Deciphering the Role of Gfi1b Gene Target Rgs18 in Erythro-Megakaryocytic Lineage Divergence
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
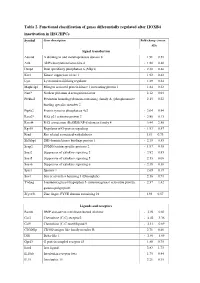
Table 2. Functional Classification of Genes Differentially Regulated After HOXB4 Inactivation in HSC/Hpcs
Table 2. Functional classification of genes differentially regulated after HOXB4 inactivation in HSC/HPCs Symbol Gene description Fold-change (mean ± SD) Signal transduction Adam8 A disintegrin and metalloprotease domain 8 1.91 ± 0.51 Arl4 ADP-ribosylation factor-like 4 - 1.80 ± 0.40 Dusp6 Dual specificity phosphatase 6 (Mkp3) - 2.30 ± 0.46 Ksr1 Kinase suppressor of ras 1 1.92 ± 0.42 Lyst Lysosomal trafficking regulator 1.89 ± 0.34 Mapk1ip1 Mitogen activated protein kinase 1 interacting protein 1 1.84 ± 0.22 Narf* Nuclear prelamin A recognition factor 2.12 ± 0.04 Plekha2 Pleckstrin homology domain-containing. family A. (phosphoinosite 2.15 ± 0.22 binding specific) member 2 Ptp4a2 Protein tyrosine phosphatase 4a2 - 2.04 ± 0.94 Rasa2* RAS p21 activator protein 2 - 2.80 ± 0.13 Rassf4 RAS association (RalGDS/AF-6) domain family 4 3.44 ± 2.56 Rgs18 Regulator of G-protein signaling - 1.93 ± 0.57 Rrad Ras-related associated with diabetes 1.81 ± 0.73 Sh3kbp1 SH3 domain kinase bindings protein 1 - 2.19 ± 0.53 Senp2 SUMO/sentrin specific protease 2 - 1.97 ± 0.49 Socs2 Suppressor of cytokine signaling 2 - 2.82 ± 0.85 Socs5 Suppressor of cytokine signaling 5 2.13 ± 0.08 Socs6 Suppressor of cytokine signaling 6 - 2.18 ± 0.38 Spry1 Sprouty 1 - 2.69 ± 0.19 Sos1 Son of sevenless homolog 1 (Drosophila) 2.16 ± 0.71 Ywhag 3-monooxygenase/tryptophan 5- monooxygenase activation protein. - 2.37 ± 1.42 gamma polypeptide Zfyve21 Zinc finger. FYVE domain containing 21 1.93 ± 0.57 Ligands and receptors Bambi BMP and activin membrane-bound inhibitor - 2.94 ± 0.62 -

Analysis of Chromatin-State Plasticity Identifies Cell-Type–Specific Regulators of H3k27me3 Patterns
Analysis of chromatin-state plasticity identifies cell-type–specific regulators of H3K27me3 patterns Luca Pinelloa,1, Jian Xub,1, Stuart H. Orkinb,c,2, and Guo-Cheng Yuana,2 aDepartment of Biostatistics and Computational Biology, Dana-Farber Cancer Institute and Harvard School of Public Heath, Boston, MA 02215; bDivision of Hematology/Oncology, Boston Children’s Hospital and Department of Pediatric Oncology, Dana-Farber Cancer Institute, Harvard Stem Cell Institute, Harvard Medical School, Boston, MA 02115; and cHoward Hughes Medical Institute, Boston, MA 02115 Contributed by Stuart H. Orkin, December 6, 2013 (sent for review August 13, 2013) Chromatin states are highly cell-type–specific, but the underlying a significant fraction of H3K27me3 is located in distal regions. mechanisms for the establishment and maintenance of their ge- It has been proposed that distal H3K27me3 marks poised nome-wide patterns remain poorly understood. Here we present enhancers, which can be activated through replacing H3K27me3 a computational approach for investigation of chromatin-state by H3K27ac (15, 16). An important question is whether there is plasticity. We applied this approach to investigate an ENCODE a general principle controlling the plastic changes of H3K27me3 ChIP-seq dataset profiling the genome-wide distributions of the patterns across different cell types. H3K27me3 mark in 19 human cell lines. We found that the high To systematically investigate the mechanisms modulating plasticity regions (HPRs) can be divided into two functionally and chromatin-state plasticity, we developed and validated a compu- mechanistically distinct subsets, which correspond to CpG island tational approach to identify distinct lineage-restricted regulators (CGI) proximal or distal regions, respectively. -

