Abnormal Embryonic Lymphatic Vessel Development in Tie1 Hypomorphic Mice Xianghu Qu, Kevin Tompkins, Lorene E
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Discovery of Orphan Receptor Tie1 and Angiopoietin Ligands Ang1 and Ang4 As Novel GAG-Binding Partners
78 Chapter 3 Discovery of Orphan Receptor Tie1 and Angiopoietin Ligands Ang1 and Ang4 as Novel GAG-Binding Partners 79 3.1 Abstract The Tie/Ang signaling axis is necessary for proper vascular development and remodeling. However, the mechanisms that modulate signaling through this receptor tyrosine kinase pathway are relatively unclear. In particular, the role of the orphan receptor Tie1 is highly disputed. Although this protein is required for survival, Tie1 has been found both to inhibit and yet be necessary for Tie2 signaling. While differing expression levels have been put forth as an explanation for its context-specific activity, the lack of known endogenous ligands for Tie1 has severely hampered understanding its molecular mode of action. Here we describe the discovery of orphan receptor Tie1 and angiopoietin ligands Ang1 and Ang4 as novel GAG binding partners. We localize the binding site of GAGs to the N- terminal region of Tie1, which may provide structural insights into the importance of this interaction regarding the formation of Tie1-Tie2 heterodimerization. Furthermore, we use our mutagenesis studies to guide the generation of a mouse model that specifically ablates GAG-Tie1 binding in vivo for further characterization of the functional outcomes of GAG-Tie1 binding. We also show that GAGs can form a trimeric complex with Ang1/4 and Tie2 using our microarray technology. Finally, we use our HaloTag glycan engineering platform to modify the cell surface of endothelial cells and demonstrate that HS GAGs can potentiate Tie2 signaling in a sulfation-specific manner, providing the first evidence of the involvement of HS GAGs in Tie/Ang signaling and delineating further the integral role of HS GAGs in angiogenesis. -

The Evolving Cardiac Lymphatic Vasculature in Development, Repair and Regeneration
REVIEWS The evolving cardiac lymphatic vasculature in development, repair and regeneration Konstantinos Klaourakis 1,2, Joaquim M. Vieira 1,2,3 ✉ and Paul R. Riley 1,2,3 ✉ Abstract | The lymphatic vasculature has an essential role in maintaining normal fluid balance in tissues and modulating the inflammatory response to injury or pathogens. Disruption of normal development or function of lymphatic vessels can have severe consequences. In the heart, reduced lymphatic function can lead to myocardial oedema and persistent inflammation. Macrophages, which are phagocytic cells of the innate immune system, contribute to cardiac development and to fibrotic repair and regeneration of cardiac tissue after myocardial infarction. In this Review, we discuss the cardiac lymphatic vasculature with a focus on developments over the past 5 years arising from the study of mammalian and zebrafish model organisms. In addition, we examine the interplay between the cardiac lymphatics and macrophages during fibrotic repair and regeneration after myocardial infarction. Finally, we discuss the therapeutic potential of targeting the cardiac lymphatic network to regulate immune cell content and alleviate inflammation in patients with ischaemic heart disease. The circulatory system of vertebrates is composed of two after MI. In this Review, we summarize the current complementary vasculatures, the blood and lymphatic knowledge on the development, structure and function vascular systems1. The blood vasculature is a closed sys- of the cardiac lymphatic vasculature, with an emphasis tem responsible for transporting gases, fluids, nutrients, on breakthroughs over the past 5 years in the study of metabolites and cells to the tissues2. This extravasation of cardiac lymphatic heterogeneity in mice and zebrafish. -
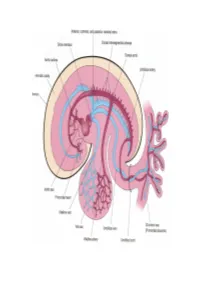
Development of HEART 4-VEINS
Development of brachiocephalic veins 1. Right brachiocephalic vein is formed by cranial part of right anterior cardinal vein and 2. Left brachiocephalic is formed by cranial part of left anterior cardinal vein and the interant.cardinal anastomosis. Development of superior vena cava 1. The part up to the opening of vena azygos develops from caudal part of right ant.cardinal vein and 2. The part below the opening (intrapericardial part) is formed by the right common cardinal vein. Development of azygos and hemiazygos veins A. 1. Vena azygos develops from right azygos line vein and 2. The arch of vena azygos is formed by the cranial end of right postcardinal vein. B. Hemiazygos veins are formed by the left azygos line vein. Development of Inferior vena cava Inferior vena cava is formed, from below upwards by: 1. Begins by the union of the two common iliac veins (postcardinal veins), 2. Right supracardinal, 3. Right supra-subcardinal anastomosis, 4. Right subcardinal, 5. New formation (hepatic segment) and 6. Hepatocardiac channel (terminal part of right vitelline vein). Congenital anomalies • Double inferior vena cava • Absence • Left SVC • Double SVC DEVELOPMENT OF PORTAL VEIN 1. The portal vein is formed behind the neck of pancreas by the union of superior mesentric and splenic vein to the left vitelline vein. 2. The part of the portal vein which is behind the Ist part of duodenum is formed by middle dorsal transverse anastomosis. 3. Part of portal vein which is in the free margin of lesser omentum is formed by cranial or distal part of right vitelline vein. -

