Zandstra.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

2,4-Dichlorophenoxyacetic Acid
2,4-Dichlorophenoxyacetic acid 2,4-Dichlorophenoxyacetic acid IUPAC (2,4-dichlorophenoxy)acetic acid name 2,4-D Other hedonal names trinoxol Identifiers CAS [94-75-7] number SMILES OC(COC1=CC=C(Cl)C=C1Cl)=O ChemSpider 1441 ID Properties Molecular C H Cl O formula 8 6 2 3 Molar mass 221.04 g mol−1 Appearance white to yellow powder Melting point 140.5 °C (413.5 K) Boiling 160 °C (0.4 mm Hg) point Solubility in 900 mg/L (25 °C) water Related compounds Related 2,4,5-T, Dichlorprop compounds Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) 2,4-Dichlorophenoxyacetic acid (2,4-D) is a common systemic herbicide used in the control of broadleaf weeds. It is the most widely used herbicide in the world, and the third most commonly used in North America.[1] 2,4-D is also an important synthetic auxin, often used in laboratories for plant research and as a supplement in plant cell culture media such as MS medium. History 2,4-D was developed during World War II by a British team at Rothamsted Experimental Station, under the leadership of Judah Hirsch Quastel, aiming to increase crop yields for a nation at war.[citation needed] When it was commercially released in 1946, it became the first successful selective herbicide and allowed for greatly enhanced weed control in wheat, maize (corn), rice, and similar cereal grass crop, because it only kills dicots, leaving behind monocots. Mechanism of herbicide action 2,4-D is a synthetic auxin, which is a class of plant growth regulators. -

Common and Chemical Names of Herbicides Approved by the WSSA
Weed Science 2010 58:511–518 Common and Chemical Names of Herbicides Approved by the Weed Science Society of America Below is the complete list of all common and chemical of herbicides as approved by the International Organization names of herbicides approved by the Weed Science Society of for Standardization (ISO). A sponsor may submit a proposal America (WSSA) and updated as of September 1, 2010. for a common name directly to the WSSA Terminology Beginning in 1996, it has been published yearly in the last Committee. issue of Weed Science with Directions for Contributors to A herbicide common name is not synonymous with Weed Science. This list is published in lieu of the selections a commercial formulation of the same herbicide, and in printed previously on the back cover of Weed Science. Only many instances, is not synonymous with the active ingredient common and chemical names included in this complete of a commercial formulation as identified on the product list should be used in WSSA publications. In the absence of label. If the herbicide is a salt or simple ester of a parent a WSSA-approved common name, the industry code number compound, the WSSA common name applies to the parent as compiled by the Chemical Abstracts Service (CAS) with compound only. CAS systematic chemical name or the systematic chemical The chemical name used in this list is that preferred by the name alone may be used. The current approved list is also Chemical Abstracts Service (CAS) according to their system of available at our web site (www.wssa.net). -

Exposure to Herbicides in House Dust and Risk of Childhood Acute Lymphoblastic Leukemia
Journal of Exposure Science and Environmental Epidemiology (2013) 23, 363–370 & 2013 Nature America, Inc. All rights reserved 1559-0631/13 www.nature.com/jes ORIGINAL ARTICLE Exposure to herbicides in house dust and risk of childhood acute lymphoblastic leukemia Catherine Metayer1, Joanne S. Colt2, Patricia A. Buffler1, Helen D. Reed3, Steve Selvin1, Vonda Crouse4 and Mary H. Ward2 We examine the association between exposure to herbicides and childhood acute lymphoblastic leukemia (ALL). Dust samples were collected from homes of 269 ALL cases and 333 healthy controls (o8 years of age at diagnosis/reference date and residing in same home since diagnosis/reference date) in California, using a high-volume surface sampler or household vacuum bags. Amounts of agricultural or professional herbicides (alachlor, metolachlor, bromoxynil, bromoxynil octanoate, pebulate, butylate, prometryn, simazine, ethalfluralin, and pendimethalin) and residential herbicides (cyanazine, trifluralin, 2-methyl-4- chlorophenoxyacetic acid (MCPA), mecoprop, 2,4-dichlorophenoxyacetic acid (2,4-D), chlorthal, and dicamba) were measured. Odds ratios (OR) and 95% confidence intervals (CI) were estimated by logistic regression. Models included the herbicide of interest, age, sex, race/ethnicity, household income, year and season of dust sampling, neighborhood type, and residence type. The risk of childhood ALL was associated with dust levels of chlorthal; compared to homes with no detections, ORs for the first, second, and third tertiles were 1.49 (95% CI: 0.82–2.72), 1.49 (95% CI: 0.83–2.67), and 1.57 (95% CI: 0.90–2.73), respectively (P-value for linear trend ¼ 0.05). The magnitude of this association appeared to be higher in the presence of alachlor. -

