Performance Evaluation of a Halogen Acid Gas Analyzer
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

MDA Scientific SPM
MDA Scientific SPM Fast response monitor for the detection of a target gas SPM Advantages • Fast response monitor specific to target gas only • Gas sensitivity to ppb levels with physical evidence • Minimum maintenance and no dynamic calibration • Customized for harsh industrial environments • More than 50 gas calibrations available Applications • Outdoor locations • Corrosive areas • Remote sampling areas • Gas storage areas • Survey work • Perimeter/fencelines • Ventilation and exhaust systems Options • Z-purge system • Duty cycle • ChemKey™ • RS422 • Remote reset • Portable • Extended sample • Heater option (operate from -20°C to ±40°C) Technical Specification Specifications Detection Technique Chemcassette® Detection System Alarm Point Dual level alarms typically set at 1/2 TLV and TLV Response Time As fast as 10 seconds Alarm Indication Local audio/visual alarms; remote capability optional Signal Outputs SPDT concentration alarm relays; SPDT fault relay; 4-20 mA; digital display Relay Rating 120VAC@10amps; 240VAC@5amps; 48VDC@5amps Operating Temperature Range 32° to 104°F; 0° to 40°C (basic unit); heating/cooling optional Power Requirements 115/230 VAC 50/60 Hz, battery operation optional Enclosure NEMA 4X fiberglass (basic unit) Dimensions 12"(H) x 12"(W) x 7”(D) (30.5 x 30.5 x 17.8 cm) (basic unit) Weight Up to 25 pounds, depending on option installed Note: options may vary the specifications Detectable Gases Range Amines Ammonia (NH3) 2.6-75.0 ppm Ammonia (NH3)-II 2.6-75.0 ppm Dimethylamine (DMA) 0.1-6 ppm n-Butylamine (n-BA) 0.4-12 -

Vibrationally Excited Hydrogen Halides : a Bibliography On
VI NBS SPECIAL PUBLICATION 392 J U.S. DEPARTMENT OF COMMERCE / National Bureau of Standards National Bureau of Standards Bldg. Library, _ E-01 Admin. OCT 1 1981 191023 / oO Vibrationally Excited Hydrogen Halides: A Bibliography on Chemical Kinetics of Chemiexcitation and Energy Transfer Processes (1958 through 1973) QC 100 • 1X57 no. 2te c l !14 c '- — | NATIONAL BUREAU OF STANDARDS The National Bureau of Standards' was established by an act of Congress March 3, 1901. The Bureau's overall goal is to strengthen and advance the Nation's science and technology and facilitate their effective application for public benefit. To this end, the Bureau conducts research and provides: (1) a basis for the Nation's physical measurement system, (2) scientific and technological services for industry and government, (3) a technical basis for equity in trade, and (4) technical services to promote public safety. The Bureau consists of the Institute for Basic Standards, the Institute for Materials Research, the Institute for Applied Technology, the Institute for Computer Sciences and Technology, and the Office for Information Programs. THE INSTITUTE FOR BASIC STANDARDS provides the central basis within the United States of a complete and consistent system of physical measurement; coordinates that system with measurement systems of other nations; and furnishes essential services leading to accurate and uniform physical measurements throughout the Nation's scientific community, industry, and commerce. The Institute consists of a Center for Radiation Research, an Office of Meas- urement Services and the following divisions: Applied Mathematics — Electricity — Mechanics — Heat — Optical Physics — Nuclear Sciences" — Applied Radiation 2 — Quantum Electronics 1 — Electromagnetics 3 — Time 3 1 1 and Frequency — Laboratory Astrophysics — Cryogenics . -

