4. Energy, Power, and Photons
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
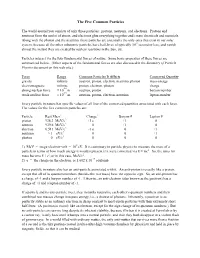
The Five Common Particles
The Five Common Particles The world around you consists of only three particles: protons, neutrons, and electrons. Protons and neutrons form the nuclei of atoms, and electrons glue everything together and create chemicals and materials. Along with the photon and the neutrino, these particles are essentially the only ones that exist in our solar system, because all the other subatomic particles have half-lives of typically 10-9 second or less, and vanish almost the instant they are created by nuclear reactions in the Sun, etc. Particles interact via the four fundamental forces of nature. Some basic properties of these forces are summarized below. (Other aspects of the fundamental forces are also discussed in the Summary of Particle Physics document on this web site.) Force Range Common Particles It Affects Conserved Quantity gravity infinite neutron, proton, electron, neutrino, photon mass-energy electromagnetic infinite proton, electron, photon charge -14 strong nuclear force ≈ 10 m neutron, proton baryon number -15 weak nuclear force ≈ 10 m neutron, proton, electron, neutrino lepton number Every particle in nature has specific values of all four of the conserved quantities associated with each force. The values for the five common particles are: Particle Rest Mass1 Charge2 Baryon # Lepton # proton 938.3 MeV/c2 +1 e +1 0 neutron 939.6 MeV/c2 0 +1 0 electron 0.511 MeV/c2 -1 e 0 +1 neutrino ≈ 1 eV/c2 0 0 +1 photon 0 eV/c2 0 0 0 1) MeV = mega-electron-volt = 106 eV. It is customary in particle physics to measure the mass of a particle in terms of how much energy it would represent if it were converted via E = mc2. -

Quantum Field Theory*
Quantum Field Theory y Frank Wilczek Institute for Advanced Study, School of Natural Science, Olden Lane, Princeton, NJ 08540 I discuss the general principles underlying quantum eld theory, and attempt to identify its most profound consequences. The deep est of these consequences result from the in nite number of degrees of freedom invoked to implement lo cality.Imention a few of its most striking successes, b oth achieved and prosp ective. Possible limitation s of quantum eld theory are viewed in the light of its history. I. SURVEY Quantum eld theory is the framework in which the regnant theories of the electroweak and strong interactions, which together form the Standard Mo del, are formulated. Quantum electro dynamics (QED), b esides providing a com- plete foundation for atomic physics and chemistry, has supp orted calculations of physical quantities with unparalleled precision. The exp erimentally measured value of the magnetic dip ole moment of the muon, 11 (g 2) = 233 184 600 (1680) 10 ; (1) exp: for example, should b e compared with the theoretical prediction 11 (g 2) = 233 183 478 (308) 10 : (2) theor: In quantum chromo dynamics (QCD) we cannot, for the forseeable future, aspire to to comparable accuracy.Yet QCD provides di erent, and at least equally impressive, evidence for the validity of the basic principles of quantum eld theory. Indeed, b ecause in QCD the interactions are stronger, QCD manifests a wider variety of phenomena characteristic of quantum eld theory. These include esp ecially running of the e ective coupling with distance or energy scale and the phenomenon of con nement. -

3.Joule's Experiments
The Force of Gravity Creates Energy: The “Work” of James Prescott Joule http://www.bookrags.com/biography/james-prescott-joule-wsd/ James Prescott Joule (1818-1889) was the son of a successful British brewer. He tinkered with the tools of his father’s trade (particularly thermometers), and despite never earning an undergraduate degree, he was able to answer two rather simple questions: 1. Why is the temperature of the water at the bottom of a waterfall higher than the temperature at the top? 2. Why does an electrical current flowing through a conductor raise the temperature of water? In order to adequately investigate these questions on our own, we need to first define “temperature” and “energy.” Second, we should determine how the measurement of temperature can relate to “heat” (as energy). Third, we need to find relationships that might exist between temperature and “mechanical” energy and also between temperature and “electrical” energy. Definitions: Before continuing, please write down what you know about temperature and energy below. If you require more space, use the back. Temperature: Energy: We have used the concept of gravity to show how acceleration of freely falling objects is related mathematically to distance, time, and speed. We have also used the relationship between net force applied through a distance to define “work” in the Harvard Step Test. Now, through the work of Joule, we can equate the concepts of “work” and “energy”: Energy is the capacity of a physical system to do work. Potential energy is “stored” energy, kinetic energy is “moving” energy. One type of potential energy is that induced by the gravitational force between two objects held at a distance (there are other types of potential energy, including electrical, magnetic, chemical, nuclear, etc). -

