Assessing the Range of Kinase Autoinhibition Mechanisms in the Insulin Receptor Family Stephen C
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

PTPRA Phosphatase Regulates GDNF-Dependent RET Signaling and Inhibits the RET Mutant MEN2A Oncogenic Potential
Journal Pre-proof PTPRA phosphatase regulates GDNF-dependent RET signaling and inhibits the RET mutant MEN2A oncogenic potential Leena Yadav, Elina Pietilä, Tiina Öhman, Xiaonan Liu, Arun K. Mahato, Yulia Sidorova, Kaisa Lehti, Mart Saarma, Markku Varjosalo PII: S2589-0042(20)30055-9 DOI: https://doi.org/10.1016/j.isci.2020.100871 Reference: ISCI 100871 To appear in: ISCIENCE Received Date: 3 August 2019 Revised Date: 15 January 2020 Accepted Date: 26 January 2020 Please cite this article as: Yadav, L., Pietilä, E., Öhman, T., Liu, X., Mahato, A.K., Sidorova, Y., Lehti, K., Saarma, M., Varjosalo, M., PTPRA phosphatase regulates GDNF-dependent RET signaling and inhibits the RET mutant MEN2A oncogenic potential, ISCIENCE (2020), doi: https://doi.org/10.1016/ j.isci.2020.100871. This is a PDF file of an article that has undergone enhancements after acceptance, such as the addition of a cover page and metadata, and formatting for readability, but it is not yet the definitive version of record. This version will undergo additional copyediting, typesetting and review before it is published in its final form, but we are providing this version to give early visibility of the article. Please note that, during the production process, errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain. © 2020 Growth factor RET PTPRA Cell surface Ras P P Complex formation RAF MEK ERK Growth Nucleus Proliferation Gene expression Migration 1 PTPRA phosphatase regulates GDNF-dependent RET signaling and inhibits the RET mutant MEN2A oncogenic potential Authors Leena Yadav 1, Elina Pietilä 3# , Tiina Öhman 1# , Xiaonan Liu 1, Arun K. -
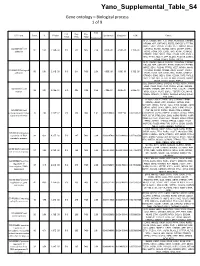
Supplemental Tables4.Pdf
Yano_Supplemental_Table_S4 Gene ontology – Biological process 1 of 9 Fold List Pop Pop GO Term Count % PValue Bonferroni Benjamini FDR Genes Total Hits Total Enrichment DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0007155~cell CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0022610~biological CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DCC, ENAH, PLXNA2, CAPZA2, ATP5B, ASTN1, PAX6, ZEB2, CDH2, CDH4, GLI3, CD24A, EPHB1, NRCAM, GO:0006928~cell CTTNBP2, EDNRB, APP, PTK2, ETV1, CLASP2, STRBP, 36 4.04 3.46E-07 510 205 7436 2.56 7.28E-04 3.64E-04 5.98E-04 motion NRG1, DCLK1, PLAT, SGPL1, TGFBR1, EVL, MYH9, YWHAE, NCKAP1, CTNNA2, SEMA6A, EPHA4, NDEL1, FYN, LRP6 PLXNA2, ADCY5, PAX6, GLI3, CTNNB1, LPHN2, EDNRB, LPHN3, APP, CSNK2A1, GPR45, NRG1, RAPGEF1, WWOX, SGPL1, TLE4, SPEN, NCAM1, DDR1, GRB10, GRM3, GNAQ, HIPK1, GNB1, HIPK2, PYGO1, GO:0007166~cell RNF138, ROR2, CNTN1, -

Ptk7-Deficient Mice Have Decreased Hematopoietic Stem Cell Pools As a Result of Deregulated Proliferation and Migration
Ptk7-Deficient Mice Have Decreased Hematopoietic Stem Cell Pools as a Result of Deregulated Proliferation and Migration This information is current as Anne-Catherine Lhoumeau, Marie-Laure Arcangeli, Maria of September 24, 2021. De Grandis, Marilyn Giordano, Jean-Christophe Orsoni, Frédérique Lembo, Florence Bardin, Sylvie Marchetto, Michel Aurrand-Lions and Jean-Paul Borg J Immunol 2016; 196:4367-4377; Prepublished online 18 April 2016; Downloaded from doi: 10.4049/jimmunol.1500680 http://www.jimmunol.org/content/196/10/4367 Supplementary http://www.jimmunol.org/content/suppl/2016/04/16/jimmunol.150068 http://www.jimmunol.org/ Material 0.DCSupplemental References This article cites 55 articles, 24 of which you can access for free at: http://www.jimmunol.org/content/196/10/4367.full#ref-list-1 Why The JI? Submit online. by guest on September 24, 2021 • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright -

Functional Analysis of Somatic Mutations Affecting Receptor Tyrosine Kinase Family in Metastatic Colorectal Cancer
Author Manuscript Published OnlineFirst on March 29, 2019; DOI: 10.1158/1535-7163.MCT-18-0582 Author manuscripts have been peer reviewed and accepted for publication but have not yet been edited. Functional analysis of somatic mutations affecting receptor tyrosine kinase family in metastatic colorectal cancer Leslie Duplaquet1, Martin Figeac2, Frédéric Leprêtre2, Charline Frandemiche3,4, Céline Villenet2, Shéhérazade Sebda2, Nasrin Sarafan-Vasseur5, Mélanie Bénozène1, Audrey Vinchent1, Gautier Goormachtigh1, Laurence Wicquart6, Nathalie Rousseau3, Ludivine Beaussire5, Stéphanie Truant7, Pierre Michel8, Jean-Christophe Sabourin9, Françoise Galateau-Sallé10, Marie-Christine Copin1,6, Gérard Zalcman11, Yvan De Launoit1, Véronique Fafeur1 and David Tulasne1 1 Univ. Lille, CNRS, Institut Pasteur de Lille, UMR 8161 - M3T – Mechanisms of Tumorigenesis and Target Therapies, F-59000 Lille, France. 2 Univ. Lille, Plateau de génomique fonctionnelle et structurale, CHU Lille, F-59000 Lille, France 3 TCBN - Tumorothèque Caen Basse-Normandie, F-14000 Caen, France. 4 Réseau Régional de Cancérologie – OncoBasseNormandie – F14000 Caen – France. 5 Normandie Univ, UNIROUEN, Inserm U1245, IRON group, Rouen University Hospital, Normandy Centre for Genomic and Personalized Medicine, F-76000 Rouen, France. 6 Tumorothèque du C2RC de Lille, F-59037 Lille, France. 7 Department of Digestive Surgery and Transplantation, CHU Lille, Univ Lille, 2 Avenue Oscar Lambret, 59037, Lille Cedex, France. 8 Department of hepato-gastroenterology, Rouen University Hospital, Normandie Univ, UNIROUEN, Inserm U1245, IRON group, F-76000 Rouen, France. 9 Department of Pathology, Normandy University, INSERM 1245, Rouen University Hospital, F 76 000 Rouen, France. 10 Department of Pathology, MESOPATH-MESOBANK, Centre León Bérard, Lyon, France. 11 Thoracic Oncology Department, CIC1425/CLIP2 Paris-Nord, Hôpital Bichat-Claude Bernard, Paris, France. -
HCC and Cancer Mutated Genes Summarized in the Literature Gene Symbol Gene Name References*
HCC and cancer mutated genes summarized in the literature Gene symbol Gene name References* A2M Alpha-2-macroglobulin (4) ABL1 c-abl oncogene 1, receptor tyrosine kinase (4,5,22) ACBD7 Acyl-Coenzyme A binding domain containing 7 (23) ACTL6A Actin-like 6A (4,5) ACTL6B Actin-like 6B (4) ACVR1B Activin A receptor, type IB (21,22) ACVR2A Activin A receptor, type IIA (4,21) ADAM10 ADAM metallopeptidase domain 10 (5) ADAMTS9 ADAM metallopeptidase with thrombospondin type 1 motif, 9 (4) ADCY2 Adenylate cyclase 2 (brain) (26) AJUBA Ajuba LIM protein (21) AKAP9 A kinase (PRKA) anchor protein (yotiao) 9 (4) Akt AKT serine/threonine kinase (28) AKT1 v-akt murine thymoma viral oncogene homolog 1 (5,21,22) AKT2 v-akt murine thymoma viral oncogene homolog 2 (4) ALB Albumin (4) ALK Anaplastic lymphoma receptor tyrosine kinase (22) AMPH Amphiphysin (24) ANK3 Ankyrin 3, node of Ranvier (ankyrin G) (4) ANKRD12 Ankyrin repeat domain 12 (4) ANO1 Anoctamin 1, calcium