Classification of Red Wines Produced from Zweigelt and Rondo Grape
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

1.2 Weingartenflächen Und Flächenanteile Der Rebsorten EN
1. Vineyard areas and areas under vine by grape variety Austrian Wine statistics report 1.2 Vineyard areas and areas under vine by grape variety 2 The data in this section is based on the 2015 Survey of Area under Vine, as well as feedback from the wine-producing federal states of Niederösterreich (Lower Austria), Burgenland, Steiermark (Styria) and Wien (Vienna). The main source of data for the 2015 Survey of Area under Vine was the Wein-ONLINE system operated by the Federal Ministry of Agriculture, Forestry, Environment and Water Management (BMNT). Data from the remaining federal states was collected by means of a questionnaire (primary data collection). Information relating to the (approved) nurseries was provided by the Burgenland and Lower Austrian Chambers of Agriculture and the Styrian state government (Agricultural Research Centre). According to the 2015 Survey of Area under Vine, Austria’s vineyards occupied 45,574 hectares. The planted vineyard area was 45,439 ha, which corresponds to 94 ha less (or a 0.2% decrease) in comparison to the 2009 Survey of Area under Vine. The long-running trend that suggested a shift away from white wine and towards red was quashed by the 2015 Survey of Area under Vine. While the white wine vineyard area increased by 2.3% to 30,502 ha compared to 2009, the red wine area decreased by 4.9% to 14,937 ha. Figure 1 shows the evolution of Austrian viticulture after the Second World War. The largest area under vine was recorded in 1980 at 59,432 ha. From 1980 onwards, the white wine vineyard area has continuously decreased, while the red wine vineyard area has expanded. -

German Red Wines – Steve Zins 11/12/2014 Final Rev 5.0 Contents
German Red Wines – Steve Zins 11/12/2014 Final Rev 5.0 Contents • Introduction • German Wine - fun facts • German Geography • Area Classification • Wine Production • Trends • Permitted Reds • Wine Classification • Wine Tasting • References Introduction • Our first visit to Germany was in 2000 to see our daughter who was attending college in Berlin. We rented a car and made a big loop from Frankfurt -Koblenz / Rhine - Black forest / Castles – Munich – Berlin- Frankfurt. • After college she took a job with Honeywell, moved to Germany, got married, and eventually had our first grandchild. • When we visit we always try to visit some new vineyards. • I was surprised how many good red wines were available. So with the help of friends and family we procured and carried this collection over. German Wine - fun facts • 90% of German reds are consumed in Germany. • Very few wine retailers in America have any German red wines. • Most of the largest red producers are still too small to export to USA. • You can pay $$$ for a fine French red or drink German reds for the entire year. • As vineyard owners die they split the vineyards between siblings. Some vineyards get down to 3 rows. Siblings take turns picking the center row year to year. • High quality German Riesling does not come in a blue bottle! German Geography • Germany is 138,000 sq mi or 357,000 sq km • Germany is approximately the size of Montana ( 146,000 sq mi ) • Germany is divided with respect to wine production into the following: • 13 Regions • 39 Districts • 167 Collective vineyard -

Leo Hillinger
Vineyards: 90+ hectares (222+ acres) www.kwselection.com/wineries/leohillinger.pdf Varieties: Grüner Veltliner, Welschriesling, Chardonnay, Pinot Gris, Sauvignon Blanc, Gelber Muskateller, Zweigelt, Blaufränkisch, St. Laurent, Merlot, Syrah, Cabernet Sauvignon and Pinot Noir Wine Region Burgenland: It is a unique wine-producing region in an Austrian context as the most recognizable wines produced here are based on white and red varieties almost at same level. On top, sweet wines are produced based on the naturally occuring Botrytis Cinerea. This region produces by far the biggest amount of red wine in Austria. This is the reason why it is also unofficially called the red wine quarter of the country. The winery Hillinger is in the north of Burgenland, between the Leithaberg and the lake Neusiedl. This subregion is known for it’s unique whites and sweets. This can be attributed to the plentiful sunshine and the cooler nights the region experiences which averages of more than 2000 hours per year Soil and Climate: The primary rock is slate and lime. The perfect vineyards for our Pinot’s are limestone and brown soil and for Blaufränkisch on lime. Hot, dry summers and cold winters express fruitiness and retain acidity in the wines combined with the temperature-regulating influence of the lake Neusiedl, guarantees long vegetation periods for full ripeness of grapes. High humidity and autumn fog and drying sun during the day helps to develop botrytis cinerea, which forms the basis for our top quality sweet wines, such as TBA Small Hill Sweet and TBA Hill 3. The success story of the Leo Hillinger wine-growing estate...With a 90+-hectare area under cultivation in Austria, the Hillinger wine-growing estate is one of the largest high-quality wine producers in Austria. -