Nomenclature of Commonly Available Herbicides in India
NOMENCLATURE OF COMMONLY AVAILABLE HERBICIDES IN INDIA Prior to the widespread use of chemical herbicides, mechanical control and cultural controls, such as altering soil pH, salinity, or fertility levels were used to control weeds. The first widely used herbicide was 2,4-dichlorophenoxyacetic acid, often abbreviated 2,4-D which kills many broadleaf plants while leaving grasses largely unaffected (high doses of 2,4-D at crucial growth periods can harm grass crops such as maize or cereals). The low cost of 2,4-D has led to continued usage today and it remains one of the most commonly used herbicides in the world. In 1950s triazine family of herbicides, which includes atrazine was introduced. Atrazine does not break down readily (within a few weeks) after being applied to soils of above neutral pH. Atrazine is said to have carryover, a generally undesirable property for herbicides. Glyphosate, frequently sold under the brand name Roundup, was introduced in 1974 for non- selective weed control. It is now a major herbicide in selective weed control in growing crop plants due to the development of crop plants that are resistant to it. Many modern chemical herbicides for agriculture are specifically formulated to decompose within a short period after application. This is desirable as it allows crops which may be affected by the herbicide to be grown on the land in future seasons. However, herbicides with low residual activity (i.e., that decompose quickly) often do not provide season-long weed control. List of herbicides with their common name -

Ecological Risk Assessment for Saflufenacil
TEXT SEARCHABLE DCOUMENT 2011 UNITED STATES ENVIRONMENTAL PROTECTION AGENCY WASHINGTON, D.C. 20460 OFFICE OF CEMICAL SAFETY AND POLLUTION PREVENTION PC Code: 118203 DP Barcode: 380638 and 381293 Thursday, April 07, 2011 MEMORANDUM SUBJECT: Ecological Risk Assessment for Saflufenacil Section 3 New Chemical Uses as a harvest aid on dry edible beans, dry peas, soybean, oilseeds "sunflower subgroup 20B", oilseeds "cotton subgroup 20C", and oilseeds canola "subgroup 20A". TO: Kathryn Montague, M.S., Product Manager 23 Herbicide Branch Registration Division (RD) (7505P) FROM: ~ Mohammed Ruhman, Ph.D., Agronomist 2 :4- . ""=- ........ 04!tJt! (I neith Sappington, Senior Biologist/Science Adviso~.... Vd- Environmental Risk Branch V O'f/ .../ II Environmental Fate and Effects Division (7507P) THROUGH: Mah Shamim, Ph.D., Branch Chief Environmental Risk Branch VI Environmental Fate and Effects Division (7507P) This ecological risk assessment for saflufenacil new uses is relying on the attached previous assessment (Attachment 1). As shown in the usage summary (Table 1), the single and seasonal rate, for all the crops range from 0.045 to 0.089 lbs a.i/A are within the range application rates used in exposure modeling for the 2009 Section 3 New Chemical Environmental Fate and Ecological Risk Assessment (DP Barcode 349855). Therefore, risk findings determined for the 2009 assessment may be used in the assessment for this submittal. Specifically, the 2009 assessment found no chronic risks to avian and mammalian species at an agricultural use rate 0 0.134 lb a.i.lA. Acute risks were not determined for birds and mammals since saflufenacil was not acutely toxic at the highest doses tested. -