activated chloride channel (4) APC Adenomatous polyposis coli (4,5,21,22,25,28) APOB Apolipoprotein B [including Ag(x) antigen] (4) AR Androgen receptor (5,21-23) ARAP1 ArfGAP with RhoGAP domain, ankyrin repeat and PH domain 1 (4) ARHGAP35 Rho GTPase activating protein 35 (21) ARID1A AT rich interactive domain 1A (SWI-like) (4,5,21,22,24,25,27,28) ARID1B AT rich interactive domain 1B (SWI1-like) (4,5,22) ARID2 AT rich interactive domain 2 (ARID, RFX-like) (4,5,22,24,25,27,28) ARID4A AT rich interactive domain 4A (RBP1-like) (28) ARID5B AT rich interactive domain 5B (MRF1-like) (21) ASPM Asp (abnormal -

Wnt5a Modulates Integrin Expression in a Receptor-Dependent Manner In
www.nature.com/scientificreports OPEN Wnt5A modulates integrin expression in a receptor‑dependent manner in ovarian cancer cells Vajihe Azimian‑Zavareh 1,2, Zeinab Dehghani‑Ghobadi1, Marzieh Ebrahimi 3*, Kian Mirzazadeh1, Irina Nazarenko4 & Ghamartaj Hossein 1,4* Wnt5A signals through various receptors that confer versatile biological functions. Here, we used Wnt5A overexpressing human ovarian SKOV‑3 and OVCAR‑3 stable clones for assessing integrin expression, cell proliferation, migration, invasion, and the ability of multicellular aggregates (MCAs) formation. We found here, that Wnt5A regulates diferently the expression of its receptors in the stable Wnt5A overexpressing clones. The expression levels of Frizzled (FZD)‑2 and ‑5, were increased in diferent clones. However ROR‑1, ‑2 expression levels were diferently regulated in clones. Wnt5A overexpressing clones showed increased cell proliferation, migration, and clonogenicity. Moreover, Wnt5A overexpressing SKOV‑3 clone showed increased MCAs formation ability. Cell invasion had been increased in OVCAR‑3‑derived clones, while this was decreased in SKOV‑3‑derived clone. Importantly, αv integrin expression levels were increased in all assessed clones, accompanied by increased cell attachment to fbronectin and focal adhesion kinase activity. Moreover, the treatment of clones with Box5 as a Wnt5A/FZD5 antagonist abrogates ITGAV increase, cell proliferation, migration, and their attachment to fbronectin. Accordingly, we observed signifcantly higher expression levels of ITGAV and ITGB3 in human high‑grade serous ovarian cancer specimens and ITGAV correlated positively with Wnt5A in metastatic serous type ovarian cancer. In summary, we hypothesize here, that Wnt5A/FZD‑5 signaling modulate αv integrin expression levels that could be associated with ovarian cancer cell proliferation, migration, and fbronectin attachment. -
Loss of Receptor Tyrosine Kinase-Like Orphan Receptor 2 Impairs The
Lei et al. Stem Cell Research & Therapy (2020) 11:137 https://doi.org/10.1186/s13287-020-01646-2 RESEARCH Open Access Loss of receptor tyrosine kinase-like orphan receptor 2 impairs the osteogenesis of mBMSCs by inhibiting signal transducer and activator of transcription 3 Lizhen Lei1,2, Zhuwei Huang1,2, Jingyi Feng1,2, Zijing Huang1,2, Yiwei Tao1,2, Xiaoli Hu1,2* and Xiaolei Zhang1,2* Abstract Background: Receptor tyrosine kinase-like orphan receptor 2 (Ror2) plays a key role in bone formation, but its signaling pathway is not completely understood. Signal transducer and activator of transcription 3 (Stat3) takes part in maintaining bone homeostasis. The aim of this study is to reveal the role and mechanism of Ror2 in the osteogenic differentiation from mouse bone marrow mesenchymal stem cells (mBMSCs) and to explore the effect of Stat3 on Ror2-mediated