7. Gamma and X-Ray Interactions in Matter
Photon interactions in matter Gamma- and X-Ray • Compton effect • Photoelectric effect Interactions in Matter • Pair production • Rayleigh (coherent) scattering Chapter 7 • Photonuclear interactions F.A. Attix, Introduction to Radiological Kinematics Physics and Radiation Dosimetry Interaction cross sections Energy-transfer cross sections Mass attenuation coefficients 1 2 Compton interaction A.H. Compton • Inelastic photon scattering by an electron • Arthur Holly Compton (September 10, 1892 – March 15, 1962) • Main assumption: the electron struck by the • Received Nobel prize in physics 1927 for incoming photon is unbound and stationary his discovery of the Compton effect – The largest contribution from binding is under • Was a key figure in the Manhattan Project, condition of high Z, low energy and creation of first nuclear reactor, which went critical in December 1942 – Under these conditions photoelectric effect is dominant Born and buried in • Consider two aspects: kinematics and cross Wooster, OH http://en.wikipedia.org/wiki/Arthur_Compton sections http://www.findagrave.com/cgi-bin/fg.cgi?page=gr&GRid=22551 3 4 Compton interaction: Kinematics Compton interaction: Kinematics • An earlier theory of -ray scattering by Thomson, based on observations only at low energies, predicted that the scattered photon should always have the same energy as the incident one, regardless of h or • The failure of the Thomson theory to describe high-energy photon scattering necessitated the • Inelastic collision • After the collision the electron departs -

1.1. Introduction the Phenomenon of Positron Annihilation Spectroscopy
PRINCIPLES OF POSITRON ANNIHILATION Chapter-1 __________________________________________________________________________________________ 1.1. Introduction The phenomenon of positron annihilation spectroscopy (PAS) has been utilized as nuclear method to probe a variety of material properties as well as to research problems in solid state physics. The field of solid state investigation with positrons started in the early fifties, when it was recognized that information could be obtained about the properties of solids by studying the annihilation of a positron and an electron as given by Dumond et al. [1] and Bendetti and Roichings [2]. In particular, the discovery of the interaction of positrons with defects in crystal solids by Mckenize et al. [3] has given a strong impetus to a further elaboration of the PAS. Currently, PAS is amongst the best nuclear methods, and its most recent developments are documented in the proceedings of the latest positron annihilation conferences [4-8]. PAS is successfully applied for the investigation of electron characteristics and defect structures present in materials, magnetic structures of solids, plastic deformation at low and high temperature, and phase transformations in alloys, semiconductors, polymers, porous material, etc. Its applications extend from advanced problems of solid state physics and materials science to industrial use. It is also widely used in chemistry, biology, and medicine (e.g. locating tumors). As the process of measurement does not mostly influence the properties of the investigated sample, PAS is a non-destructive testing approach that allows the subsequent study of a sample by other methods. As experimental equipment for many applications, PAS is commercially produced and is relatively cheap, thus, increasingly more research laboratories are using PAS for basic research, diagnostics of machine parts working in hard conditions, and for characterization of high-tech materials. -