A to Z Glossary of Wine Terms
A to Z Glossary of Wine Terms A ABV (Alcohol by Volume) - A measure of the alcohol concentration of a wine, usually expressed as a percentage. Acidic - A tart, sour, or fresh feeling in the mouth when you taste the wine. Aeration - Adding oxygen to the wine to soften tannins. Aggressive – Wine tasting term describing a wine that is either too tannic, too acidic, or a combina- tion of both. Alcohol - Ethanol; in wine it is produced via fermentation with yeast and sugar. Alcoholic – Wine tasting term indicating high alcohol. A wine with a noticeably high alcohol con- tent; perceived as a hotness in the wine. Angular – Wine tasting term used to describe young wines that display predominately sharp, bitter, or tart flavor characteristics. AOC (Appellation d’origine contrôlée) - A term in the French wine designation system which means “controlled designation of origin.” It identifies the location or region where a wine is made. Appearance - In general, the term appearance is used to describe the clarity of a wine. Appellation - A designated wine growing area governed by specific rules regarding the wine grapes grown and wine produced in the specific appellation areas. Aroma - A wine’s scent characteristics; very closely tied in with the flavors. Aromatic - Varieties of grapes that have especially noticeable aromas. Some aromatic grapes in- clude Viognier, Torrontés, Gewürztraminer, Riesling, Muscat, and Pinot Gris. Aromatized – A wine that is infused with botanicals. Vermouth is an aromatized wine. Assemblage - A method of blending wine before bottling. Astringent – A wine tasting term meaning the wine leaves the mouth feeling overwhelmingly dry. -
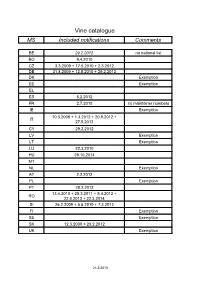
Vine Catalogue MS Included Notifications Comments
Vine catalogue MS Included notifications Comments BE 29.2.2012 no national list BG 9.4.2010 CZ 3.3.2009 + 17.5.2010 + 2.3.2012 DE 31.8.2009 + 12.5.2010 + 29.2.2012 DK Exemption EE Exemption EL ES 8.2.2012 FR 2.7.2010 no maintainer numbers IE Exemption 10.3.2008 + 1.3.2012 + 20.9.2012 + IT 27.5.2013 CY 29.2.2012 LV Exemption LT Exemption LU 22.3.2010 HU 29.10.2014 MT NL Exemption AT 2.2.2012 PL Exemption PT 28.2.2012 13.4.2010 + 25.3.2011 + 5.4.2012 + RO 22.4.2013 + 22.3.2014 SI 26.2.2009 + 5.5.2010 + 7.3.2012 FI Exemption SE Exemption SK 12.3.2009 + 20.2.2012 UK Exemption 21.5.2015 Common catalogue of varieties of vine 1 2 3 4 5 Known synonyms Variety Clone Maintainer Observations in other MS A Abbuoto N. IT 1 B, wine, pas de Abondant B FR matériel certifiable Abouriou B FR B, wine 603, 604 FR B, wine Abrusco N. IT 15 Accent 1 Gm DE 771 N Acolon CZ 1160 N We 725 DE 765 B, table, pas de Admirable de Courtiller B FR matériel certifiable Afuz Ali = Regina Agiorgitiko CY 163 wine, black Aglianico del vulture N. I – VCR 11, I – VCR 14 IT 2 I - Unimi-vitis-AGV VV401, I - Unimi-vitis- IT 33 AGV VV404 I – VCR 7, I – VCR 2, I – Glianica, Glianico, Aglianico N. VCR 13, I – VCR 23, I – IT 2 wine VCR 111, I – VCR 106, I Ellanico, Ellenico – VCR 109, I – VCR 103 I - AV 02, I - AV 05, I - AV 09, I - BN 2.09.014, IT 31 wine I - BN 2.09.025 I - Unimi-vitis-AGT VV411, I - Unimi-vitis- IT 33 wine AGTB VV421 I - Ampelos TEA 22, I - IT 60 wine Ampelos TEA 23 I - CRSA - Regione Puglia D382, I - CRSA - IT 66 wine Regione Puglia D386 Aglianicone N. -