osteogenesis. Methods: Ror2 CKO mice were generated via the Cre-loxp recombination system using Prrx1-Cre transgenic mice. Quantitative real-time PCR and western blot were performed to assess the expression of Stat3 and osteogenic markers in Ror2-knockdown mBMSCs (mBMSC-sh-Ror2). After being incubated in osteogenic induction medium for 3 weeks, Alizarin Red staining and western blot were used to examine the calcium deposit and osteogenic markers in Stat3 overexpression in mBMSC-sh-Ror2. Results: Loss of Ror2 in mesenchymal or osteoblast progenitor cells led to a dwarfism phenotype in vivo. The mRNA expression of osteogenic markers (osteocalcin, osteopontin (OPN), and collagen I) in the ulna proximal epiphysis of Ror2 CKO mice was significantly decreased (P < 0.05). -

Global Regulation of Differential Gene Expression by C-Abl/Arg Oncogenic
LAB/IN VITRO RESEARCH e-ISSN 1643-3750 © Med Sci Monit, 2017; 23: 2625-2635 DOI: 10.12659/MSM.904888 Received: 2017.04.16 Accepted: 2017.05.08 Global Regulation of Differential Gene Published: 2017.05.30 Expression by c-Abl/Arg Oncogenic Kinases Authors’ Contribution: ABCE 1 Qincai Dong* 1 Laboratory of Genetic Engineering, Beijing Institute of Biotechnology, Beijing, Study Design A BC 2 Chenggong Li* P.R. China Data Collection B 2 Institute of Health Sciences, Anhui University, Hefei, Anhui, P.R. China Statistical Analysis C BC 3 Xiuhua Qu 3 Center of Basic Medical Sciences, Navy General Hospital of PLA, Beijing, Data Interpretation D AFG 1 Cheng Cao P.R. China Manuscript Preparation E AEG 1 Xuan Liu Literature Search F Funds Collection G * These 2 authors contributed equally to this work Corresponding Authors: Xuan Liu, e-mail: [email protected], Cheng Cao, e-mail: [email protected] Source of support: This work was supported by the National Natural Science Foundation of China [31070674] Background: Studies have found that c-Abl oncogenic kinases may regulate gene transcription by RNA polymerase II phos- phorylation or by direct regulation of specific transcription factors or coactivators. However, the global regu- lation of differential gene expression by c-Abl/Arg is largely unknown. In this study, differentially expressed genes (DEGs) regulated by c-Abl/Arg were identified, and related cellular functions and associated pathways were investigated. Material/Methods: RNA obtained from wild-type and c-Abl/Arg gene-silenced MCF-7 cells was analyzed by RNA-Seq. DEGs were identified using edgeR software and partially validated by qRT-PCR. -

Ror2 Functions As a Noncanonical Wnt Receptor That Regulates NMDAR-Mediated Synaptic Transmission
RoR2 functions as a noncanonical Wnt receptor that regulates NMDAR-mediated synaptic transmission Waldo Cerpaa,1, Elena Latorre-Estevesa,b, and Andres Barriaa,2 aDepartment of Physiology and Biophysics and bMolecular and Cellular Biology Program, University of Washington, Seattle, WA 98195 Edited* by Richard L. Huganir, The Johns Hopkins University School of Medicine, Baltimore, MD, and approved March 6, 2015 (received for review September 16, 2014) Wnt signaling has a well-established role as a regulator of nervous receptor for noncanonical Wnt ligands capable of regulating syn- system development, but its role in the maintenance and regula- aptic NMDARs. In hippocampal neurons, activation of RoR2 by tion of established synapses in the mature brain remains poorly noncanonical Wnt ligand Wnt5a activates PKC and JNK, two understood. At excitatory glutamatergic synapses, NMDA receptors