Work and Energy Summary Sheet Chapter 6
Work and Energy Summary Sheet Chapter 6 Work: work is done when a force is applied to a mass through a displacement or W=Fd. The force and the displacement must be parallel to one another in order for work to be done. F (N) W =(Fcosθ)d F If the force is not parallel to The area of a force vs. the displacement, then the displacement graph + W component of the force that represents the work θ d (m) is parallel must be found. done by the varying - W d force. Signs and Units for Work Work is a scalar but it can be positive or negative. Units of Work F d W = + (Ex: pitcher throwing ball) 1 N•m = 1 J (Joule) F d W = - (Ex. catcher catching ball) Note: N = kg m/s2 • Work – Energy Principle Hooke’s Law x The work done on an object is equal to its change F = kx in kinetic energy. F F is the applied force. 2 2 x W = ΔEk = ½ mvf – ½ mvi x is the change in length. k is the spring constant. F Energy Defined Units Energy is the ability to do work. Same as work: 1 N•m = 1 J (Joule) Kinetic Energy Potential Energy Potential energy is stored energy due to a system’s shape, position, or Kinetic energy is the energy of state. motion. If a mass has velocity, Gravitational PE Elastic (Spring) PE then it has KE 2 Mass with height Stretch/compress elastic material Ek = ½ mv 2 EG = mgh EE = ½ kx To measure the change in KE Change in E use: G Change in ES 2 2 2 2 ΔEk = ½ mvf – ½ mvi ΔEG = mghf – mghi ΔEE = ½ kxf – ½ kxi Conservation of Energy “The total energy is neither increased nor decreased in any process. -

1 the LOCALIZED QUANTUM VACUUM FIELD D. Dragoman
1 THE LOCALIZED QUANTUM VACUUM FIELD D. Dragoman – Univ. Bucharest, Physics Dept., P.O. Box MG-11, 077125 Bucharest, Romania, e-mail: [email protected] ABSTRACT A model for the localized quantum vacuum is proposed in which the zero-point energy of the quantum electromagnetic field originates in energy- and momentum-conserving transitions of material systems from their ground state to an unstable state with negative energy. These transitions are accompanied by emissions and re-absorptions of real photons, which generate a localized quantum vacuum in the neighborhood of material systems. The model could help resolve the cosmological paradox associated to the zero-point energy of electromagnetic fields, while reclaiming quantum effects associated with quantum vacuum such as the Casimir effect and the Lamb shift; it also offers a new insight into the Zitterbewegung of material particles. 2 INTRODUCTION The zero-point energy (ZPE) of the quantum electromagnetic field is at the same time an indispensable concept of quantum field theory and a controversial issue (see [1] for an excellent review of the subject). The need of the ZPE has been recognized from the beginning of quantum theory of radiation, since only the inclusion of this term assures no first-order temperature-independent correction to the average energy of an oscillator in thermal equilibrium with blackbody radiation in the classical limit of high temperatures. A more rigorous introduction of the ZPE stems from the treatment of the electromagnetic radiation as an ensemble of harmonic quantum oscillators. Then, the total energy of the quantum electromagnetic field is given by E = åk,s hwk (nks +1/ 2) , where nks is the number of quantum oscillators (photons) in the (k,s) mode that propagate with wavevector k and frequency wk =| k | c = kc , and are characterized by the polarization index s. -

Active Short Circuit - Chassis Short Characterization and Potential Mitigation Technique for the MMRTG
Jet Propulsion Laboratory California Institute of Technology Multi-Mission RTG Active Short Circuit - Chassis Short Characterization and Potential Mitigation Technique for the MMRTG February 25, 2015 Gary Bolotin1 Nicholas Keyawa1 1Jet Propulsion Laboratory, California Institute of Technology 1 Jet Propulsion Laboratory California Institute of Technology Agenda Multi-Mission RTG • Introduction – MMRTG – Internal MMRTG Chassis Shorts • Active Short Circuit Purpose • Active Short Circuit Theory • Active Short Design and Component Layout • Conclusion and Future Work 2 Jet Propulsion Laboratory California Institute of Technology Introduction – MMRTG Multi-Mission RTG • The Multi-Mission Radioisotope Thermoelectric Generator (MMRTG) utilizes a combination of PbTe, PbSnTe, and TAGS thermoelectric couples to produce electric current from the heat generated by the radioactive decay of plutonium – 238. 3 Jet Propulsion Laboratory Introduction – Internal MMRTG Chassis California Institute of Technology Shorts Multi-Mission RTG • Shorts inside the MMRTG between the electrical power circuit and the MMRTG chassis have been detected via isolation checks and/or changes in the bus balance voltage. • Example location of short shown below MMRTG Chassis Internal MMRTG Short 4 Jet Propulsion Laboratory California Institute of Technology Active Short Circuit Purpose Multi-Mission RTG • The leading hypothesis suggests that the FOD which is causing the internal shorts are extremely small pieces of material that could potentially melt and/or sublimate away given a sufficient amount of current. • By inducing a controlled second short (in the presence of an internal MMRTG chassis short), a significant amount of current flow can be generated to achieve three main design goals: 1) Measure and characterize the MMRTG internal short to chassis, 2) Safely determine if the MMRTG internal short can be cleared in the presence of another controlled short 3) Quantify the amount of energy required to clear the MMRTG internal short. -