Influence of Water on Conductivity of Aluminium Halide Solutions in Acetonitrile
Influence of water on conductivity of aluminium halide solutions in acetonitrile L. LUX Department of Chemistry, Faculty of Metallurgy, Technical University, CS-043 85 Košice Received 7 September 1981 Accepted for publication 8 February 1982 Equilibria of the substitution and association reactions of AICI3 and AlBr3 with acetonitrile were investigated by measuring the conductivity of solutions at varying concentrations of A1C13, AlBr3, and water in the acetonitrile solution. Both halides behave in acetonitrile in the absence of water as relatively strong electrolytes. It has been revealed that water displaces acetoni trile from the solvate sphere of cation of both halides. As for AlBr3, the bromide ion is also displaced from the coordination sphere of aluminium and replaced by a neutral water molecule, which simultaneously results in an increased number of ionized particles in solution. The hydrate A1X3 -6H20 was identified as final product which separates from the solution by adding water. Изучены равновесия реакций замещения и ассоциации А1С13 и А1Вг3 с ацетонитрилом посредством измерения электропроводности раствора в зависимости от концентрации А1С13, А1Вг3, как и в зависимости от концентрации воды в растворе ацетонитрила. Оба галогенида ведут себя в ацетонитриле как сравнительно сильные электролиты в отсутствии воды. Обнаружено, что вода вытесняет ацетонитрил из сольватной обо лочки катиона у обоих галогенидов. У А1Вг3 доходит и к вытеснению иона брома из координационной сферы алюминия и его замещению нейтраль ной молекулой воды, что одновременно приводит к увеличению числа ионизированных частиц в растворе. Конечным продуктом, выделенным из раствора при добавлении воды был идентифицированный гидрат А1Х3-6Н20. Acetonitrile is a typical aprotic solvent which is widely used in electrochemical research because of its profitable properties, i.e. -

Thursday 10 January 2019
Please check the examination details below before entering your candidate information Candidate surname Other names Pearson Edexcel Centre Number Candidate Number International Advanced Level Thursday 10 January 2019 Afternoon (Time: 1 hour 40 minutes) Paper Reference WCH04/01 Chemistry Advanced Unit 4: General Principles of Chemistry I – Rates, Equilibria and Further Organic Chemistry (including synoptic assessment) Candidates must have: Scientific calculator Total Marks Data Booklet Instructions • Use black ink or black ball-point pen. • Fill in the boxes at the top of this page with your name, centre number and candidate number. • Answer all questions. • Answer the questions in the spaces provided – there may be more space than you need. Information • The total mark for this paper is 90. • The marks for each question are shown in brackets – use this as a guide as to how much time to spend on each question. • Questions labelled with an asterisk (*) are ones where the quality of your written communication will be assessed – you should take particular care with your spelling, punctuation and grammar, as well as the clarity of expression, on these questions. • A Periodic Table is printed on the back cover of this paper. Advice • Read each question carefully before you start to answer it. • Show all your working in calculations and include units where appropriate. • Check your answers if you have time at the end. Turn over P54560A ©2019 Pearson Education Ltd. *P54560A0128* 2/1/1/1/1/1/ SECTION A Answer ALL the questions in this section. You should aim to spend no more than 20 minutes on THIS AREA WRITE IN DO NOT this section. -

The Summer Assignment Will Receive a GRADE on the First Day of Class – August 9
Bishop Moore AP Chemistry Summer Assignment June 2017 Future AP Chemistry Student, Welcome to AP Chemistry. In order to ensure the best start for everyone next fall, I have prepared a summer assignment that reviews basic chemistry concepts some of which you may have forgotten you learned. For those topics you need help with there are a multitude of tremendous chemistry resources available on the Internet. With access to hundreds of websites either in your home or at the local library, I am confident that you will have sufficient resources to prepare adequately for the fall semester. The reference text book as part of AP course is “Chemistry: The Central Science” by Brown LeMay 14th Edition for AP. Much of the material in this summer packet will be familiar to you. It will be important for everyone to come to class the first day prepared. While I review throughout the course, extensive remediation is not an option as we work towards our goal of being 100% prepared for the AP Exam in early May. There will be a test covering the basic concepts included in the summer packet during the first or second week of school. You may contact me by email: ([email protected]) this summer. I will do my best to answer your questions ASAP. I hope you are looking forward to an exciting year of chemistry. You are all certainly excellent students, and with plenty of motivation and hard work you should find AP Chemistry a successful and rewarding experience. Finally, I recommend that you spread out the summer assignment. -