Weissweine Aus Der Bodenseeregion
WEISSWEINE AUS DER BODENSEEREGION Weine vom Weingut Möth in Bregenz Die besten und größten Lagen befinden sich in unmittelbarer Nähe zum Bodensee in der Lage Neu Amerika. Hier profitiert man von den klimatischen Gegebenheiten des großen Sees (Wärmespeicher, Reflexionswärme, Fön). Die Reben stehen auf sandigem Kiesboden. Mit etwa 3,5 ha ist dieser Weinbaubetrieb der größte und auch einzige, der als Haupterwerb in Vorarlberg geführt wird. Chardonnay 2018 34.00 Im Duft leicht rauchig, würzig, am Gaumen cremig, mineralisch, mit zarter Säuretextur. Seebrünzler 2018 32.00 Duftig, fruchtige Nase nach Pfirsich und Muskat. Am Gaumen Fruchtsüsse mit weicher Säure unterlegt. Weingut Aufricht in Stetten Das Weingut Aufricht ist ein privates Weingut der Brüder Robert und Manfred Aufricht im Weinanbaugebiet Baden mit Sitz in Stetten am Bodensee. Es bewirtschaftet eine Ertragsrebfläche von 35 Hektar und produziert von 250.000 bis 300.000 Flaschen Wein. Auxerrois 2018 35.00 Faszinierende gelbe Steinfrucht, cremig unterlegt von einem dezenten, weichen Pinot-Ton. Angenehm wenig Säure. Winzerverein Hagnau Der Winzerverein Hagnau ist die älteste Winzergenossenschaft im Weinanbaugebiet Baden mit Sitz in Hagnau am Bodensee. Er bewirtschaftet eine Ertragsrebfläche von 166 Hektar. Müller Thurgau „Felchen“ 2017 | 2018 27.00 Hagnauer Burgstall WEISSWEINE AUS ÖSTERREICH In allen österreichischen Weinbauregionen gehen kleine, aber auch größere Winzer den Weg der kompromisslosen Qualität und finden damit großen Anklang. Einige der Besten davon haben wir für Sie ausgesucht. -

7 Elements Introduction to Austrian Wine
7 ELEMENTS INTRODUCTION TO AUSTRIAN WINE © AWMB SEVEN ELEMENTS OF UNIQUENESS Key Facts © AWMB/Philipp Forster AUSTRIAN VINEYARD AREA IN 2015 Total: 46,515 ha / 115.000 acres BASIC FACTS • Production: 250 million litres • Consumption: 250 million litres • Import: 50 – 70 million litres • Export: 50 – 70 million litres AUSTRIA‘S BOOMING WINE EXPORTS Value: € 170 million Volume: 53 million litres Ø Price: 3.24 Euro/liter Source: Statistics Austria, preliminary export figures I-XII 2018 (as of March 2019). The data capture method used by Statistics Austria also includes re-exports of non-Austrian wine. 1995 no data available. AUSTRIAN WINE EXPORTS: BOTTLE VS. BULK 65.000 60.000 55.000 50.000 45.000 40.000 35.000 30.000 25.000 inL 1.000 20.000 15.000 10.000 5.000 0 2000 2001 2002 2003 2011 2012 2013 2014 2015 2016 2017 2004 2005 2006 2007 2008 2009 2010 MengeBottles Flasche MengeBulk Fass AUSTRIA‘S TOP 10 EXPORT MARKETS (REVENUE) Source: Statistik Austria, preliminary export numbers for 2018; March 2019 1. THE CLIMATE 2. THE LAND 3. THE GRAPES 4. THE CULTURE 5. NATURE 6. VALUE FOR MONEY 7. THE TASTE © AWMB/Philipp Forster 1. THE CLIMATE 2. THE LAND 3. THE GRAPES 4. THE CULTURE 5. NATURE 6. VALUE FOR MONEY 7. THE TASTE © AWMB/Anna Stöcher ([email protected]) THE TENSION OF OPPOSITES Skiing in the West Wine in the East © AWMB © AWMB THE TENSION OF OPPOSITES 3 2 1 1. Continental-pannonian 2. Temperate atlantic 4 3. Cool air from the north 4. -