Global H3k4me3 Genome Mapping Reveals Alterations of Innate Immunity Signaling and Overexpression of JMJD3 in Human Myelodysplastic Syndrome CD34 Þ Cells
Leukemia (2013) 27, 2177–2186 & 2013 Macmillan Publishers Limited All rights reserved 0887-6924/13 www.nature.com/leu ORIGINAL ARTICLE Global H3K4me3 genome mapping reveals alterations of innate immunity signaling and overexpression of JMJD3 in human myelodysplastic syndrome CD34 þ cells YWei1, R Chen2, S Dimicoli1, C Bueso-Ramos3, D Neuberg4, S Pierce1, H Wang2, H Yang1, Y Jia1, H Zheng1, Z Fang1, M Nguyen3, I Ganan-Gomez1,5, B Ebert6, R Levine7, H Kantarjian1 and G Garcia-Manero1 The molecular bases of myelodysplastic syndromes (MDS) are not fully understood. Trimethylated histone 3 lysine 4 (H3K4me3) is present in promoters of actively transcribed genes and has been shown to be involved in hematopoietic differentiation. We performed a genome-wide H3K4me3 CHIP-Seq (chromatin immunoprecipitation coupled with whole genome sequencing) analysis of primary MDS bone marrow (BM) CD34 þ cells. This resulted in the identification of 36 genes marked by distinct higher levels of promoter H3K4me3 in MDS. A majority of these genes are involved in nuclear factor (NF)-kB activation and innate immunity signaling. We then analyzed expression of histone demethylases and observed significant overexpression of the JmjC-domain histone demethylase JMJD3 (KDM6b) in MDS CD34 þ cells. Furthermore, we demonstrate that JMJD3 has a positive effect on transcription of multiple CHIP-Seq identified genes involved in NF-kB activation. Inhibition of JMJD3 using shRNA in primary BM MDS CD34 þ cells resulted in an increased number of erythroid colonies in samples isolated from patients with lower-risk MDS. Taken together, these data indicate the deregulation of H3K4me3 and associated abnormal activation of innate immunity signals have a role in the pathogenesis of MDS and that targeting these signals may have potential therapeutic value in MDS. -

Rabbit Mab A
C 0 2 - t GFI1b (D3G2) Rabbit mAb a e r o t S Orders: 877-616-CELL (2355) [email protected] Support: 877-678-TECH (8324) 9 4 Web: [email protected] 8 www.cellsignal.com 5 # 3 Trask Lane Danvers Massachusetts 01923 USA For Research Use Only. Not For Use In Diagnostic Procedures. Applications: Reactivity: Sensitivity: MW (kDa): Source/Isotype: UniProt ID: Entrez-Gene Id: WB, F H M R Mk Endogenous 35, 42 Rabbit IgG Q5VTD9 8328 Product Usage Information Application Dilution Western Blotting 1:1000 Flow Cytometry 1:400 - 1:1600 Storage Supplied in 10 mM sodium HEPES (pH 7.5), 150 mM NaCl, 100 µg/ml BSA, 50% glycerol and less than 0.02% sodium azide. Store at –20°C. Do not aliquot the antibody. Specificity / Sensitivity GFI1b (D3G2) Rabbit mAb recognizes endogenous levels of total GFI1b protein. Species Reactivity: Human, Mouse, Rat, Monkey Source / Purification Monoclonal antibody is produced by immunizing animals with a synthetic peptide corresponding to residues surrounding Tyr119 of human GFI1b protein. Background GFI1b and its homolog GFI1 are transcriptional repressors and important regulators of erythroid and megakaryocytic development and differentiation (1,2). GFI1b negatively regulates transcription by recruiting chromatin regulatory proteins including CoREST, the histone demethylase LSD1 and HDACs 1 and 2, which associate with GFI1b via its SNAG repression domain (3). GFI1b has also been shown to control the differentiation of erythroid and megakaryocytic progenitors by regulating TGF-β signaling at the bipotent progenitor stage (4). Inactivation of GFI1b in mice leads to embryonic lethality due to failure to produce functional erythrocytes and megakaryocytes (2). -