Molecular Dynamics Simulation of Ion Mobility. 2. Alkali Metal and Halide Ions Using the SPC/E Model for Water at 25 °C†
+ + 1420 J. Phys. Chem. 1996, 100, 1420-1425 Molecular Dynamics Simulation of Ion Mobility. 2. Alkali Metal and Halide Ions Using the SPC/E Model for Water at 25 °C† Song Hi Lee Department of Chemistry, Kyungsung UniVersity, Pusan 608-736, Korea Jayendran C. Rasaiah* Department of Chemistry, UniVersity of Maine, Orono, Maine 04469 ReceiVed: October 16, 1995; In Final Form: NoVember 10, 1995X We present results of computer simulations of the mobilities of the alkali metal ions (Li+, Na+,K+, Rb+, and Cs+) and the halides (F-, Cl-, Br-, and I-) at 25 °C using the SPC/E model for water and ion-water parameters fitted to the binding energies of small clusters of ions. A simple truncation of the ion-water and water- water potentials was used, and the mobilities calculated from the mean square displacement and the velocity autocorrelation functions, respectively, were found to be in good agreement with each other. The calculations demonstrate, for the first time, cation and anion mobilities that fall on separate curves, as functions of ion size, with distinct maxima. This is in complete accord with experimental trends observed in water at 25 °C. The cation mobilities are also in better agreement with the measured values than the calculations done earlier (J. Chem. Phys. 1994, 101, 6964) using the TIP4P model. The mobilities of the halides calculated here for the SPC/E model are however slightly lower than the experimental results. The residence times of water in the hydration shells around an ion are found to decrease dramatically with its size. Stereoscopic pictures show that the structure of the solvent cage around an ion is qualitatively different for the larger ions, implicating both solvent dynamics and structure as important factors in explaining ion mobility in aqueous systems. -

IODINE Its Properties and Technical Applications
IODINE Its Properties and Technical Applications CHILEAN IODINE EDUCATIONAL BUREAU, INC. 120 Broadway, New York 5, New York IODINE Its Properties and Technical Applications ¡¡iiHiüíiüüiütitittüHiiUitítHiiiittiíU CHILEAN IODINE EDUCATIONAL BUREAU, INC. 120 Broadway, New York 5, New York 1951 Copyright, 1951, by Chilean Iodine Educational Bureau, Inc. Printed in U.S.A. Contents Page Foreword v I—Chemistry of Iodine and Its Compounds 1 A Short History of Iodine 1 The Occurrence and Production of Iodine ....... 3 The Properties of Iodine 4 Solid Iodine 4 Liquid Iodine 5 Iodine Vapor and Gas 6 Chemical Properties 6 Inorganic Compounds of Iodine 8 Compounds of Electropositive Iodine 8 Compounds with Other Halogens 8 The Polyhalides 9 Hydrogen Iodide 1,0 Inorganic Iodides 10 Physical Properties 10 Chemical Properties 12 Complex Iodides .13 The Oxides of Iodine . 14 Iodic Acid and the Iodates 15 Periodic Acid and the Periodates 15 Reactions of Iodine and Its Inorganic Compounds With Organic Compounds 17 Iodine . 17 Iodine Halides 18 Hydrogen Iodide 19 Inorganic Iodides 19 Periodic and Iodic Acids 21 The Organic Iodo Compounds 22 Organic Compounds of Polyvalent Iodine 25 The lodoso Compounds 25 The Iodoxy Compounds 26 The Iodyl Compounds 26 The Iodonium Salts 27 Heterocyclic Iodine Compounds 30 Bibliography 31 II—Applications of Iodine and Its Compounds 35 Iodine in Organic Chemistry 35 Iodine and Its Compounds at Catalysts 35 Exchange Catalysis 35 Halogenation 38 Isomerization 38 Dehydration 39 III Page Acylation 41 Carbón Monoxide (and Nitric Oxide) Additions ... 42 Reactions with Oxygen 42 Homogeneous Pyrolysis 43 Iodine as an Inhibitor 44 Other Applications 44 Iodine and Its Compounds as Process Reagents ... -