Src-Family Kinases Impact Prognosis and Targeted Therapy in Flt3-ITD+ Acute Myeloid Leukemia
Src-Family Kinases Impact Prognosis and Targeted Therapy in Flt3-ITD+ Acute Myeloid Leukemia Title Page by Ravi K. Patel Bachelor of Science, University of Minnesota, 2013 Submitted to the Graduate Faculty of School of Medicine in partial fulfillment of the requirements for the degree of Doctor of Philosophy University of Pittsburgh 2019 Commi ttee Membership Pa UNIVERSITY OF PITTSBURGH SCHOOL OF MEDICINE Commi ttee Membership Page This dissertation was presented by Ravi K. Patel It was defended on May 31, 2019 and approved by Qiming (Jane) Wang, Associate Professor Pharmacology and Chemical Biology Vaughn S. Cooper, Professor of Microbiology and Molecular Genetics Adrian Lee, Professor of Pharmacology and Chemical Biology Laura Stabile, Research Associate Professor of Pharmacology and Chemical Biology Thomas E. Smithgall, Dissertation Director, Professor and Chair of Microbiology and Molecular Genetics ii Copyright © by Ravi K. Patel 2019 iii Abstract Src-Family Kinases Play an Important Role in Flt3-ITD Acute Myeloid Leukemia Prognosis and Drug Efficacy Ravi K. Patel, PhD University of Pittsburgh, 2019 Abstract Acute myelogenous leukemia (AML) is a disease characterized by undifferentiated bone-marrow progenitor cells dominating the bone marrow. Currently the five-year survival rate for AML patients is 27.4 percent. Meanwhile the standard of care for most AML patients has not changed for nearly 50 years. We now know that AML is a genetically heterogeneous disease and therefore it is unlikely that all AML patients will respond to therapy the same way. Upregulation of protein-tyrosine kinase signaling pathways is one common feature of some AML tumors, offering opportunities for targeted therapy. -

