Genome Sequencing Unveils a Regulatory Landscape of Platelet Reactivity
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Identification of Novel Genetic Variants Associated with Short Stature in A
Human Genetics https://doi.org/10.1007/s00439-020-02191-x ORIGINAL INVESTIGATION Identifcation of novel genetic variants associated with short stature in a Baka Pygmies population Matteo Zoccolillo1 · Claudia Moia2 · Sergio Comincini2 · Davide Cittaro3 · Dejan Lazarevic3 · Karen A. Pisani1 · Jan M. Wit4 · Mauro Bozzola5 Received: 1 April 2020 / Accepted: 30 May 2020 © The Author(s) 2020 Abstract Human growth is a complex trait determined by genetic factors in combination with external stimuli, including environment, nutrition and hormonal status. In the past, several genome-wide association studies (GWAS) have collectively identifed hundreds of genetic variants having a putative efect on determining adult height in diferent worldwide populations. Theo- retically, a valuable approach to better understand the mechanisms of complex traits as adult height is to study a population exhibiting extreme stature phenotypes, such as African Baka Pygmies. After phenotypic characterization, we sequenced the whole exomes of a cohort of Baka Pygmies and their non-Pygmies Bantu neighbors to highlight genetic variants associated with the reduced stature. Whole exome data analysis revealed 29 single nucleotide polymorphisms (SNPs) signifcantly associated with the reduced height in the Baka group. Among these variants, we focused on SNP rs7629425, located in the 5′-UTR of the Hyaluronidase-2 (HYAL2) gene. The frequency of the alternative allele was signifcantly increased compared to African and non-African populations. In vitro luciferase assay showed signifcant diferences in transcription modulation by rs7629425 C/T alleles. In conclusion, our results suggested that the HYAL2 gene variants may play a role in the etiology of short stature in Baka Pygmies population. -
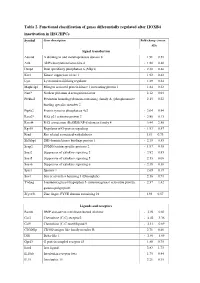
Table 2. Functional Classification of Genes Differentially Regulated After HOXB4 Inactivation in HSC/Hpcs
Table 2. Functional classification of genes differentially regulated after HOXB4 inactivation in HSC/HPCs Symbol Gene description Fold-change (mean ± SD) Signal transduction Adam8 A disintegrin and metalloprotease domain 8 1.91 ± 0.51 Arl4 ADP-ribosylation factor-like 4 - 1.80 ± 0.40 Dusp6 Dual specificity phosphatase 6 (Mkp3) - 2.30 ± 0.46 Ksr1 Kinase suppressor of ras 1 1.92 ± 0.42 Lyst Lysosomal trafficking regulator 1.89 ± 0.34 Mapk1ip1 Mitogen activated protein kinase 1 interacting protein 1 1.84 ± 0.22 Narf* Nuclear prelamin A recognition factor 2.12 ± 0.04 Plekha2 Pleckstrin homology domain-containing. family A. (phosphoinosite 2.15 ± 0.22 binding specific) member 2 Ptp4a2 Protein tyrosine phosphatase 4a2 - 2.04 ± 0.94 Rasa2* RAS p21 activator protein 2 - 2.80 ± 0.13 Rassf4 RAS association (RalGDS/AF-6) domain family 4 3.44 ± 2.56 Rgs18 Regulator of G-protein signaling - 1.93 ± 0.57 Rrad Ras-related associated with diabetes 1.81 ± 0.73 Sh3kbp1 SH3 domain kinase bindings protein 1 - 2.19 ± 0.53 Senp2 SUMO/sentrin specific protease 2 - 1.97 ± 0.49 Socs2 Suppressor of cytokine signaling 2 - 2.82 ± 0.85 Socs5 Suppressor of cytokine signaling 5 2.13 ± 0.08 Socs6 Suppressor of cytokine signaling 6 - 2.18 ± 0.38 Spry1 Sprouty 1 - 2.69 ± 0.19 Sos1 Son of sevenless homolog 1 (Drosophila) 2.16 ± 0.71 Ywhag 3-monooxygenase/tryptophan 5- monooxygenase activation protein. - 2.37 ± 1.42 gamma polypeptide Zfyve21 Zinc finger. FYVE domain containing 21 1.93 ± 0.57 Ligands and receptors Bambi BMP and activin membrane-bound inhibitor - 2.94 ± 0.62 -