Halides and Halogens. What Do I Need to Know? John Vivari, Nordson EFD
Halides and Halogens. What do I need to know? John Vivari, Nordson EFD Abstract With halogen-containing substances in the public eye due to scrutiny by the European Union and a variety of non- governmental organizations (NGOs) as possible additions to the list of substances banned from electronics, we at EFD have received numerous inquiries from customers asking how this subject will affect them and their processes. Having just overcome the hurdle of RoHS (Restriction of Hazardous Substances), they want to know what halogens and halides are, and what changes they should be prepared for if required to stop using them. Halide-free materials are not new. Some segments of the electronics industry have been sensitive to halides and their significance for decades. This paper will give the reader a working knowledge of halogens and halides. Armed with this education, the reader will be able to make informed decisions when required to use halogen-free materials, either because regulations dictate it or social pressure makes acceptance preferable to resistance. Key Words: halide, halogen, bromine, chlorine, flame retardant, RoHS What are halogens and halides? damage. Brominated flame retardant use is not limited to electronics. It is also in common usage in furniture, At their most basic level, halogens are the electronegative construction materials and textiles. elements in column 17 of the periodic table, including fluorine (F), chlorine, (Cl), bromine (Br), iodine (I) and Other sources of halogens in circuit boards include astatine (At). In electronics fiberglass sizing, epoxy curing agents and accelerators, applications, iodine and resin wetting and de-foaming agents, flux residues, and astatine are rarely if ever contamination from handling. -

Gas Phase Chemistry and Removal of CH3I During a Severe Accident
DK0100070 Nordisk kernesikkerhedsforskning Norraenar kjarnoryggisrannsoknir Pohjoismainenydinturvallisuustutkimus Nordiskkjernesikkerhetsforskning Nordisk karnsakerhetsforskning Nordic nuclear safety research NKS-25 ISBN 87-7893-076-6 Gas Phase Chemistry and Removal of CH I during a Severe Accident Anna Karhu VTT Energy, Finland 2/42 January 2001 Abstract The purpose of this literature review was to gather valuable information on the behavior of methyl iodide on the gas phase during a severe accident. The po- tential of transition metals, especially silver and copper, to remove organic io- dides from the gas streams was also studied. Transition metals are one of the most interesting groups in the contex of iodine mitigation. For example silver is known to react intensively with iodine compounds. Silver is also relatively inert material and it is thermally stable. Copper is known to react with some radioio- dine species. However, it is not reactive toward methyl iodide. In addition, it is oxidized to copper oxide under atmospheric conditions. This may limit the in- dustrial use of copper. Key words Methyl iodide, gas phase, severe accident mitigation NKS-25 ISBN 87-7893-076-6 Danka Services International, DSI, 2001 The report can be obtained from NKS Secretariat P.O. Box 30 DK-4000RoskiIde Denmark Phone +45 4677 4045 Fax +45 4677 4046 http://www.nks.org e-mail: [email protected] Gas Phase Chemistry and Removal of CH3I during a Severe Accident VTT Energy, Finland Anna Karhu Abstract The purpose of this literature review was to gather valuable information on the behavior of methyl iodide on the gas phase during a severe accident. -
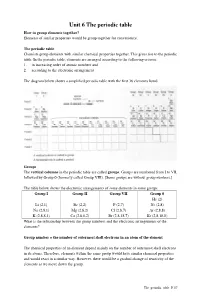
Unit 6 the Periodic Table How to Group Elements Together? Elements of Similar Properties Would Be Group Together for Convenience
Unit 6 The periodic table How to group elements together? Elements of similar properties would be group together for convenience. The periodic table Chemists group elements with similar chemical properties together. This gives rise to the periodic table. In the periodic table, elements are arranged according to the following criteria: 1. in increasing order of atomic numbers and 2. according to the electronic arrangement The diagram below shows a simplified periodic table with the first 36 elements listed. Groups The vertical columns in the periodic table are called groups . Groups are numbered from I to VII, followed by Group 0 (formerly called Group VIII). [Some groups are without group numbers.] The table below shows the electronic arrangements of some elements in some groups. Group I Group II Group VII Group 0 He (2) Li (2,1) Be (2,2) F (2,7) Ne (2,8) Na (2,8,1) Mg (2,8,2) Cl (2,8,7) Ar (2,8,8) K (2,8,8,1) Ca (2,8,8,2) Br (2,8,18,7) Kr (2,8,18,8) What is the relationship between the group numbers and the electronic arrangements of the elements? Group number = the number of outermost shell electrons in an atom of the element The chemical properties of an element depend mainly on the number of outermost shell electrons in its atoms. Therefore, elements within the same group would have similar chemical properties and would react in a similar way. However, there would be a gradual change of reactivity of the elements as we move down the group. -