Cardiovascular System Heart Development Cardiovascular System Heart Development
Cardiovascular System Heart Development Cardiovascular System Heart Development In human embryos, the heart begins to beat at approximately 22-23 days, with blood flow beginning in the 4th week. The heart is one of the earliest differentiating and functioning organs. • This emphasizes the critical nature of the heart in distributing blood through the vessels and the vital exchange of nutrients, oxygen, and wastes between the developing baby and the mother. • Therefore, the first system that completes its development in the embryo is called cardiovascular system. https://www.slideshare.net/DrSherifFahmy/intraembryonic-mesoderm-general-embryology Mesoderm is one of the three • Connective tissue primary germ layers that • Smooth and striated muscle • Cardiovascular System differentiates early in • Kidneys development that collectively • Spleen • Genital organs, ducts gives rise to all subsequent • Adrenal gland cortex tissues and organs. The cardiovascular system begins to develop in the third week of gestation. Blood islands develop in the newly formed mesoderm, and consist of (a) a central group of haemoblasts, the embryonic precursors of blood cells; (b) endothelial cells. Development of the heart and vascular system is often described together as the cardiovascular system. Development begins very early in mesoderm both within (embryonic) and outside (extra embryonic, vitelline, umblical and placental) the embryo. Vascular development occurs in many places. • Blood islands coalesce to form a vascular plexus. Preferential channels form arteries and veins. • Day 17 - Blood islands form first in the extra-embryonic mesoderm • Day 18 - Blood islands form next in the intra-embryonic mesoderm • Day 19 - Blood islands form in the cardiogenic mesoderm and coalesce to form a pair of endothelial heart tubes Development of a circulation • A circulation is established during the 4th week after the myocardium is differentiated. -
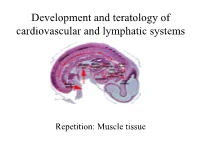
Development and Teratology of Cardiovascular and Lymphatic Systems
Development and teratology of cardiovascular and lymphatic systems Repetition: Muscle tissue Beginning of the cardiovascular system development – the 3rd week: Hemangiogenesis (day 15 – 16) – blood islets (insulae sanguinae) in extraembryonic mesoderm and splanchnic mesenchyme of embryo Clusters of mesenchyme cells (angiogenic cells) differentiate into: - angioblasts endothelium (at the periphery of blood islets) - hemoblasts primitive erythrocytes (in the center of blood islets) Clusters of angiogenic cells form a "horseshoe-shaped" space between somatic and splanchnic layer of mesoderm = pericardial cavity. Two endothelial tubes arrise in splanchnic mesoderm. The ventral portion of these tubes forms the cardiogenic area with two heart tubes, while the lateral portions form the dorsal aortae. Germ disc: prosencephalon mesencephalon eye rhombencephalon heart lateral mesoderm somites small blood vessels blood islands 8,9 Spine primitive streak Initially, the cardiogenic area is located anterior to the prechordal plate and the neural plate. The growth of the central nervous system pulls the cardiogenic area and prechordal plate (buccopharyngeal membrane ventrally and caudally ( ). Cardiogenic region just cranial to the prechordal plate. The canalization of cardiogenic clusters in the splanchnic mesoderm results in the formation of the paired heart tubes. Folding of embryo and primitive gut separation from yolk sac. Fusion of the heart tubes a single heart tube is, temporarily attached to the dorsal side of the pericardial cavity by the -

6 Development of the Great Vessels and Conduction Tissue
Development of the Great Vessels and Conduc6on Tissue Development of the heart fields • h:p://php.med.unsw.edu.au/embryology/ index.php?6tle=Advanced_-_Heart_Fields ! 2 Septa6on of the Bulbus Cordis Bulbus Cordis AV Canal Ventricle Looking at a sagital sec6on of the heart early in development the bulbus cordis is con6nuous with the ventricle which is con6nuous with the atria. As the AV canal shiOs to the right the bulbus move to the right as well. Septa6on of the Bulbus Cordis A P A P The next three slides make the point via cross sec6ons that the aorta and pulmonary arteries rotate around each other. This means the septum between them changes posi6on from superior to inferior as well. Septa6on of the Bulbus Cordis P A A P Septa6on of the Bulbus Cordis P A P A Migra6on of neural crest cells Neural crest cells migrate from the 3ed, 4th and 6th pharyngeal arches to form some of the popula6on of cells forming the aor6copulmonary septum. Septa6on of the Bulbus Cordis Truncal (Conal) Swellings Bulbus Cordis The cardiac jelly in the region of the truncus and conus adds the neural crest cells and expands as truncal swellings. Septa6on of the Bulbus Cordis Aorticopulmonary septum These swellings grow toward each other to meet and form the septum between the aorta and pulmonary artery. Aorta Pulmonary Artery Septa6on of the Bulbus Cordis Anterior 1 2 3 1 2 3 The aor6copulmonary septum then rotates as it moves inferiorly. However, the exact mechanism for that rota6on remains unclear. Septa6on of the Bulbus Cordis Aorta Pulmonary Artery Conal Ridges IV Foramen Membranous Muscular IV Endocarial Septum Interventricular Cushion Septum However, the aor6copulmonary septum must form properly for the IV septum to be completed. -