kinases involved in the regulation of NMDAR currents. In ad- (NMDARs) have a fundamental role in synaptogenesis, synaptic dition, we show that signaling through RoR2 is necessary for plasticity, and learning and memory; however, it is not known what the maintenance of basal NMDAR-mediated synaptic trans- controls their number and subunit composition. Here we show that mission and the acute regulation of NMDAR synaptic responses the receptor tyrosine kinase-like orphan receptor 2 (RoR2) func- by Wnt5a. tions as a Wnt receptor required to maintain basal NMDAR- Identification of RoR2 as a Wnt receptor that regulates syn- aptic NMDARs provides a mechanism for Wnt signaling to mediated synaptic transmission. In addition, RoR2 activation by a control synaptic transmission and synaptic plasticity acutely, and noncanonical Wnt ligand activates PKC and JNK and acutely en- is a critical first step toward understanding the role played by hances NMDAR synaptic responses. -

ROR2 Is Epigenetically Inactivated in the Early Stages of Colorectal Neoplasia and Is Associated with Proliferation and Migration Sean S
Ma et al. BMC Cancer (2016) 16:508 DOI 10.1186/s12885-016-2576-7 RESEARCH ARTICLE Open Access ROR2 is epigenetically inactivated in the early stages of colorectal neoplasia and is associated with proliferation and migration Sean S. Q. Ma1, Sameer Srivastava2,3, Estelle Llamosas1, Nicholas J. Hawkins4, Luke B. Hesson2, Robyn L. Ward2 and Caroline E. Ford1* Abstract Background: Colorectal cancer (CRC) is closely linked to Wnt signalling, with 94 % of cases exhibiting a Wnt related mutation. ROR2 is a receptor tyrosine kinase that is thought to repress β-catenin dependent Wnt signalling. Our study aims to determine if ROR2 is epigenetically silenced in CRC and determine if in vitro silencing of ROR2 potentiates Wnt signalling, and alters the proliferative, migratory or invasive potential of cells. Methods: ROR2 expression was examined in CRC cell lines and patient adenomas using qRT-PCR, while COBRA and bisulphite sequencing was used to analyse ROR2 promoter methylation. 258 patient primary tumour samples from publicly available databases were also examined for ROR2 expression and methylation. In addition, the functional effects of ROR2 modulation were investigated in HCT116 cells following ROR2 siRNA knockdown and in RKO and SW620 cells following ectopic ROR2 expression. Results: Reduced ROR2 expression was found to correlate with ROR2 promoter hypermethylation in colorectal cancer cell lines, carcinomas and adenomas. ROR2 expression was downregulated in 76.7 % (23/30) of CRC cell lines with increasing ROR2 promoter hypermethylation correlating with progressively lower expression. Analysis of 239 primary tumour samples from a publicly available cohort also found a significant correlation between reduced ROR2 expression and increased promoter methylation. -

Agrin-Lrp4-Ror2 Signaling Regulates Adult Hippocampal Neurogenesis In
RESEARCH ARTICLE Agrin-Lrp4-Ror2 signaling regulates adult hippocampal neurogenesis in mice Hongsheng Zhang1, Anupama Sathyamurthy2, Fang Liu2, Lei Li1, Lei Zhang1, Zhaoqi Dong1, Wanpeng Cui1, Xiangdong Sun2, Kai Zhao2, Hongsheng Wang1, Hsin-Yi Henry Ho3, Wen-Cheng Xiong1,2,4, Lin Mei1,2,4* 1Department of Neurosciences, School of Medicine, Case Western Reserve University, Cleveland, United States; 2Department of Neuroscience and Regenerative Medicine, Medical College of Georgia, Augusta University, Augusta, United States; 3Department