Agricultural Biotechnology: Benefits of Transgenic Soybeans
AGRICULTURAL BIOTECHNOLOGY: BENEFITS OF TRANSGENIC SOYBEANS Leonard P. Gianessi Janet E. Carpenter April 2000 National Center for Food and Agricultural Policy 1616 P Street, NW, First Floor Washington, DC 20036 Tel: 202-328-5048 Fax: 202-328-5133 [email protected] Preparation of this report was supported financially with a grant from the Rockefeller Foundation TABLE OF CONTENTS 1. Introduction 2. U.S. Soybean Production 3. Soybean Products 4. Soybean Physiology 5. Soybeans – Agronomic Factors 6. Soybean Genetic Improvements A. Introduction B. Reproductive Process C. Artificial Cross Breeding D. Mutation Breeding E. Transgenic Plants 7. Weed Competition – Soybeans 8. Weed Control in Soybeans: 1940’s – 1950’s 9. Herbicides – An Overview 10. Herbicide Use in Soybeans: 1960’s – 1995 A. Introduction B. Historical Overview 1. The Early 1960’s 2. Soil Applied Herbicides 3. Postemergence Herbicides 4. Sulfonylurea/Imidazolinone Herbicides 5. Burndown Herbicides C. Summary of Usage: 1995 11. Transgenic Herbicide Tolerant Soybeans A. Glyphosate – An Overview B. Performance of Roundup Ready Soybeans C. Herbicide Ratings D. Adoption Impacts: 1995 – 1998 1. Herbicide Costs 2. Soybean Yields 3. Returns 4. Other Aggregate Studies 5. Herbicide Treatments 6. Herbicide Use Amounts 7. Other Impacts 12. Summary and Conclusions 13. References Appendix 1: Soybean Processing – A Description 1. Introduction Soybeans and other crops have been improved genetically for many decades through traditional crop breeding – a technique that requires that species be sexually compatible. With the development of biotechnology methods, scientists have the ability to transfer single genes from one living organism into another, regardless of species or sexual compatibility. Varieties that are developed through the transfer of genes between species that are not sexually compatible are referred to as “transgenic.” Transgenic soybean plants have been developed with a gene from a soil bacteria that allows the use of an herbicide that would normally kill soybeans. -

MC(' Potential Exposure of Humans to 2
(MC( FUNDAMENTAL AND APPLIED TOXICOLOGY 1:3 3 9-3 4 6 (1981) Potential Exposure of Humans to 2,4,5-T and TCDD in the Oregon Coast Ranges MICHAEL NEWTON" and LOGAN A. NORRISB "Professor of Forest Ecology, Oregon State University, Corvallis; BChief Research Chemist, USDA Forest Service, Corvallis, Oregon ABSTRACT Potential Exposure of Humans to 2,4,5-T and TCDD in Humans may be exposed to herbicides through drift; inges- the Oregon Coast Ranges. Newton, M. and Norris, L.A. tion of wild and domestic meat, vegetables, and fruit; con- (1981). F.undam. AppL Toxicol. 1:339-346. Research on the sumption of water; and dermal contact while handling the use of 2,4,5-trichlorophenoxyacetic acid (2,4,5-T) contami- chemicals, equipment, and treated vegetation. The range of -8 nated with 2.5 X 10 parts 2,3,7,8-tetrachlorodibenzo-p- potential exposure extends from zero, if there is no encounter dioxin (TCDD) in forests of the Oregon Coast Ranges per- with the herbicide, to the worst situation where the person has mits estimates of human exposures for both compounds. encountered the highest levels of water contamination, drift Estimated total exposure of nearby ( ^ 1/8 mile distant) resi- exposure, meat contamination, and dermal exposure simul- dents during the first week after application is 0.0039 mg/kg taneously. We have brought estimates of all sources together of 2,4,5-T for a 70-kg adult. Exposure to TCDD in the same to determine the possible range of total exposure from episode would be 1.9 X 10 b ° mg/kg. -