Gfi1b (NM 008114) Mouse Tagged ORF Clone – MG204861 | Origene
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for MG204861 Gfi1b (NM_008114) Mouse Tagged ORF Clone Product data: Product Type: Expression Plasmids Product Name: Gfi1b (NM_008114) Mouse Tagged ORF Clone Tag: TurboGFP Symbol: Gfi1b Synonyms: Gfi-1B Vector: pCMV6-AC-GFP (PS100010) E. coli Selection: Ampicillin (100 ug/mL) Cell Selection: Neomycin ORF Nucleotide >MG204861 representing NM_008114 Sequence: Red=Cloning site Blue=ORF Green=Tags(s) TTTTGTAATACGACTCACTATAGGGCGGCCGGGAATTCGTCGACTGGATCCGGTACCGAGGAGATCTGCC GCCGCGATCGCC ATGCCACGGTCCTTTCTAGTGAAGAGTAAGAAGGCACACACTTACCACCAGCCCCGGGCACAGGGTGATG AGCTGGTCTGGCCTCCTGCTGTAATTCCTGTGGCAAAAGAGCATAGCCAGAGTGCCAGCCCTCTTCTCAG CACACCGCTTCCAAGCCAGACCTTGGACTGGAACACAATCAAACAGGAGCGGGAGATGTTGCTGAACCAG AGCCTTCCCAAGATGGCCTCAGCCCCAGAGGGGCCTCTCGTGACACCCCAACCCCAGGATGGGGAATCAC CACTCTCTGAGTCACCCCCTTTCTACAAGCCCAGCTTCTCCTGGGATACCTTGGCCTCCTCCTACAGCCA CAGCTACACACAGACCCCCTCCACCATGCAGTCCGCCTTCCTGGAGCGCTCCGTGAGGCTGTACGGCAGC CCCCTCGTGCCCAGCACAGAGTCTCCCTTGGACTTCCGCCTCCGCTACTCTCCAGGCATGGACACTTACC ACTGTGTCAAGTGCAACAAGGTGTTCTCCACCCCTCATGGGCTAGAAGTGCATGTCCGCCGCTCTCACAG CGGAACCCGGCCCTTTGCCTGTGATGTCTGTGGCAAAACCTTTGGCCACGCTGTGAGCTTGGAGCAGCAT ACTCACGTCCACTCACAGGAGCGAAGCTTCGAGTGCCGGATGTGTGGCAAAGCCTTCAAGCGTTCATCCA CCCTGTCCACCCACCTGCTCATCCACTCGGACACTCGGCCCTACCCCTGCCAGTTCTGTGGGAAGCGCTT CCACCAGAAGTCGGACATGAAGAAACACACCTACATCCACACAGGTGAGAAGCCCCACAAGTGCCAGGTG TGTGGGAAAGCCTTCAGCCAGAGCTCCAACCTCATCACCCACAGCCGCAAGCACACAGGCTTCAAGCCGT -

