Mouse Models of Altered Protein Kinase a Signaling
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplemental Information to Mammadova-Bach Et Al., “Laminin Α1 Orchestrates VEGFA Functions in the Ecosystem of Colorectal Carcinogenesis”
Supplemental information to Mammadova-Bach et al., “Laminin α1 orchestrates VEGFA functions in the ecosystem of colorectal carcinogenesis” Supplemental material and methods Cloning of the villin-LMα1 vector The plasmid pBS-villin-promoter containing the 3.5 Kb of the murine villin promoter, the first non coding exon, 5.5 kb of the first intron and 15 nucleotides of the second villin exon, was generated by S. Robine (Institut Curie, Paris, France). The EcoRI site in the multi cloning site was destroyed by fill in ligation with T4 polymerase according to the manufacturer`s instructions (New England Biolabs, Ozyme, Saint Quentin en Yvelines, France). Site directed mutagenesis (GeneEditor in vitro Site-Directed Mutagenesis system, Promega, Charbonnières-les-Bains, France) was then used to introduce a BsiWI site before the start codon of the villin coding sequence using the 5’ phosphorylated primer: 5’CCTTCTCCTCTAGGCTCGCGTACGATGACGTCGGACTTGCGG3’. A double strand annealed oligonucleotide, 5’GGCCGGACGCGTGAATTCGTCGACGC3’ and 5’GGCCGCGTCGACGAATTCACGC GTCC3’ containing restriction site for MluI, EcoRI and SalI were inserted in the NotI site (present in the multi cloning site), generating the plasmid pBS-villin-promoter-MES. The SV40 polyA region of the pEGFP plasmid (Clontech, Ozyme, Saint Quentin Yvelines, France) was amplified by PCR using primers 5’GGCGCCTCTAGATCATAATCAGCCATA3’ and 5’GGCGCCCTTAAGATACATTGATGAGTT3’ before subcloning into the pGEMTeasy vector (Promega, Charbonnières-les-Bains, France). After EcoRI digestion, the SV40 polyA fragment was purified with the NucleoSpin Extract II kit (Machery-Nagel, Hoerdt, France) and then subcloned into the EcoRI site of the plasmid pBS-villin-promoter-MES. Site directed mutagenesis was used to introduce a BsiWI site (5’ phosphorylated AGCGCAGGGAGCGGCGGCCGTACGATGCGCGGCAGCGGCACG3’) before the initiation codon and a MluI site (5’ phosphorylated 1 CCCGGGCCTGAGCCCTAAACGCGTGCCAGCCTCTGCCCTTGG3’) after the stop codon in the full length cDNA coding for the mouse LMα1 in the pCIS vector (kindly provided by P. -

Tumour-Agnostic Therapy for Pancreatic Cancer and Biliary Tract Cancer
diagnostics Review Tumour-Agnostic Therapy for Pancreatic Cancer and Biliary Tract Cancer Shunsuke Kato Department of Clinical Oncology, Juntendo University Graduate School of Medicine, 2-1-1, Hongo, Bunkyo-ku, Tokyo 113-8421, Japan; [email protected]; Tel.: +81-3-5802-1543 Abstract: The prognosis of patients with solid tumours has remarkably improved with the develop- ment of molecular-targeted drugs and immune checkpoint inhibitors. However, the improvements in the prognosis of pancreatic cancer and biliary tract cancer is delayed compared to other carcinomas, and the 5-year survival rates of distal-stage disease are approximately 10 and 20%, respectively. How- ever, a comprehensive analysis of tumour cells using The Cancer Genome Atlas (TCGA) project has led to