INDEX to PESTICIDE TYPES and FAMILIES and PART 180 TOLERANCE INFORMATION of PESTICIDE CHEMICALS in FOOD and FEED COMMODITIES
US Environmental Protection Agency Office of Pesticide Programs INDEX to PESTICIDE TYPES and FAMILIES and PART 180 TOLERANCE INFORMATION of PESTICIDE CHEMICALS in FOOD and FEED COMMODITIES Note: Pesticide tolerance information is updated in the Code of Federal Regulations on a weekly basis. EPA plans to update these indexes biannually. These indexes are current as of the date indicated in the pdf file. For the latest information on pesticide tolerances, please check the electronic Code of Federal Regulations (eCFR) at http://www.access.gpo.gov/nara/cfr/waisidx_07/40cfrv23_07.html 1 40 CFR Type Family Common name CAS Number PC code 180.163 Acaricide bridged diphenyl Dicofol (1,1-Bis(chlorophenyl)-2,2,2-trichloroethanol) 115-32-2 10501 180.198 Acaricide phosphonate Trichlorfon 52-68-6 57901 180.259 Acaricide sulfite ester Propargite 2312-35-8 97601 180.446 Acaricide tetrazine Clofentezine 74115-24-5 125501 180.448 Acaricide thiazolidine Hexythiazox 78587-05-0 128849 180.517 Acaricide phenylpyrazole Fipronil 120068-37-3 129121 180.566 Acaricide pyrazole Fenpyroximate 134098-61-6 129131 180.572 Acaricide carbazate Bifenazate 149877-41-8 586 180.593 Acaricide unclassified Etoxazole 153233-91-1 107091 180.599 Acaricide unclassified Acequinocyl 57960-19-7 6329 180.341 Acaricide, fungicide dinitrophenol Dinocap (2, 4-Dinitro-6-octylphenyl crotonate and 2,6-dinitro-4- 39300-45-3 36001 octylphenyl crotonate} 180.111 Acaricide, insecticide organophosphorus Malathion 121-75-5 57701 180.182 Acaricide, insecticide cyclodiene Endosulfan 115-29-7 79401 -
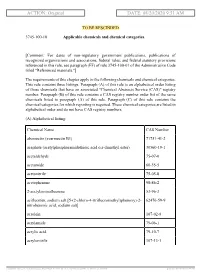
ACTION: Original DATE: 08/20/2020 9:51 AM
ACTION: Original DATE: 08/20/2020 9:51 AM TO BE RESCINDED 3745-100-10 Applicable chemicals and chemical categories. [Comment: For dates of non-regulatory government publications, publications of recognized organizations and associations, federal rules, and federal statutory provisions referenced in this rule, see paragraph (FF) of rule 3745-100-01 of the Administrative Code titled "Referenced materials."] The requirements of this chapter apply to the following chemicals and chemical categories. This rule contains three listings. Paragraph (A) of this rule is an alphabetical order listing of those chemicals that have an associated "Chemical Abstracts Service (CAS)" registry number. Paragraph (B) of this rule contains a CAS registry number order list of the same chemicals listed in paragraph (A) of this rule. Paragraph (C) of this rule contains the chemical categories for which reporting is required. These chemical categories are listed in alphabetical order and do not have CAS registry numbers. (A) Alphabetical listing: Chemical Name CAS Number abamectin (avermectin B1) 71751-41-2 acephate (acetylphosphoramidothioic acid o,s-dimethyl ester) 30560-19-1 acetaldehyde 75-07-0 acetamide 60-35-5 acetonitrile 75-05-8 acetophenone 98-86-2 2-acetylaminofluorene 53-96-3 acifluorfen, sodium salt [5-(2-chloro-4-(trifluoromethyl)phenoxy)-2- 62476-59-9 nitrobenzoic acid, sodium salt] acrolein 107-02-8 acrylamide 79-06-1 acrylic acid 79-10-7 acrylonitrile 107-13-1 [ stylesheet: rule.xsl 2.14, authoring tool: RAS XMetaL R2_0F1, (dv: 0, p: 185720, pa: -

Reregistration Eligibility Decision (RED) for Glyphosate
GLYPHOSATE RED September 1993 GLYPHOSATE REREGISTRATION ELIGIBILITY TEAM Office of Pesticide Programs: Special Review and Reregistration Division Eric Feris .................................................. Reregistration Branch Health Effects Division Jane Smith ........................................ Chemical Coordination Branch Krystyna Locke .............................................. Toxicology Branch I Jeff Evans ........................... Occupational and Residential Exposure Branch Randolph Perfetti ......................... Chemistry Branch - Reregistration Support Biological and Economic Analysis Division James G. Saulmon .....................................Biological Analysis Branch Eric Maurer ........................................... Economic Analysis Branch Environmental Fate and Effects Division Candace Brassard ..................................... Ecological Effects Branch Kevin Poff ............................. Environmental Fate and Groundwater Branch Bernice Slutsky ............................ Science Analysis and Coordination Staff Registration Division Mark Perry .......................................... Registration Support Branch Karen P. Hicks ....................................... Fungicide-Herbicide Branch Policy and Special Projects Staff Jean Frane ............................. Food Safety & Regulation Tracking Section Office of General Counsel: Pesticides and Toxic Substances Division Debra Burton ................................................. Pesticides Branch Office of Compliance Monitoring: -

