................................................................................................................................1 II. BACKGROUND ................................................................................................................................................1 A. PET DRUGS .....................................................................................................................................................1 B. IND..................................................................................................................................................................2 -

1.1. Introduction the Phenomenon of Positron Annihilation Spectroscopy
PRINCIPLES OF POSITRON ANNIHILATION Chapter-1 __________________________________________________________________________________________ 1.1. Introduction The phenomenon of positron annihilation spectroscopy (PAS) has been utilized as nuclear method to probe a variety of material properties as well as to research problems in solid state physics. The field of solid state investigation with positrons started in the early fifties, when it was recognized that information could be obtained about the properties of solids by studying the annihilation of a positron and an electron as given by Dumond et al. [1] and Bendetti and Roichings [2]. In particular, the discovery of the interaction of positrons with defects in crystal solids by Mckenize et al. [3] has given a strong impetus to a further elaboration of the PAS. Currently, PAS is amongst the best nuclear methods, and its most recent developments are documented in the proceedings of the latest positron annihilation conferences [4-8]. PAS is successfully applied for the investigation of electron characteristics and defect structures present in materials, magnetic structures of solids, plastic deformation at low and high temperature, and phase transformations in alloys, semiconductors, polymers, porous material, etc. Its applications extend from advanced problems of solid state physics and materials science to industrial use. It is also widely used in chemistry, biology, and medicine (e.g. locating tumors). As the process of measurement does not mostly influence the properties of the investigated sample, PAS is a non-destructive testing approach that allows the subsequent study of a sample by other methods. As experimental equipment for many applications, PAS is commercially produced and is relatively cheap, thus, increasingly more research laboratories are using PAS for basic research, diagnostics of machine parts working in hard conditions, and for characterization of high-tech materials. -

A New Gamma Camera for Positron Emission Tomography
INIS-mf—11552 A new gamma camera for Positron Emission Tomography NL89C0813 P. SCHOTANUS A new gamma camera for Positron Emission Tomography A new gamma camera for Positron Emission Tomography PROEFSCHRIFT TER VERKRIJGING VAN DE GRAAD VAN DOCTOR AAN DE TECHNISCHE UNIVERSITEIT DELFT, OP GEZAG VAN DE RECTOR MAGNIFICUS, PROF.DRS. P.A. SCHENCK, IN HET OPENBAAR TE VERDEDIGEN TEN OVERSTAAN VAN EEN COMMISSIE, AANGEWEZEN DOOR HET COLLEGE VAN DECANEN, OP DINSDAG 20 SEPTEMBER 1988TE 16.00 UUR. DOOR PAUL SCHOTANUS '$ DOCTORANDUS IN DE NATUURKUNDE GEBOREN TE EINDHOVEN Dit proefschrift is goedgekeurd door de promotor Prof.dr. A.H. Wapstra s ••I Sommige boeken schijnen geschreven te zijn.niet opdat men er iets uit zou leren, maar opdat men weten zal, dat de schrijver iets geweten heeft. Goethe Contents page 1 Introduction 1 2 Nuclear diagnostics as a tool in medical science; principles and applications 2.1 The position of nuclear diagnostics in medical science 2 2.2 The detection of radiation in nuclear diagnostics: 5 standard techniques 2.3 Positron emission tomography 7 2.4 Positron emitting isotopes 9 2.5 Examples of radiodiagnostic studies with PET 11 2.6 Comparison of PET with other diagnostic techniques 12 3 Detectors for positron emission tomography 3.1 The absorption d 5H keV annihilation radiation in solids 15 3.2 Scintillators for the detection of annihilation radiation 21 3.3 The detection of scintillation light 23 3.4 Alternative ways to detect annihilation radiation 28 3-5 Determination of the point of annihilation: detector geometry, -

Electron Capture in Stars
Electron capture in stars K Langanke1;2, G Mart´ınez-Pinedo1;2;3 and R.G.T. Zegers4;5;6 1GSI Helmholtzzentrum f¨urSchwerionenforschung, D-64291 Darmstadt, Germany 2Institut f¨urKernphysik (Theoriezentrum), Department of Physics, Technische Universit¨atDarmstadt, D-64298 Darmstadt, Germany 3Helmholtz Forschungsakademie Hessen f¨urFAIR, GSI Helmholtzzentrum f¨ur Schwerionenforschung, D-64291 Darmstadt, Germany 4 National Superconducting Cyclotron Laboratory, Michigan State University, East Lansing, Michigan 48824, USA 5 Joint Institute for Nuclear Astrophysics: Center for the Evolution of the Elements, Michigan State University, East Lansing, Michigan 48824, USA 6 Department of Physics and Astronomy, Michigan State University, East Lansing, Michigan 48824, USA E-mail: [email protected], [email protected], [email protected] Abstract. Electron captures on nuclei play an essential role for the dynamics of several astrophysical objects, including core-collapse and thermonuclear supernovae, the crust of accreting neutron stars in binary systems and the final core evolution of intermediate mass stars. In these astrophysical objects, the capture occurs at finite temperatures and at densities at which the electrons form a degenerate relativistic electron gas. The capture rates can be derived in perturbation theory where allowed nuclear transitions (Gamow-Teller transitions) dominate, except at the higher temperatures achieved in core-collapse supernovae where also forbidden transitions contribute significantly to the rates. There has been decisive progress in recent years in measuring Gamow-Teller (GT) strength distributions using novel experimental techniques based on charge-exchange reactions. These measurements provide not only data for the GT distributions of ground states for many relevant nuclei, but also serve as valuable constraints for nuclear models which are needed to derive the capture rates for the arXiv:2009.01750v1 [nucl-th] 3 Sep 2020 many nuclei, for which no data exist yet. -