the identification of various driver mutations. Evidently, few mutations exist across organs, and basket trials targeting driver mutations regardless of the primary organ are being actively conducted. Such basket trials not only focus on the gate keeper-type oncogene mutations, such as HER2 and BRAF, but also focus on the caretaker-type tumour suppressor genes, such as BRCA1/2, mismatch repair-related genes, which cause hereditary cancer syndrome. As oncogene panel testing is a vital approach in routine practice, clinicians should devise a strategy for improved understanding of the cancer genome. Here, the gene mutation profiles of pancreatic cancer and biliary tract cancer have been outlined and the current status of tumour-agnostic therapy in these cancers has been reported. Keywords: pancreatic cancer; biliary tract cancer; targeted therapy; solid tumours; driver mutations; agonist therapy Citation: Kato, S. Tumour-Agnostic Therapy for Pancreatic Cancer and 1. -

Gpr161 Anchoring of PKA Consolidates GPCR and Camp Signaling
Gpr161 anchoring of PKA consolidates GPCR and cAMP signaling Verena A. Bachmanna,1, Johanna E. Mayrhofera,1, Ronit Ilouzb, Philipp Tschaiknerc, Philipp Raffeinera, Ruth Röcka, Mathieu Courcellesd,e, Federico Apeltf, Tsan-Wen Lub,g, George S. Baillieh, Pierre Thibaultd,i, Pia Aanstadc, Ulrich Stelzlf,j, Susan S. Taylorb,g,2, and Eduard Stefana,2 aInstitute of Biochemistry and Center for Molecular Biosciences, University of Innsbruck, 6020 Innsbruck, Austria; bDepartment of Chemistry and Biochemistry, University of California, San Diego, CA 92093; cInstitute of Molecular Biology, University of Innsbruck, 6020 Innsbruck, Austria; dInstitute for Research in Immunology and Cancer, Université de Montréal, Montreal, QC, Canada H3C 3J7; eDépartement de Biochimie, Université de Montréal, Montreal, QC, Canada H3C 3J7; fOtto-Warburg Laboratory, Max Planck Institute for Molecular Genetics, 14195 Berlin, Germany; gDepartment of Pharmacology, University of California, San Diego, CA 92093; hInstitute of Cardiovascular and Medical Sciences, University of Glasgow, Glasgow, G12 8QQ, United Kingdom; iDepartment of Chemistry, Université de Montréal, Montreal, QC, Canada H3C 3J7; and jInstitute of Pharmaceutical Sciences, Pharmaceutical Chemistry, University of Graz, 8010 Graz, Austria Contributed by Susan S. Taylor, May 24, 2016 (sent for review February 18, 2016; reviewed by John J. G. Tesmer and Mark von Zastrow) Scaffolding proteins organize the information flow from activated G accounts for nanomolar binding affinities to PKA R subunit dimers protein-coupled receptors (GPCRs) to intracellular effector cascades (12, 13). Moreover, additional components of the cAMP signaling both spatially and temporally. By this means, signaling scaffolds, such machinery, such as GPCRs, adenylyl cyclases, and phosphodiester- as A-kinase anchoring proteins (AKAPs), compartmentalize kinase ases, physically interact with AKAPs (1, 5, 11, 14). -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Emerging Genetic Alterations Linked to Male Infertility: X-Chromosome Copy Number Variation and Spermatogenesis Regulatory Genes’ Expression