Emergence of the Noncoding Cancer Genome: a Target of Genetic and Epigenetic Alterations
Published OnlineFirst October 19, 2016; DOI: 10.1158/2159-8290.CD-16-0745 REVIEW Emergence of the Noncoding Cancer Genome: A Target of Genetic and Epigenetic Alterations Stanley Zhou 1 , 2 , Aislinn E. Treloar 1 , 2 , and Mathieu Lupien 1 , 2 , 3 ABSTRACT The emergence of whole-genome annotation approaches is paving the way for the comprehensive annotation of the human genome across diverse cell and tissue types exposed to various environmental conditions. This has already unmasked the positions of thou- sands of functional cis-regulatory elements integral to transcriptional regulation, such as enhancers, promoters, and anchors of chromatin interactions that populate the noncoding genome. Recent studies have shown that cis-regulatory elements are commonly the targets of genetic and epigenetic altera- tions associated with aberrant gene expression in cancer. Here, we review these fi ndings to showcase the contribution of the noncoding genome and its alteration in the development and progression of can- cer. We also highlight the opportunities to translate the biological characterization of genetic and epi- genetic alterations in the noncoding cancer genome into novel approaches to treat or monitor disease. Signifi cance: The majority of genetic and epigenetic alterations accumulate in the noncoding genome throughout oncogenesis. Discriminating driver from passenger events is a challenge that holds great promise to improve our understanding of the etiology of different cancer types. Advancing our under- standing of the noncoding cancer genome may thus identify new therapeutic opportunities and acceler- ate our capacity to fi nd improved biomarkers to monitor various stages of cancer development. Cancer Discov; 6(11); 1215–29. -

Identification of Key Pathways and Genes in Dementia Via Integrated Bioinformatics Analysis
bioRxiv preprint doi: https://doi.org/10.1101/2021.04.18.440371; this version posted July 19, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Identification of Key Pathways and Genes in Dementia via Integrated Bioinformatics Analysis Basavaraj Vastrad1, Chanabasayya Vastrad*2 1. Department of Biochemistry, Basaveshwar College of Pharmacy, Gadag, Karnataka 582103, India. 2. Biostatistics and Bioinformatics, Chanabasava Nilaya, Bharthinagar, Dharwad 580001, Karnataka, India. * Chanabasayya Vastrad [email protected] Ph: +919480073398 Chanabasava Nilaya, Bharthinagar, Dharwad 580001 , Karanataka, India bioRxiv preprint doi: https://doi.org/10.1101/2021.04.18.440371; this version posted July 19, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract To provide a better understanding of dementia at the molecular level, this study aimed to identify the genes and key pathways associated with dementia by using integrated bioinformatics analysis. Based on the expression profiling by high throughput sequencing dataset GSE153960 derived from the Gene Expression Omnibus (GEO), the differentially expressed genes (DEGs) between patients with dementia and healthy controls were identified. With DEGs, we performed a series of functional enrichment analyses. Then, a protein–protein interaction (PPI) network, modules, miRNA-hub gene regulatory network and TF-hub gene regulatory network was constructed, analyzed and visualized, with which the hub genes miRNAs and TFs nodes were screened out. Finally, validation of hub genes was performed by using receiver operating characteristic curve (ROC) analysis. -