Toxicological Profile for Glyphosate Were
A f Toxicological Profile for Glyphosate August 2020 GLYPHOSATE II DISCLAIMER Use of trade names is for identification only and does not imply endorsement by the Agency for Toxic Substances and Disease Registry, the Public Health Service, or the U.S. Department of Health and Human Services. GLYPHOSATE III FOREWORD This toxicological profile is prepared in accordance with guidelines developed by the Agency for Toxic Substances and Disease Registry (ATSDR) and the Environmental Protection Agency (EPA). The original guidelines were published in the Federal Register on April 17, 1987. Each profile will be revised and republished as necessary. The ATSDR toxicological profile succinctly characterizes the toxicologic and adverse health effects information for these toxic substances described therein. Each peer-reviewed profile identifies and reviews the key literature that describes a substance's toxicologic properties. Other pertinent literature is also presented, but is described in less detail than the key studies. The profile is not intended to be an exhaustive document; however, more comprehensive sources of specialty information are referenced. The focus of the profiles is on health and toxicologic information; therefore, each toxicological profile begins with a relevance to public health discussion which would allow a public health professional to make a real-time determination of whether the presence of a particular substance in the environment poses a potential threat to human health. The adequacy of information to determine a substance's -

US EPA, Pesticide Product Label, SHARDA GLYPHOSATE 20.5 % +
86(19,5210(17$/3527(&7,21$*(1&< (3$5HJ1XPEHU 'DWHRI,VVXDQFH 2IILFHRI3HVWLFLGH3URJUDPV 5HJLVWUDWLRQ'LYLVLRQ 3 3HQQV\OYDQLD$YH1: :DVKLQJWRQ'& 127,&(2)3(67,&,'( 7HUPRI,VVXDQFH ;5HJLVWUDWLRQ 5HUHJLVWUDWLRQ &RQGLWLRQDO XQGHU),)5$DVDPHQGHG 1DPHRI3HVWLFLGH3URGXFW 6KDUGD*O\SKRVDWH 0HWRODFKORU0HVRWULRQH =& 1DPHDQG$GGUHVVRI5HJLVWUDQW LQFOXGH=,3&RGH $QQD$UPVWURQJ 6KDUGD86$//& 32%R[ +RFNHVVLQ'( 1RWH&KDQJHVLQODEHOLQJGLIIHULQJLQVXEVWDQFHIURPWKDWDFFHSWHGLQFRQQHFWLRQZLWKWKLVUHJLVWUDWLRQPXVWEHVXEPLWWHGWRDQGDFFHSWHGE\WKH 5HJLVWUDWLRQ'LYLVLRQSULRUWRXVHRIWKHODEHOLQFRPPHUFH,QDQ\FRUUHVSRQGHQFHRQWKLVSURGXFWDOZD\VUHIHUWRWKHDERYH(3$UHJLVWUDWLRQQXPEHU 2QWKHEDVLVRILQIRUPDWLRQIXUQLVKHGE\WKHUHJLVWUDQWWKHDERYHQDPHGSHVWLFLGHLVKHUHE\UHJLVWHUHG XQGHUWKH)HGHUDO,QVHFWLFLGH)XQJLFLGHDQG5RGHQWLFLGH$FW 5HJLVWUDWLRQLVLQQRZD\WREHFRQVWUXHGDVDQHQGRUVHPHQWRUUHFRPPHQGDWLRQRIWKLVSURGXFWE\WKH $JHQF\,QRUGHUWRSURWHFWKHDOWKDQGWKHHQYLURQPHQWWKH$GPLQLVWUDWRURQKLVPRWLRQPD\DWDQ\ WLPHVXVSHQGRUFDQFHOWKHUHJLVWUDWLRQRIDSHVWLFLGHLQDFFRUGDQFHZLWKWKH$FW7KHDFFHSWDQFHRIDQ\ QDPHLQFRQQHFWLRQZLWKWKHUHJLVWUDWLRQRIDSURGXFWXQGHUWKLV$FWLVQRWWREHFRQVWUXHGDVJLYLQJWKH UHJLVWUDQWDULJKWWRH[FOXVLYHXVHRIWKHQDPHRUWRLWVXVHLILWKDVEHHQFRYHUHGE\RWKHUV 7KLVSURGXFWLVFRQGLWLRQDOO\UHJLVWHUHGLQDFFRUGDQFHZLWK),)5$VHFWLRQ F $ <RXPXVWFRPSO\ ZLWKWKHIROORZLQJFRQGLWLRQV 6XEPLWDQGRUFLWHDOOGDWDUHTXLUHGIRUUHJLVWUDWLRQUHUHJLVWUDWLRQUHJLVWUDWLRQUHYLHZRI\RXU SURGXFWXQGHU),)5$ZKHQWKH$JHQF\UHTXLUHVDOOUHJLVWUDQWVRIVLPLODUSURGXFWVWRVXEPLWVXFK GDWD 6LJQDWXUHRI$SSURYLQJ2IILFLDO 'DWH (PLO\6FKPLG$FWLQJ3URGXFW0DQDJHU