Radioactive Decay
North Berwick High School Department of Physics Higher Physics Unit 2 Particles and Waves Section 3 Fission and Fusion Section 3 Fission and Fusion Note Making Make a dictionary with the meanings of any new words. Einstein and nuclear energy 1. Write down Einstein’s famous equation along with units. 2. Explain the importance of this equation and its relevance to nuclear power. A basic model of the atom 1. Copy the components of the atom diagram and state the meanings of A and Z. 2. Copy the table on page 5 and state the difference between elements and isotopes. Radioactive decay 1. Explain what is meant by radioactive decay and copy the summary table for the three types of nuclear radiation. 2. Describe an alpha particle, including the reason for its short range and copy the panel showing Plutonium decay. 3. Describe a beta particle, including its range and copy the panel showing Tritium decay. 4. Describe a gamma ray, including its range. Fission: spontaneous decay and nuclear bombardment 1. Describe the differences between the two methods of decay and copy the equation on page 10. Nuclear fission and E = mc2 1. Explain what is meant by the terms ‘mass difference’ and ‘chain reaction’. 2. Copy the example showing the energy released during a fission reaction. 3. Briefly describe controlled fission in a nuclear reactor. Nuclear fusion: energy of the future? 1. Explain why nuclear fusion might be a preferred source of energy in the future. 2. Describe some of the difficulties associated with maintaining a controlled fusion reaction. -

Heavy Element Nucleosynthesis
Heavy Element Nucleosynthesis A summary of the nucleosynthesis of light elements is as follows 4He Hydrogen burning 3He Incomplete PP chain (H burning) 2H, Li, Be, B Non-thermal processes (spallation) 14N, 13C, 15N, 17O CNO processing 12C, 16O Helium burning 18O, 22Ne α captures on 14N (He burning) 20Ne, Na, Mg, Al, 28Si Partly from carbon burning Mg, Al, Si, P, S Partly from oxygen burning Ar, Ca, Ti, Cr, Fe, Ni Partly from silicon burning Isotopes heavier than iron (as well as some intermediate weight iso- topes) are made through neutron captures. Recall that the prob- ability for a non-resonant reaction contained two components: an exponential reflective of the quantum tunneling needed to overcome electrostatic repulsion, and an inverse energy dependence arising from the de Broglie wavelength of the particles. For neutron cap- tures, there is no electrostatic repulsion, and, in complex nuclei, virtually all particle encounters involve resonances. As a result, neutron capture cross-sections are large, and are very nearly inde- pendent of energy. To appreciate how heavy elements can be built up, we must first consider the lifetime of an isotope against neutron capture. If the cross-section for neutron capture is independent of energy, then the lifetime of the species will be ( )1=2 1 1 1 µn τn = ≈ = Nnhσvi NnhσivT Nnhσi 2kT For a typical neutron cross-section of hσi ∼ 10−25 cm2 and a tem- 8 9 perature of 5 × 10 K, τn ∼ 10 =Nn years. Next consider the stability of a neutron rich isotope. If the ratio of of neutrons to protons in an atomic nucleus becomes too large, the nucleus becomes unstable to beta-decay, and a neutron is changed into a proton via − (Z; A+1) −! (Z+1;A+1) + e +ν ¯e (27:1) The timescale for this decay is typically on the order of hours, or ∼ 10−3 years (with a factor of ∼ 103 scatter). -

Cooperative Internal Conversion Process by Proton Exchange
Cooperative internal conversion process by proton exchange P´eter K´alm´an∗ and Tam´as Keszthelyi† Budapest University of Technology and Economics, Institute of Physics, Budafoki ´ut 8. F., H-1521 Budapest, Hungary A generalization of the recently discovered cooperative internal conversion process is investigated theoretically. In the cooperative internal conversion process by proton exchange investigated the coupling of bound-free electron and proton transitions due to the dipole term of their Coulomb interaction permits cooperation of two nuclei leading to proton exchange and an electron emission. General expression of the cross section of the process obtained in the one particle spherical nuclear shell model is presented. As a numerical example the cooperative internal conversion process by proton exchange in Al is dealt with. As a further generalization, cooperative internal conversion process by heavy charged particle exchange and as an example of it the cooperative internal con- version process by triton exchange is discussed. The process is also connected to the field of nuclear waste disposal. PACS numbers: 23.20.Nx, 25.90.+k, 28.41.Kw, Keywords: internal conversion and extranuclear effects, other topics of nuclear reactions: specific reactions, radioactive wastes, waste disposal In a recent paper [1] a new phenomenon, the coop- erative internal conversion process (CICP) is discussed which is a special type of the well known internal conver- sion process [2]. In CICP two nuclei cooperate by neutron exchange creating final nuclei of energy lower than the en- ergy of the initial nuclei. The process is initiated by the coupling of bound-free electron and neutron transitions due to the dipole term of their Coulomb interaction in the initial atom leading to the creation of a virtual free neutron which is captured through strong interaction by an other nucleus.