Genome Sequencing Unveils a Regulatory Landscape of Platelet Reactivity
ARTICLE https://doi.org/10.1038/s41467-021-23470-9 OPEN Genome sequencing unveils a regulatory landscape of platelet reactivity Ali R. Keramati 1,2,124, Ming-Huei Chen3,4,124, Benjamin A. T. Rodriguez3,4,5,124, Lisa R. Yanek 2,6, Arunoday Bhan7, Brady J. Gaynor 8,9, Kathleen Ryan8,9, Jennifer A. Brody 10, Xue Zhong11, Qiang Wei12, NHLBI Trans-Omics for Precision (TOPMed) Consortium*, Kai Kammers13, Kanika Kanchan14, Kruthika Iyer14, Madeline H. Kowalski15, Achilleas N. Pitsillides4,16, L. Adrienne Cupples 4,16, Bingshan Li 12, Thorsten M. Schlaeger7, Alan R. Shuldiner9, Jeffrey R. O’Connell8,9, Ingo Ruczinski17, Braxton D. Mitchell 8,9, ✉ Nauder Faraday2,18, Margaret A. Taub17, Lewis C. Becker1,2, Joshua P. Lewis 8,9,125 , 2,14,125✉ 3,4,125✉ 1234567890():,; Rasika A. Mathias & Andrew D. Johnson Platelet aggregation at the site of atherosclerotic vascular injury is the underlying patho- physiology of myocardial infarction and stroke. To build upon prior GWAS, here we report on 16 loci identified through a whole genome sequencing (WGS) approach in 3,855 NHLBI Trans-Omics for Precision Medicine (TOPMed) participants deeply phenotyped for platelet aggregation. We identify the RGS18 locus, which encodes a myeloerythroid lineage-specific regulator of G-protein signaling that co-localizes with expression quantitative trait loci (eQTL) signatures for RGS18 expression in platelets. Gene-based approaches implicate the SVEP1 gene, a known contributor of coronary artery disease risk. Sentinel variants at RGS18 and PEAR1 are associated with thrombosis risk and increased gastrointestinal bleeding risk, respectively. Our WGS findings add to previously identified GWAS loci, provide insights regarding the mechanism(s) by which genetics may influence cardiovascular disease risk, and underscore the importance of rare variant and regulatory approaches to identifying loci contributing to complex phenotypes. -

Supplementary Information.Pdf
Supplementary Information Whole transcriptome profiling reveals major cell types in the cellular immune response against acute and chronic active Epstein‐Barr virus infection Huaqing Zhong1, Xinran Hu2, Andrew B. Janowski2, Gregory A. Storch2, Liyun Su1, Lingfeng Cao1, Jinsheng Yu3, and Jin Xu1 Department of Clinical Laboratory1, Children's Hospital of Fudan University, Minhang District, Shanghai 201102, China; Departments of Pediatrics2 and Genetics3, Washington University School of Medicine, Saint Louis, Missouri 63110, United States. Supplementary information includes the following: 1. Supplementary Figure S1: Fold‐change and correlation data for hyperactive and hypoactive genes. 2. Supplementary Table S1: Clinical data and EBV lab results for 110 study subjects. 3. Supplementary Table S2: Differentially expressed genes between AIM vs. Healthy controls. 4. Supplementary Table S3: Differentially expressed genes between CAEBV vs. Healthy controls. 5. Supplementary Table S4: Fold‐change data for 303 immune mediators. 6. Supplementary Table S5: Primers used in qPCR assays. Supplementary Figure S1. Fold‐change (a) and Pearson correlation data (b) for 10 cell markers and 61 hypoactive and hyperactive genes identified in subjects with acute EBV infection (AIM) in the primary cohort. Note: 23 up‐regulated hyperactive genes were highly correlated positively with cytotoxic T cell (Tc) marker CD8A and NK cell marker CD94 (KLRD1), and 38 down‐regulated hypoactive genes were highly correlated positively with B cell, conventional dendritic cell -

The Genetic Basis of Emotional Behaviour in Mice
European Journal of Human Genetics (2006) 14, 721–728 & 2006 Nature Publishing Group All rights reserved 1018-4813/06 $30.00 www.nature.com/ejhg REVIEW The genetic basis of emotional behaviour in mice Saffron AG Willis-Owen*,1 and Jonathan Flint1 1Wellcome Trust Centre for Human Genetics, Roosevelt Drive, Headington, Oxford, UK The last decade has witnessed a steady expansion in the number of quantitative trait loci (QTL) mapped for complex phenotypes. However, despite this proliferation, the number of successfully cloned QTL has remained surprisingly low, and to a great extent limited to large effect loci. In this review, we follow the progress of one complex trait locus; a low magnitude moderator of murine emotionality identified some 10 years ago in a simple two-strain intercross, and successively resolved using a variety of crosses and fear-related phenotypes. These experiments have revealed a complex underlying genetic architecture, whereby genetic effects fractionate into several separable QTL with some evidence of phenotype specificity. Ultimately, we describe a method of assessing gene candidacy, and show that given sufficient access to genetic diversity and recombination, progression from QTL to gene can be achieved even for low magnitude genetic effects. European Journal of Human Genetics (2006) 14, 721–728. doi:10.1038/sj.ejhg.5201569 Keywords: mouse; emotionality; quantitative trait locus; QTL Introduction tude genetic effects and their interactions (both with other Emotionality is a psychological trait of complex aetiology, genetic loci (ie epistasis) and nongenetic (environmental) which moderates an organism’s response to stress. Beha- factors) ultimately producing a quasi-continuously distrib- vioural evidence of emotionality has been documented uted phenotype. -