First Determination of the Electric Charge of the Top Quark
First Determination of the Electric Charge of the Top Quark PER HANSSON arXiv:hep-ex/0702004v1 1 Feb 2007 Licentiate Thesis Stockholm, Sweden 2006 Licentiate Thesis First Determination of the Electric Charge of the Top Quark Per Hansson Particle and Astroparticle Physics, Department of Physics Royal Institute of Technology, SE-106 91 Stockholm, Sweden Stockholm, Sweden 2006 Cover illustration: View of a top quark pair event with an electron and four jets in the final state. Image by DØ Collaboration. Akademisk avhandling som med tillst˚and av Kungliga Tekniska H¨ogskolan i Stock- holm framl¨agges till offentlig granskning f¨or avl¨aggande av filosofie licentiatexamen fredagen den 24 november 2006 14.00 i sal FB54, AlbaNova Universitets Center, KTH Partikel- och Astropartikelfysik, Roslagstullsbacken 21, Stockholm. Avhandlingen f¨orsvaras p˚aengelska. ISBN 91-7178-493-4 TRITA-FYS 2006:69 ISSN 0280-316X ISRN KTH/FYS/--06:69--SE c Per Hansson, Oct 2006 Printed by Universitetsservice US AB 2006 Abstract In this thesis, the first determination of the electric charge of the top quark is presented using 370 pb−1 of data recorded by the DØ detector at the Fermilab Tevatron accelerator. tt¯ events are selected with one isolated electron or muon and at least four jets out of which two are b-tagged by reconstruction of a secondary decay vertex (SVT). The method is based on the discrimination between b- and ¯b-quark jets using a jet charge algorithm applied to SVT-tagged jets. A method to calibrate the jet charge algorithm with data is developed. A constrained kinematic fit is performed to associate the W bosons to the correct b-quark jets in the event and extract the top quark electric charge. -

Electric Units and Standards
DEPARTMENT OF COMMERCE OF THE Bureau of Standards S. W. STRATTON, Director No. 60 ELECTRIC UNITS AND STANDARDS list Edition 1 Issued September 25. 1916 WASHINGTON GOVERNMENT PRINTING OFFICE DEPARTMENT OF COMMERCE Circular OF THE Bureau of Standards S. W. STRATTON, Director No. 60 ELECTRIC UNITS AND STANDARDS [1st Edition] Issued September 25, 1916 WASHINGTON GOVERNMENT PRINTING OFFICE 1916 ADDITIONAL COPIES OF THIS PUBLICATION MAY BE PROCURED FROM THE SUPERINTENDENT OF DOCUMENTS GOVERNMENT PRINTING OFFICE WASHINGTON, D. C. AT 15 CENTS PER COPY A complete list of the Bureau’s publications may be obtained free of charge on application to the Bureau of Standards, Washington, D. C. ELECTRIC UNITS AND STANDARDS CONTENTS Page I. The systems of units 4 1. Units and standards in general 4 2. The electrostatic and electromagnetic systems 8 () The numerically different systems of units 12 () Gaussian systems, and ratios of the units 13 “ ’ (c) Practical ’ electromagnetic units 15 3. The international electric units 16 II. Evolution of present system of concrete standards 18 1. Early electric standards 18 2. Basis of present laws 22 3. Progress since 1893 24 III. Units and standards of the principal electric quantities 29 1. Resistance 29 () International ohm 29 Definition 29 Mercury standards 30 Secondary standards 31 () Resistance standards in practice 31 (c) Absolute ohm 32 2. Current 33 (a) International ampere 33 Definition 34 The silver voltameter 35 ( b ) Resistance standards used in current measurement 36 (c) Absolute ampere 37 3. Electromotive force 38 (a) International volt 38 Definition 39 Weston normal cell 39 (b) Portable Weston cells 41 (c) Absolute and semiabsolute volt 42 4. -