Central Annals of Reproductive Medicine and Treatment Bringing Excellence in Open Access Research Article *Corresponding author Susana Fernandes, Department of Genetics, Faculty of Medicine of University of Porto, Al. Prof. Hernâni Emerging Genetic Alterations Monteiro, 4200 - 319 Porto, Portugal; Tel: 351 22 551 36 47; Email: Submitted: 12 October 2016 Linked to Male Infertility: Accepted: 10 February 2017 Published: 14 February 2017 X-Chromosome Copy Number Copyright © 2017 Fernandes et al. Variation and Spermatogenesis OPEN ACCESS Keywords Regulatory Genes’ Expression • Male infertility • Spermatogenesis Catarina Costa1, Maria João Pinho1,2, Alberto Barros1,2,3, and • DNA copy number variations • Gene expression Susana Fernandes1,2* 1Department of Genetics, Faculty of Medicine of University of Porto, Portugal 2i3S – Instituto de Investigação e Inovação em Saúde, University of Porto, Portugal 3Centre for Reproductive Genetics A Barros, Portugal Abstract The etiopathogenesis of primary testicular failure remains undefined in 50% of cases. Most of these idiopathic cases probably result from genetic mutations/anomalies. Novel causes, like Copy Number Variation and gene expression profile, are being explored thanks to recent advances in the field of genetics. Our aim was to study Copy Number Variation (CNV) 67, a patient-specific CNV related to spermatogenic anomaly and evaluate the expression of regulatory genes AKAP4, responsible for sperm fibrous sheet assembly, and STAG3, essential for sister chromatid cohesion during meiosis. One hundred infertile men were tested for CNV67 with quantitative PCR (qPCR). Quantitative real-time PCR was performed to evaluate gene expression patterns of the two mentioned genes in testicular biopsies from 22 idiopathic infertile patients. CNV67 deletion was found in 2% of patients, with the same semen phenotype described in previous studies. -

Identification and Characterization of RHOA-Interacting Proteins in Bovine Spermatozoa1
BIOLOGY OF REPRODUCTION 78, 184–192 (2008) Published online before print 10 October 2007. DOI 10.1095/biolreprod.107.062943 Identification and Characterization of RHOA-Interacting Proteins in Bovine Spermatozoa1 Sarah E. Fiedler, Malini Bajpai, and Daniel W. Carr2 Department of Medicine, Oregon Health & Sciences University and Veterans Affairs Medical Center, Portland, Oregon 97239 ABSTRACT Guanine nucleotide exchange factors (GEFs) catalyze the GDP for GTP exchange [2]. Activation is negatively regulated by In somatic cells, RHOA mediates actin dynamics through a both guanine nucleotide dissociation inhibitors (RHO GDIs) GNA13-mediated signaling cascade involving RHO kinase and GTPase-activating proteins (GAPs) [1, 2]. Endogenous (ROCK), LIM kinase (LIMK), and cofilin. RHOA can be RHO can be inactivated via C3 exoenzyme ADP-ribosylation, negatively regulated by protein kinase A (PRKA), and it and studies have demonstrated RHO involvement in actin-based interacts with members of the A-kinase anchoring (AKAP) cytoskeletal response to extracellular signals, including lyso- family via intermediary proteins. In spermatozoa, actin