C Ertificate 2020 Weinbau Gruber OG
Cert. No: 2-00186-2021 Weinbau Gruber OG Winzerstraße 46, 3743 Röschitz National farm No: 4284721 The above mentioned operator with the main activity "agricultural production" has been in possession of a valid inspection contract with Austria Bio Garantie GmbH since 2013-09-07. On the basis of the inspection conducted on 2020-08-12 the operator is certified according to the following rules: • Council Regulation (EC) No 834/2007 and its implementing rules, as amended • Guideline Agricultural Products from Organic Production and Derived Products, as amended This document has been issued on the basis of Article 29(1) of Regulation (EC) No 834/2007 and of Regulation (EC) No 889/2008. The declared operator has submitted his activities under control, and meets the requirements laid down in the named Regulations. The operator's products listed below may be declared as follows: Product(group): Declaration: Plant production: field bean (1,3423 ha) organic product green fallow and fallow land (5,0827 ha) conventional product vegetated not used area (0,1769 ha) conventional product grapes (1,0667 ha) plots 36, 183, 184 conventional product Grüner Veltliner Certificate 2020 Certificate grapes (1,2828 ha) plot 1 young plants without in-conversion product (1) yield Pinot Noir grapes (1,0864 ha) plot 10 young plants in-conversion product (1) without yield Rhine Riesling grapes (0,2165 ha) plot 171 in-conversion product (1) Müller Thurgau grapes (2,9046 ha) plots 6, 113 young plants in-conversion product (1) without yield Grüner Veltliner During the named period, this certificate retains Validity of the certificate from 2021-01-08: validity only if: Plant products: harvest 2020, not later than 2022-01-31 a) The products must be in accordance with the named regulations. -

Technical Specifications for Registration of Geographical Indications
TECHNICAL SPECIFICATIONS FOR REGISTRATION OF GEOGRAPHICAL INDICATIONS NAME OF GEOGRAPHICAL INDICATION Franken PRODUCT CATEGORY Wine, Quality sparkling wine, Semi-sparkling wine, Liqueur wine, Partially fermented grape must COUNTRY OF ORIGIN Germany APPLICANT Bundesland Bayern Bayerisches Staatsministerium fürErnährung, Landwirtschaft und Forsten 2 Ludwigstraße 80539 München Deutschland/Germany Tel: +49 (0) 89 2182 0 Fax: +49 (0) 89 2182 2714 [email protected] PROTECTION IN COUNTRY OF ORIGIN Date of protection in the European Union: 18/09/1973 Date of protection in the Member State: Anordnung des Reichsnährstandes für Wein of 7 January 1936 (RNVBl. p. 17) PRODUCT DESCRIPTION Raw material Most significant vine varieties: Gutedel Regent Blauer Limberger Schwarzriesling Blauer Trollinger Kerner Weißer Burgunder Blauer Portugieser Ruländer Grüner Silvaner Dornfelder Blauer Spätburgunder Müller Thurgau Weißer Riesling Alcohol content Wine with special Partially Liqueur attributes fermented wine Wine („Beerenauslese“, grape must (Qualitäts- (Qualitäts- „Trockenbeeren- (Federweißer) likörwein) wein) auslese“ und „Eiswein“) Minimum alcohol 7.0 5.5 1.0 15.0 content (% vol.) Physical appearance o Franken white (colour: yellowish green to golden yellow) o Franken red (colour: light red to brick red, ruby red, garnet red, purple, bluish to dark violet, also brownish tinges) o Franken Blanc de noir (colour: colourless to yellowish or golden yellow tinges) o Franken Rotling/Franken rosé: (colour: light red to pale red, in some cases with bluish tinges) DESCRIPTION OF GEOGRAPHICAL AREA The Franken winegrowing area is located in northern Bavaria and is bordered by the upland areas of Spessart, Odenwald, Rhön, Hassberge and Steigerwald. The River Main provides the drainage system for the whole area and is at the same time a prominent feature of the region. -

Phenolic Compounds As Markers of Wine Quality and Authenticity
foods Review Phenolic Compounds as Markers of Wine Quality and Authenticity Vakare˙ Merkyte˙ 1,2 , Edoardo Longo 1,2,* , Giulia Windisch 1,2 and Emanuele Boselli 1,2 1 Faculty of Science and Technology, Free University of Bozen-Bolzano, Piazza Università 5, 39100 Bozen-Bolzano, Italy; [email protected] (V.M.); [email protected] (G.W.); [email protected] (E.B.) 2 Oenolab, NOI Techpark South Tyrol, Via A. Volta 13B, 39100 Bozen-Bolzano, Italy * Correspondence: [email protected]; Tel.: +39-0471-017691 Received: 29 October 2020; Accepted: 28 November 2020; Published: 1 December 2020 Abstract: Targeted and untargeted determinations are being currently applied to different classes of natural phenolics to develop an integrated approach aimed at ensuring compliance to regulatory prescriptions related to specific quality parameters of wine production. The regulations are particularly severe for wine and include various aspects of the viticulture practices and winemaking techniques. Nevertheless, the use of phenolic profiles for quality control is still fragmented and incomplete, even if they are a promising tool for quality evaluation. Only a few methods have been already validated and widely applied, and an integrated approach is in fact still missing because of the complex dependence of the chemical profile of wine on many viticultural and enological factors, which have not been clarified yet. For example, there is a lack of studies about the phenolic composition in relation to the wine authenticity of white and especially rosé wines. This review is a bibliographic account on the approaches based on phenolic species that have been developed for the evaluation of wine quality and frauds, from the grape varieties (of V. -