Dosage-Sensitive Requirement for Mouse Dll4 in Artery Development
Downloaded from genesdev.cshlp.org on September 26, 2021 - Published by Cold Spring Harbor Laboratory Press RESEARCH COMMUNICATION Dosage-sensitive requirement 2000), which were also observed, to a lesser degree, in Hey1/Hey2 double mutants, the putative downstream for mouse Dll4 targets of Notch signaling (Fischer et al. 2004). Here we in artery development show, in contrast, that the Dll4 ligand alone is required in a dosage-sensitive manner for normal arterial pattern- António Duarte,1,5 Masanori Hirashima,3,5,6 ing in development. Rui Benedito,1 Alexandre Trindade,1 The Dll4 gene was inactivated by targeted disruption 1 2 1 in embryonic stem (ES) cells. The targeting vector was Patrícia Diniz, Evguenia Bekman, Luís Costa, designed to replace the initiation codon and the first 2 3,4,7 Domingos Henrique, and Janet Rossant three coding exons with the -galactosidase (lacZ) re- porter gene (Fig. 1A). Electroporation of R1 ES cells with 1CIISA, Faculdade de Medicina Veterina´ria, 1300-0-477 the targeting vector followed by G418 selection resulted Lisboa, Portugal; 2Instituto de Medicina Molecular, Faculdade in the derivation of four correctly gene-targeted ES cell de Medicina, 1649-9-028 Lisboa, Portugal; 3Samuel Lunenfeld lines. Chimeric mice were generated by aggregation be- Research Institute, Mount Sinai Hospital, Toronto, Ontario, tween Dll4+/− ES cells and ICR host embryos. When Canada M5G1X5; 4Department of Molecular and Medical crossed with wild-type ICR females, five male chimeras Genetics, University of Toronto, Toronto, Ontario, produced F1 agouti pups at 100% frequency. Genotyping Canada M5S 1A8 of F1 agouti mice by Southern blotting or polymerase +/− Involvement of the Notch signaling pathway in vascular chain reaction (PCR; Fig. -

Protein Tyrosine Kinases: Their Roles and Their Targeting in Leukemia
cancers Review Protein Tyrosine Kinases: Their Roles and Their Targeting in Leukemia Kalpana K. Bhanumathy 1,*, Amrutha Balagopal 1, Frederick S. Vizeacoumar 2 , Franco J. Vizeacoumar 1,3, Andrew Freywald 2 and Vincenzo Giambra 4,* 1 Division of Oncology, College of Medicine, University of Saskatchewan, Saskatoon, SK S7N 5E5, Canada; [email protected] (A.B.); [email protected] (F.J.V.) 2 Department of Pathology and Laboratory Medicine, College of Medicine, University of Saskatchewan, Saskatoon, SK S7N 5E5, Canada; [email protected] (F.S.V.); [email protected] (A.F.) 3 Cancer Research Department, Saskatchewan Cancer Agency, 107 Wiggins Road, Saskatoon, SK S7N 5E5, Canada 4 Institute for Stem Cell Biology, Regenerative Medicine and Innovative Therapies (ISBReMIT), Fondazione IRCCS Casa Sollievo della Sofferenza, 71013 San Giovanni Rotondo, FG, Italy * Correspondence: [email protected] (K.K.B.); [email protected] (V.G.); Tel.: +1-(306)-716-7456 (K.K.B.); +39-0882-416574 (V.G.) Simple Summary: Protein phosphorylation is a key regulatory mechanism that controls a wide variety of cellular responses. This process is catalysed by the members of the protein kinase su- perfamily that are classified into two main families based on their ability to phosphorylate either tyrosine or serine and threonine residues in their substrates. Massive research efforts have been invested in dissecting the functions of tyrosine kinases, revealing their importance in the initiation and progression of human malignancies. Based on these investigations, numerous tyrosine kinase inhibitors have been included in clinical protocols and proved to be effective in targeted therapies for various haematological malignancies. -

Structural Basis of Tie2 Activation and Tie2/Tie1 Heterodimerization
Structural basis of Tie2 activation and Tie2/Tie1 heterodimerization Veli-Matti Leppänena,1, Pipsa Saharinena,b, and Kari Alitaloa,b,1 aWihuri Research Institute, Biomedicum Helsinki, Haartmaninkatu 8, 00290 Helsinki, Finland; and bTranslational Cancer Biology Program, Research Programs Unit, University of Helsinki, 00014 Helsinki, Finland Contributed by Kari Alitalo, March 8, 2017 (sent for review September 28, 2016; reviewed by Joseph Schlessinger and Michel O. Steinmetz) The endothelial cell (EC)-specific receptor tyrosine kinases mice lacking Tie1 develop severe edema around E13.5 because Tie1 and Tie2 are necessary for the remodeling and maturation of compromised microvessel integrity and defects in lymphatic of blood and lymphatic vessels. Angiopoietin-1 (Ang1) growth vasculature and die subsequently (15, 16). Furthermore, Tie1 has factor is a Tie2 agonist, whereas Ang2 functions as a context- critical functions in vascular pathologies, e.g., in tumor angio- dependent agonist/antagonist. The orphan receptor Tie1 modu- genesis and atherosclerosis progression (12, 17). lates Tie2 activation, which is induced by association of angio- In EC monolayers, angiopoietins stimulate Tie receptor trans- cis – trans location to cell–cell junctions for Tie2 trans-association, whereas poietins with Tie2 in and across EC EC junctions in . – Except for the binding of the C-terminal angiopoietin domains in the absence of cell cell adhesion the Tie receptors are an- to the Tie2 ligand-binding domain, the mechanisms for Tie2 chored to the extracellular matrix (ECM) by Ang1-induced Tie2 cis-association (10, 18). Integrins also have been implicated in activation are poorly understood. We report here the structural α β – basis of Ang1-induced Tie2 dimerization in cis and provide Tie2 signaling, and the 5 1 integrin heterodimer enhances Ang1-induced EC adhesion and Tie2 activation (13, 19, 20). -