Production Process for Refined Hydrogen Iodide
^ ^ ^ ^ I ^ ^ ^ ^ ^ ^ II ^ II ^ ^ ^ ^ ^ ^ ^ ^ ^ ^ ^ ^ ^ I ^ European Patent Office Office europeen des brevets EP 0 714 849 A1 EUROPEAN PATENT APPLICATION (43) Date of publication: (51) |nt CI.6: C01B7/13 05.06.1996 Bulletin 1996/23 (21) Application number: 95308524.8 (22) Date of filing: 28.11.1995 (84) Designated Contracting States: • Tanaka, Yoshinori DE FR GB NL Mobara-shi, Chiba (JP) • Omura, Masahiro (30) Priority: 28.11.1994 J P 292771/95 Mobara-shi, Chiba (JP) • Tomoshige, Naoki (71) Applicant: MITSUI TOATSU CHEMICALS, Mobara-shi, Chiba (JP) INCORPORATED Tokyo (JP) (74) Representative: Nicholls, Kathryn Margaret et al MEWBURN ELLIS (72) Inventors: York House • Utsunomiya, Atsushi 23 Kingsway Takaishi-shi, Osaka (JP) London WC2B 6HP (GB) • Sasaki, Kenju Mobara-shi, Chiba (JP) (54) Production process for refined hydrogen iodide (57) A process for producing refined hydrogen io- amount of which is at least 1/3 (weight ratio) relative to dide by contacting crude hydrogen iodide with a zeolite the amount of the zeolite, to convert impurities of sulfur is disclosed. Crude hydrogen iodide is obtained by re- components contained in the zeolite to hydrogen ducing iodine with a hydrogenated naphthalene, where- sulfide, thereby removing the sulfur components. The in all of iodine is dissolved in advance in a part of the kind of the zeolite is an A type zeolite (an average pore hydrogenated naphthalene to prepare an iodine solu- diameter: 3 angstrom, 4 angstrom or 5 angstrom) or a tion, and the reaction is carried out while adding contin- mordenite type zeolite. Further, an activated carbon uously or intermittently the above solution to the balance may be combined with the zeolite at the rear stage there- of the hydrogenated naphthalene; and the same oper- of. -

Chapter 9 Hydrogen
Chapter 9 Hydrogen Diborane, B2 H6• is the simplest member of a large class of compounds, the electron-deficient boron hydrid Like all boron hydrides, it has a positive standard free energy of formation, and so cannot be prepared direct from boron and hydrogen. The bridge B-H bonds are longer and weaker than the tenninal B-H bonds (13: vs. 1.19 A). 89.1 Reactions of hydrogen compounds? (a) Ca(s) + H2Cg) ~ CaH2Cs). This is the reaction of an active s-me: with hydrogen, which is the way that saline metal hydrides are prepared. (b) NH3(g) + BF)(g) ~ H)N-BF)(g). This is the reaction of a Lewis base and a Lewis acid. The product I Lewis acid-base complex. (c) LiOH(s) + H2(g) ~ NR. Although dihydrogen can behave as an oxidant (e.g., with Li to form LiH) or reductant (e.g., with O2 to form H20), it does not behave as a Br0nsted or Lewis acid or base. It does not r with strong bases, like LiOH, or with strong acids. 89.2 A procedure for making Et3MeSn? A possible procedure is as follows: ~ 2Et)SnH + 2Na 2Na+Et3Sn- + H2 Ja'Et3Sn- + CH3Br ~ Et3MeSn + NaBr 9.1 Where does Hydrogen fit in the periodic chart? (a) Hydrogen in group 1? Hydrogen has one vale electron like the group 1 metals and is stable as W, especially in aqueous media. The other group 1 m have one valence electron and are quite stable as ~ cations in solution and in the solid state as simple 1 salts.