Wppi: Weighting Protein-Protein Interactions
Package ‘wppi’ September 23, 2021 Type Package Title Weighting protein-protein interactions Version 1.1.2 Description Protein-protein interaction data is essential for omics data analysis and modeling. Database knowledge is general, not specific for cell type, physiological condition or any other context determining which connections are functional and contribute to the signaling. Functional annotations such as Gene Ontology and Human Phenotype Ontology might help to evaluate the relevance of interactions. This package predicts functional relevance of protein-protein interactions based on functional annotations such as Human Protein Ontology and Gene Ontology, and prioritizes genes based on network topology, functional scores and a path search algorithm. License MIT + file LICENSE URL https://github.com/AnaGalhoz37/wppi BugReports https://github.com/AnaGalhoz37/wppi/issues biocViews GraphAndNetwork, Network, Pathways, Software, GeneSignaling, GeneTarget, SystemsBiology, Transcriptomics, Annotation Encoding UTF-8 VignetteBuilder knitr Depends R(>= 4.1) Imports dplyr, igraph, logger, methods, magrittr, Matrix, OmnipathR(>= 2.99.8), progress, purrr, rlang, RCurl, stats, tibble, tidyr Suggests knitr, testthat, rmarkdown RoxygenNote 7.1.1 NeedsCompilation no git_url https://git.bioconductor.org/packages/wppi git_branch master git_last_commit 6aa91be git_last_commit_date 2021-07-15 1 2 common_neighbors Date/Publication 2021-09-23 Author Ana Galhoz [cre, aut] (<https://orcid.org/0000-0001-7402-5292>), Denes Turei [aut] (<https://orcid.org/0000-0002-7249-9379>), Michael P. Menden [aut] (<https://orcid.org/0000-0003-0267-5792>), Albert Krewinkel [ctb, cph] (pagebreak Lua filter) Maintainer Ana Galhoz <[email protected]> R topics documented: common_neighbors . .2 count_genes . .3 filter_annot_with_network . .4 functional_annot . .5 graph_from_op . .6 in_omnipath . .6 prioritization_genes . .7 process_annot . -

Biological Role and Disease Impact of Copy Number Variation in Complex Disease
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 2014 Biological Role and Disease Impact of Copy Number Variation in Complex Disease Joseph Glessner University of Pennsylvania, [email protected] Follow this and additional works at: https://repository.upenn.edu/edissertations Part of the Bioinformatics Commons, and the Genetics Commons Recommended Citation Glessner, Joseph, "Biological Role and Disease Impact of Copy Number Variation in Complex Disease" (2014). Publicly Accessible Penn Dissertations. 1286. https://repository.upenn.edu/edissertations/1286 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/edissertations/1286 For more information, please contact [email protected]. Biological Role and Disease Impact of Copy Number Variation in Complex Disease Abstract In the human genome, DNA variants give rise to a variety of complex phenotypes. Ranging from single base mutations to copy number variations (CNVs), many of these variants are neutral in selection and disease etiology, making difficult the detection of true common orar r e frequency disease-causing mutations. However, allele frequency comparisons in cases, controls, and families may reveal disease associations. Single nucleotide polymorphism (SNP) arrays and exome sequencing are popular assays for genome-wide variant identification. oT limit bias between samples, uniform testing is crucial, including standardized platform versions and sample processing. Bases occupy single points while copy variants occupy segments.