LAC.12 Embryology 2019-2020 Dr.Mahdi Alheety
LAC.12 Embryology 2019-2020 Dr.Mahdi ALheety Cardiovascular System Establishment of the Cardiogenic Field The vascular system appears in the middle of the third week, when the embryo is no longer able to satisfy its nutritional requirements by diffusion alone. Progenitor heart cells lie in the epiblast, immediately adjacent to the cranial end of the primitive streak. From there, they migrate through the streak and into the splanchnic layer of lateral plate mesoderm where they form a horseshoe-shaped cluster of cells called the primary heart field (PHF) cranial to the neural folds. As the progenitor heart cells migrate and form the PHF during days 16 to18, they are specified on both sides from lateral to medial to become the atria, left ventricle, and most of the right ventricle. Patterning of these cells occurs at the same time that laterality (left-right sidedness) is being established for the entire embryo and this process and the signaling pathway it is dependent upon is essential for normal heart development. The remainder of the heart, including part of the right ventricle and outflow tract (conus cordis and truncus arteriosus), is derived from the secondary heart field (SHF). This field of cells appears slightly later (days 20 to 21) than those in the PHF, resides in splanchnic mesoderm ventral to the posterior pharynx, and is responsible for lengthening the outflow tract. Cells in the SHF also exhibit laterality, such that those on the right side contribute to the left of the outflow tract region and those on the left contribute to the right. -

Supplementary Data ASXL2 Regulates Hematopoiesis in Mice and Its
Supplementary data ASXL2 regulates hematopoiesis in mice and its deficiency promotes myeloid expansion Supplementary Methods Genomic DNA extraction Genomic DNA was extracted from BM mononuclear cells using a DNA extraction kit (Puregene Gentra System, Minneapolis, MN, USA) according to the manufacturer’s instructions. Genomic DNA was quantified using Qubit Fluorometer (Life Technologies) and DNA integrity was assessed by agarose gel electrophoresis. For samples with low quantity, DNA was amplified using REPLI-g Ultrafast mini kit (Qiagen). Peripheral blood analysis Complete peripheral blood counts were analysed using Abbott Cell-Dyn 3700 hematology analyzer (Abbott Laboratories). Expression analysis of Asxl2 and Asxl1 Transcript levels of Asxl2 and Asxl1 were estimated using quantitative RT-PCR with following primers: Asxl2 primer set 1, ATTCGACAAGAGATTGAGAAGGAG (forward) and TTTCTGTGAATCTTCAAGGCTTAG (reverse); Asxl2 primer set 2, GCCCTTAACAATGAGTTCTTCACT (forward) and TCCACAGCTCTACTTTCTTCTCCT (reverse); Asxl1 primers, GGTGGAACAATGGAAGGAAA (forward) and CTGGCCGAGAACGTTTCTTA (reverse). Asxl2 protein was detected in immunoblot using anti-ASXL2 antibody (Bethyl). In vitro differentiation of bone marrow cells Bone marrow (BM) cells were cultured in IMDM containing 20% FBS and 10 ng/ml IL3, 10 ng/ml IL6, 20 ng/ml SCF and 20 ng/ml GMCSF for two weeks. For FACS analysis, cells were washed, stained with fluorochrome-conjugated antibodies and analysed on FACS LSR II flow cytometer (BD Biosciences) using FACSDIVA software (BD Biosciences). Colony re-plating assay Bone marrow cells were plated in methylcellulose medium containing mouse stem cell factor (SCF), mouse interleukin 3 (IL-3), human interleukin 6 (IL-6) and human erythropoietin (MethoCult GF M3434; StemCell Technologies). Colonies were enumerated after 9-12 days and cells were harvested for re-plating.