Irrigation and Leaf Removal Effects on Polyphenolic Content of Grapes and Wines Produced from Cv
Available online: www.notulaebotanicae.ro Print ISSN 0255-965X; Electronic 1842-4309 Not Bot Horti Agrobo, 2016, 44(1):133-139. DOI:10.15835/nbha44110254 Original Article Irrigation and Leaf Removal Effects on Polyphenolic Content of Grapes and Wines Produced from cv. ‘Agiorgitiko’ (Vitis vinifera L.) Evangelia CHORTI1, Maria KYRALEOU2, Stamatina KALLITHRAKA2, Manolis PAVLIDIS2, Stefanos KOUNDOURAS3, Ioannis KANAKIS4, Yorgos KOTSERIDIS2* 1Cooperative Winery of Nemea, 130 Papaconstantinou av., 20500 Nemea, Greece; [email protected] 2Agricultural University of Athens, Department of Food Science & Technology, 75 Iera Odos, 11855 Athens, Greece ; [email protected]; [email protected]; [email protected]; [email protected] (*corresponding author) 3Aristotle University of Thessaloniki, School of Agriculture, Laboratory of Viticulture, 54124, Thessaloniki, Greece; [email protected] 4Fassoulis Grapevine Nurseries, 20500 Leontio - Nemea, Greece; [email protected] Abstract Vitis vinifera L. cv. ‘Agiorgitiko’ is one of the most important red grape varieties of Greece, cultivated almost exclusively in the region of Nemea in north-eastern Peloponnese. This work aimed to study the influence of some commonly applied viticultural practices on the polyphenolic composition of ‘Agiorgitiko’. Leaf removal at veraison, irrigation, and a combination of both, were applied and the phenolic content of the grapes and of the produced wines was compared. The results showed that leaf removal decreased berry size, enhanced total anthocyanin, total phenol and malvidin 3-O-monoglucoside accumulation in skins and increased the amount of extractable anthocyanins in the juice. The combination of irrigation and leaf removal caused a significant increase in total phenols in the skin and in the amount of extractable anthocyanins in juice. -

The Effect of Grape Ripening Stage on Red Wine Color
THE EFFECT OF GRAPE RIPENING STAGE ON RED WINE COLOR EFFET DE LA MATURATION SUR LA COULEUR DES VINS ROUGES A.B. BAUTISTA-ORTÍN1, J.I. FERNÁNDEZ-FERNÁNDEZ2, J.M. LÓPEZ-ROCA1 and E. GÓMEZ-PLAZA*1 1 : Departamento de Tecnología de Alimentos, Nutrición y Bromatología, Facultad de Veterinaria, Universidad de Murcia. Campus de Espinardo, 30071 Murcia, Espagne 2 : I.M.I.D.A. Ctra. La Alberca s/n, 30150 Murcia, Espagne Abstract : The physico-chemical and chromatic characteristics of grapes (Vitis vinifera L. cv. Monastrell) harvested at six dif- ferent degree of ripeness (from August 16 to October 24, 2002) and that of the wines obtained from these grapes have been stu- died. The grape anthocyanins content (mg/kg of berry fresh weight) was maximum in those grapes harvested on September 11 and 16 (804.1 and 822.6 mg/kg, respectively) and decreased for grapes harvested in October. However, the results showed that the grapes with the highest anthocyanin concentration did not lead to the highest colored wines. The wines elaborated from grapes harvested on October 16 (671.9 mg of anthocyanins per kg of berry fresh weight) had the best chromatic characteristics and better withstood aging in the bottle; the extent of cell wall degradation in overly matured grapes probably facilitated the extraction of phenolic compounds from skins. However, the chromatic quality of wines made from grapes harvested one week later (October 24, the most mature grapes) was lower than that from October 16, with lower color intensity (13 % lower in the wine elaborated from grapes harvested in October 24) and a percentage of yellow color 6 % higher in this wine.