poly- merization precedes the acrosome reaction, which is necessary phosphatidic acid (LPA) [2–4]. LPA is known to signal through for normal fertility. The present study was undertaken to G-protein-coupled receptors (GPCRs) [4, 5]; specifically, LPA- determine whether the GNA13-mediated RHOA signaling activated GNA13 (formerly Ga13) promotes RHO activation pathway may be involved in acrosome reaction in bovine through GEFs [4, 6]. Activated RHO-GTP then signals RHO caudal sperm, and whether AKAPs may be involved in its kinase (ROCK), resulting in the phosphorylation and activation targeting and regulation. GNA13, RHOA, ROCK2, LIMK2, and of LIM-kinase (LIMK), which in turn phosphorylates and cofilin were all detected by Western blot in bovine caudal inactivates cofilin, an actin depolymerizer, the end result being sperm. -

Glycogen Synthase Kinase-3 Is Activated in Neuronal Cells by G 12
The Journal of Neuroscience, August 15, 2002, 22(16):6863–6875 Glycogen Synthase Kinase-3 Is Activated in Neuronal Cells by G␣ ␣ 12 and G 13 by Rho-Independent and Rho-Dependent Mechanisms C. Laura Sayas, Jesu´ s Avila, and Francisco Wandosell Centro de Biologı´a Molecular “Severo Ochoa”, Consejo Superior de Investigaciones Cientı´ficas, Universidad Auto´ noma de Madrid, Cantoblanco, Madrid 28049, Spain ␣ ␣ ␣ ␣ Glycogen synthase kinase-3 (GSK-3) was generally considered tively active G 12 (G 12QL) and G 13 (G 13QL) in Neuro2a cells a constitutively active enzyme, only regulated by inhibition. induces upregulation of GSK-3 activity. Furthermore, overex- Here we describe that GSK-3 is activated by lysophosphatidic pression of constitutively active RhoA (RhoAV14) also activates ␣ acid (LPA) during neurite retraction in rat cerebellar granule GSK-3 However, the activation of GSK-3 by G 13 is blocked by neurons. GSK-3 activation correlates with an increase in GSK-3 coexpression with C3 transferase, whereas C3 does not block ␣ tyrosine phosphorylation. In addition, LPA induces a GSK-3- GSK-3 activation by G 12. Thus, we demonstrate that GSK-3 is ␣ ␣ mediated hyperphosphorylation of the microtubule-associated activated by both G 12 and G 13 in neuronal cells. However, ␣ protein tau. Inhibition of GSK-3 by lithium partially blocks neu- GSK-3 activation by G 13 is Rho-mediated, whereas GSK-3 ␣ rite retraction, indicating that GSK-3 activation is important but activation by G 12 is Rho-independent. The results presented not essential for the neurite retraction progress. GSK-3 activa- here imply the existence of a previously unknown mechanism of ␣ tion by LPA in cerebellar granule neurons is neither downstream GSK-3 activation by G 12/13 subunits. -

Table S1. List of Oligonucleotide Primers Used