Photon Cross Sections, Attenuation Coefficients, and Energy Absorption Coefficients from 10 Kev to 100 Gev*
1 of Stanaaros National Bureau Mmin. Bids- r'' Library. Ml gEP 2 5 1969 NSRDS-NBS 29 . A111D1 ^67174 tioton Cross Sections, i NBS Attenuation Coefficients, and & TECH RTC. 1 NATL INST OF STANDARDS _nergy Absorption Coefficients From 10 keV to 100 GeV U.S. DEPARTMENT OF COMMERCE NATIONAL BUREAU OF STANDARDS T X J ". j NATIONAL BUREAU OF STANDARDS 1 The National Bureau of Standards was established by an act of Congress March 3, 1901. Today, in addition to serving as the Nation’s central measurement laboratory, the Bureau is a principal focal point in the Federal Government for assuring maximum application of the physical and engineering sciences to the advancement of technology in industry and commerce. To this end the Bureau conducts research and provides central national services in four broad program areas. These are: (1) basic measurements and standards, (2) materials measurements and standards, (3) technological measurements and standards, and (4) transfer of technology. The Bureau comprises the Institute for Basic Standards, the Institute for Materials Research, the Institute for Applied Technology, the Center for Radiation Research, the Center for Computer Sciences and Technology, and the Office for Information Programs. THE INSTITUTE FOR BASIC STANDARDS provides the central basis within the United States of a complete and consistent system of physical measurement; coordinates that system with measurement systems of other nations; and furnishes essential services leading to accurate and uniform physical measurements throughout the Nation’s scientific community, industry, and com- merce. The Institute consists of an Office of Measurement Services and the following technical divisions: Applied Mathematics—Electricity—Metrology—Mechanics—Heat—Atomic and Molec- ular Physics—Radio Physics -—Radio Engineering -—Time and Frequency -—Astro- physics -—Cryogenics. -

Section 22-3: Energy, Momentum and Radiation Pressure
Answer to Essential Question 22.2: (a) To find the wavelength, we can combine the equation with the fact that the speed of light in air is 3.00 " 108 m/s. Thus, a frequency of 1 " 1018 Hz corresponds to a wavelength of 3 " 10-10 m, while a frequency of 90.9 MHz corresponds to a wavelength of 3.30 m. (b) Using Equation 22.2, with c = 3.00 " 108 m/s, gives an amplitude of . 22-3 Energy, Momentum and Radiation Pressure All waves carry energy, and electromagnetic waves are no exception. We often characterize the energy carried by a wave in terms of its intensity, which is the power per unit area. At a particular point in space that the wave is moving past, the intensity varies as the electric and magnetic fields at the point oscillate. It is generally most useful to focus on the average intensity, which is given by: . (Eq. 22.3: The average intensity in an EM wave) Note that Equations 22.2 and 22.3 can be combined, so the average intensity can be calculated using only the amplitude of the electric field or only the amplitude of the magnetic field. Momentum and radiation pressure As we will discuss later in the book, there is no mass associated with light, or with any EM wave. Despite this, an electromagnetic wave carries momentum. The momentum of an EM wave is the energy carried by the wave divided by the speed of light. If an EM wave is absorbed by an object, or it reflects from an object, the wave will transfer momentum to the object.