of Neurobiology, Harvard Medical School, Boston, United States; 4Louis Stokes Cleveland Veterans Affairs Medical Center, Cleveland, United States Abstract Adult neurogenesis in the hippocampus may represent a form of plasticity in brain functions including mood, learning and memory. However, mechanisms underlying neural stem/ progenitor cells (NSPCs) proliferation are not well understood. We found that Agrin, a factor critical for neuromuscular junction formation, is elevated in the hippocampus of mice that are stimulated by enriched environment (EE). Genetic deletion of the Agrn gene in excitatory neurons decreases NSPCs proliferation and increases depressive-like behavior. Low-density lipoprotein receptor-related protein 4 (Lrp4), a receptor for Agrin, is expressed in hippocampal NSPCs and its mutation blocked basal as well as EE-induced NSPCs proliferation and maturation of newborn neurons. Finally, we show that Lrp4 interacts with and activates receptor tyrosine kinase-like orphan receptor 2 (Ror2); and Ror2 mutation impairs NSPCs proliferation. Together, these observations identify a role of Agrin-Lrp4-Ror2 signaling for adult neurogenesis, uncovering previously unexpected functions of Agrin and Lrp4 in the brain. *For correspondence: DOI: https://doi.org/10.7554/eLife.45303.001 [email protected] Competing interests: The authors declare that no Introduction competing interests exist. -

Dysregulation of WNT5A/ROR2 Signaling Characterizes The
Published OnlineFirst April 15, 2016; DOI: 10.1158/1541-7786.MCR-15-0484 Signal Transduction Molecular Cancer Research Dysregulation of WNT5A/ROR2 Signaling Characterizes the Progression of Barrett- Associated Esophageal Adenocarcinoma Orestis Lyros1,2, Linghui Nie3, Tami Moore3, Rituparna Medda3, Mary Otterson3, Behnaz Behmaram4, Alexander Mackinnon4, Ines Gockel2, Reza Shaker1, and Parvaneh Rafiee3 Abstract The mechanism underlying the progression of normal survival, and migration in a dose-dependent fashion. esophageal mucosa to esophageal adenocarcinoma remains rhWNT5AwasfoundtoinhibitTOPflash activity in ROR2 elusive. WNT5A is a noncanonical WNT, which mainly func- wild-type cells, whereas increased TOPflash activity in ROR2- tionsviathereceptortyrosinekinase-likeorphanreceptor2 knockdown OE33 cells. In addition, ROR2 knockdown alone (ROR2), and has an unclear role in carcinogenesis. In this abolished cell proliferation andweakenedthemigrationprop- study, we aimed to determine the role of WNT5A/ROR2 erties of OE33 cells. These findings support an early dysregula- signaling in esophageal adenocarcinoma. Analysis of WNT5A tion of the noncanonical WNT5A/ROR2 pathway in the path- and ROR2 expression patterns in healthy controls, Barrett and ogenesis of esophageal adenocarcinoma, with the loss of esophageal adenocarcinoma patients' esophageal clinical spe- WNT5A expression together with the ROR2 overexpression to cimens as well as in various esophageal cell lines demonstrated be consistent with tumor promotion. a ROR2 overexpression in esophageal adenocarcinoma tissues compared with Barrett and healthy mucosa, whereas WNT5A Implications: The dysregulation of WNT5A/ROR2 noncanonical expression was found significantly downregulated toward WNT signaling in Barrett-associated esophageal adenocarcinoma esophageal adenocarcinoma formation. Treatment of esoph- introduces possible prognostic markers and novel targets for ageal adenocarcinoma OE33 cells with human recombinant tailored therapy of this malignancy.