Table S1. List of oligonucleotide primers used. Cla4 LF-5' GTAGGATCCGCTCTGTCAAGCCTCCGACC M629Arev CCTCCCTCCATGTACTCcgcGATGACCCAgAGCTCGTTG M629Afwd CAACGAGCTcTGGGTCATCgcgGAGTACATGGAGGGAGG LF-3' GTAGGCCATCTAGGCCGCAATCTCGTCAAGTAAAGTCG RF-5' GTAGGCCTGAGTGGCCCGAGATTGCAACGTGTAACC RF-3' GTAGGATCCCGTACGCTGCGATCGCTTGC Ukc1 LF-5' GCAATATTATGTCTACTTTGAGCG M398Arev CCGCCGGGCAAgAAtTCcgcGAGAAGGTACAGATACGc M398Afwd gCGTATCTGTACCTTCTCgcgGAaTTcTTGCCCGGCGG LF-3' GAGGCCATCTAGGCCATTTACGATGGCAGACAAAGG RF-5' GTGGCCTGAGTGGCCATTGGTTTGGGCGAATGGC RF-3' GCAATATTCGTACGTCAACAGCGCG Nrc2 LF-5' GCAATATTTCGAAAAGGGTCGTTCC M454Grev GCCACCCATGCAGTAcTCgccGCAGAGGTAGAGGTAATC M454Gfwd GATTACCTCTACCTCTGCggcGAgTACTGCATGGGTGGC LF-3' GAGGCCATCTAGGCCGACGAGTGAAGCTTTCGAGCG RF-5' GAGGCCTGAGTGGCCTAAGCATCTTGGCTTCTGC RF-3' GCAATATTCGGTCAACGCTTTTCAGATACC Ipl1 LF-5' GTCAATATTCTACTTTGTGAAGACGCTGC M629Arev GCTCCCCACGACCAGCgAATTCGATagcGAGGAAGACTCGGCCCTCATC M629Afwd GATGAGGGCCGAGTCTTCCTCgctATCGAATTcGCTGGTCGTGGGGAGC LF-3' TGAGGCCATCTAGGCCGGTGCCTTAGATTCCGTATAGC RF-5' CATGGCCTGAGTGGCCGATTCTTCTTCTGTCATCGAC RF-3' GACAATATTGCTGACCTTGTCTACTTGG Ire1 LF-5' GCAATATTAAAGCACAACTCAACGC D1014Arev CCGTAGCCAAGCACCTCGgCCGAtATcGTGAGCGAAG D1014Afwd CTTCGCTCACgATaTCGGcCGAGGTGCTTGGCTACGG LF-3' GAGGCCATCTAGGCCAACTGGGCAAAGGAGATGGA RF-5' GAGGCCTGAGTGGCCGTGCGCCTGTGTATCTCTTTG RF-3' GCAATATTGGCCATCTGAGGGCTGAC Kin28 LF-5' GACAATATTCATCTTTCACCCTTCCAAAG L94Arev TGATGAGTGCTTCTAGATTGGTGTCggcGAAcTCgAGCACCAGGTTG L94Afwd CAACCTGGTGCTcGAgTTCgccGACACCAATCTAGAAGCACTCATCA LF-3' TGAGGCCATCTAGGCCCACAGAGATCCGCTTTAATGC RF-5' CATGGCCTGAGTGGCCAGGGCTAGTACGACCTCG -

WO 2019/079361 Al 25 April 2019 (25.04.2019) W 1P O PCT
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization I International Bureau (10) International Publication Number (43) International Publication Date WO 2019/079361 Al 25 April 2019 (25.04.2019) W 1P O PCT (51) International Patent Classification: CA, CH, CL, CN, CO, CR, CU, CZ, DE, DJ, DK, DM, DO, C12Q 1/68 (2018.01) A61P 31/18 (2006.01) DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, HN, C12Q 1/70 (2006.01) HR, HU, ID, IL, IN, IR, IS, JO, JP, KE, KG, KH, KN, KP, KR, KW, KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, (21) International Application Number: MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, PCT/US2018/056167 OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, (22) International Filing Date: SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, 16 October 2018 (16. 10.2018) TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (25) Filing Language: English (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (26) Publication Language: English GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, ST, SZ, TZ, (30) Priority Data: UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, TJ, 62/573,025 16 October 2017 (16. 10.2017) US TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, EE, ES, FI, FR, GB, GR, HR, HU, ΓΕ , IS, IT, LT, LU, LV, (71) Applicant: MASSACHUSETTS INSTITUTE OF MC, MK, MT, NL, NO, PL, PT, RO, RS, SE, SI, SK, SM, TECHNOLOGY [US/US]; 77 Massachusetts Avenue, TR), OAPI (BF, BJ, CF, CG, CI, CM, GA, GN, GQ, GW, Cambridge, Massachusetts 02139 (US). -
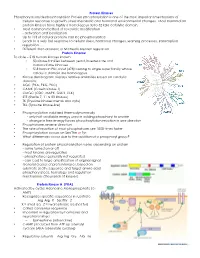
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

Supplementary Table 1. Pain and PTSS Associated Genes (N = 604
Supplementary Table 1. Pain and PTSS associated genes (n = 604) compiled from three established pain gene databases (PainNetworks,[61] Algynomics,[52] and PainGenes[42]) and one PTSS gene database (PTSDgene[88]). These genes were used in in silico analyses aimed at identifying miRNA that are predicted to preferentially target this list genes vs. a random set of genes (of the same length). ABCC4 ACE2 ACHE ACPP ACSL1 ADAM11 ADAMTS5 ADCY5 ADCYAP1 ADCYAP1R1 ADM ADORA2A ADORA2B ADRA1A ADRA1B ADRA1D ADRA2A ADRA2C ADRB1 ADRB2 ADRB3 ADRBK1 ADRBK2 AGTR2 ALOX12 ANO1 ANO3 APOE APP AQP1 AQP4 ARL5B ARRB1 ARRB2 ASIC1 ASIC2 ATF1 ATF3 ATF6B ATP1A1 ATP1B3 ATP2B1 ATP6V1A ATP6V1B2 ATP6V1G2 AVPR1A AVPR2 BACE1 BAMBI BDKRB2 BDNF BHLHE22 BTG2 CA8 CACNA1A CACNA1B CACNA1C CACNA1E CACNA1G CACNA1H CACNA2D1 CACNA2D2 CACNA2D3 CACNB3 CACNG2 CALB1 CALCRL CALM2 CAMK2A CAMK2B CAMK4 CAT CCK CCKAR CCKBR CCL2 CCL3 CCL4 CCR1 CCR7 CD274 CD38 CD4 CD40 CDH11 CDK5 CDK5R1 CDKN1A CHRM1 CHRM2 CHRM3 CHRM5 CHRNA5 CHRNA7 CHRNB2 CHRNB4 CHUK CLCN6 CLOCK CNGA3 CNR1 COL11A2 COL9A1 COMT COQ10A CPN1 CPS1 CREB1 CRH CRHBP CRHR1 CRHR2 CRIP2 CRYAA CSF2 CSF2RB CSK CSMD1 CSNK1A1 CSNK1E CTSB CTSS CX3CL1 CXCL5 CXCR3 CXCR4 CYBB CYP19A1 CYP2D6 CYP3A4 DAB1 DAO DBH DBI DICER1 DISC1 DLG2 DLG4 DPCR1 DPP4 DRD1 DRD2 DRD3 DRD4 DRGX DTNBP1 DUSP6 ECE2 EDN1 EDNRA EDNRB EFNB1 EFNB2 EGF EGFR EGR1 EGR3 ENPP2 EPB41L2 EPHB1 EPHB2 EPHB3 EPHB4 EPHB6 EPHX2 ERBB2 ERBB4 EREG ESR1 ESR2 ETV1 EZR F2R F2RL1 F2RL2 FAAH FAM19A4 FGF2 FKBP5 FLOT1 FMR1 FOS FOSB FOSL2 FOXN1 FRMPD4 FSTL1 FYN GABARAPL1 GABBR1 GABBR2 GABRA2 GABRA4 -

© Copyright 2021 Heather Raquel Dahlin
© Copyright 2021 Heather Raquel Dahlin The Structure of Sperm Autoantigenic Protein (SPA17): An R2D2 Protein Critical to Cilia and Implicated in Oncogenesis Heather Raquel Dahlin A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy University of Washington 2021 Reading Committee: John D. Scott, Chair Ning Zheng Linda Wordeman Program Authorized to Offer Degree: Pharmacology University of Washington Abstract Structure of SPA17: An R2D2 Protein Critical to Cilia and Implicated in Oncogenesis Heather Raquel Dahlin Chair of the Supervisory Committee: John D. Scott, Ph.D., Edwin G. Krebs- Speights Professor of Cell Signaling and Cancer Biology Pharmacology A-Kinase Anchoring proteins (AKAPs) localize the activity of cyclic AMP (cAMP)-Dependent Protein Kinase (PKA) through interaction of an amphipathic helix that binds to a conserved RIIα docking and dimerization (R2D2) domain on the N-terminus of PKA. Genome analysis indicates that at least thirteen other RIIα superfamily proteins exist in humans, which are not coupled to cyclic nucleotide binding domains and are largely localized to cilia and flagella. The newly reported R2D2 proteins exist in two lineages differing by their similarity to Type I or Type II PKA. Moreover, R2D2 domains bind to AKAPs and can contain extra regulatory sequences conferring novel functions and binding specificity. Here we detail the structure of one such domain comprising the N-terminus of Sperm Autoantigenic Protein 17 (SPA17) resolved to 1.72 Å. The structure of core hydrophobic sites for dimerization and AKAP binding are highly conserved between PKA and SPA17. Additional flanking sequences outside of the core R2D2 domain occlude the AKAP binding site and reduce the affinity for AKAP helices in the absence of heterodimerization with another R2D2 protein, ROPN1L.