HYAL2 Antibody (R30915)
HYAL2 Antibody (R30915) Catalog No. Formulation Size R30915 0.5mg/ml if reconstituted with 0.2ml sterile DI water 100 ug Bulk quote request Availability 1-3 business days Species Reactivity Human, Mouse, Rat. Format Antigen affinity purified Clonality Polyclonal (rabbit origin) Isotype Rabbit IgG Purity Antigen affinity Buffer Lyophilized from 1X PBS with 2.5% BSA and 0.025% sodium azide/thimerosal UniProt Q12891 Applications Western blot : 0.5-1ug/ml Limitations This HYAL2 antibody is available for research use only. Western blot testing of HYAL2 antibody and Lane 1: HeLa; 2: SMMC-7721; 3: COLO320; 4: MCF-7; 5: HT1080 cell lysate Description Hyaluronoglucosaminidase 2, also known as hyaluronidase 2, and LuCa-2, is an enzyme that in humans is encoded by the HYAL2 gene. HYAL2 cDNAs encode a preprotein with an N-terminal signal peptide. Northern blot analysis indicated that the gene was expressed in all human tissues tested except adult brain, and Western blot analysis detected the protein in all mouse tissues examined except adult brain. HYAL2 is a glycosylphosphatidylinositol (GPI)-anchored protein on the cell surface and serves as a receptor for entry into the cell of the jaagsiekte sheep retrovirus (JSRV). The findings of Rai et al.(2001) that HYAL2 is a GPI-anchored protein on the cell surface showed that GFP would likely be cleaved from HYAL2 during GPI addition, leaving HYAL2 on the cell surface and resulting in GFP transit to the lysosome for degradation. Application Notes The stated application concentrations are suggested starting amounts. Titration of the HYAL2 antibody may be required due to differences in protocols and secondary/substrate sensitivity. -

Global H3k4me3 Genome Mapping Reveals Alterations of Innate Immunity Signaling and Overexpression of JMJD3 in Human Myelodysplastic Syndrome CD34 Þ Cells
Leukemia (2013) 27, 2177–2186 & 2013 Macmillan Publishers Limited All rights reserved 0887-6924/13 www.nature.com/leu ORIGINAL ARTICLE Global H3K4me3 genome mapping reveals alterations of innate immunity signaling and overexpression of JMJD3 in human myelodysplastic syndrome CD34 þ cells YWei1, R Chen2, S Dimicoli1, C Bueso-Ramos3, D Neuberg4, S Pierce1, H Wang2, H Yang1, Y Jia1, H Zheng1, Z Fang1, M Nguyen3, I Ganan-Gomez1,5, B Ebert6, R Levine7, H Kantarjian1 and G Garcia-Manero1 The molecular bases of myelodysplastic syndromes (MDS) are not fully understood. Trimethylated histone 3 lysine 4 (H3K4me3) is present in promoters of actively transcribed genes and has been shown to be involved in hematopoietic differentiation. We performed a genome-wide H3K4me3 CHIP-Seq (chromatin immunoprecipitation coupled with whole genome sequencing) analysis of primary MDS bone marrow (BM) CD34 þ cells. This resulted in the identification of 36 genes marked by distinct higher levels of promoter H3K4me3 in MDS. A majority of these genes are involved in nuclear factor (NF)-kB activation and innate immunity signaling. We then analyzed expression of histone demethylases and observed significant overexpression of the JmjC-domain histone demethylase JMJD3 (KDM6b) in MDS CD34 þ cells. Furthermore, we demonstrate that JMJD3 has a positive effect on transcription of multiple CHIP-Seq identified genes involved in NF-kB activation. Inhibition of JMJD3 using shRNA in primary BM MDS CD34 þ cells resulted in an increased number of erythroid colonies in samples isolated from patients with lower-risk MDS. Taken together, these data indicate the deregulation of H3K4me3 and associated abnormal activation of innate immunity signals have a role in the pathogenesis of MDS and that targeting these signals may have potential therapeutic value in MDS. -

Expression of the POTE Gene Family in Human Ovarian Cancer Carter J Barger1,2, Wa Zhang1,2, Ashok Sharma 1,2, Linda Chee1,2, Smitha R
www.nature.com/scientificreports OPEN Expression of the POTE gene family in human ovarian cancer Carter J Barger1,2, Wa Zhang1,2, Ashok Sharma 1,2, Linda Chee1,2, Smitha R. James3, Christina N. Kufel3, Austin Miller 4, Jane Meza5, Ronny Drapkin 6, Kunle Odunsi7,8,9, 2,10 1,2,3 Received: 5 July 2018 David Klinkebiel & Adam R. Karpf Accepted: 7 November 2018 The POTE family includes 14 genes in three phylogenetic groups. We determined POTE mRNA Published: xx xx xxxx expression in normal tissues, epithelial ovarian and high-grade serous ovarian cancer (EOC, HGSC), and pan-cancer, and determined the relationship of POTE expression to ovarian cancer clinicopathology. Groups 1 & 2 POTEs showed testis-specifc expression in normal tissues, consistent with assignment as cancer-testis antigens (CTAs), while Group 3 POTEs were expressed in several normal tissues, indicating they are not CTAs. Pan-POTE and individual POTEs showed signifcantly elevated expression in EOC and HGSC compared to normal controls. Pan-POTE correlated with increased stage, grade, and the HGSC subtype. Select individual POTEs showed increased expression in recurrent HGSC, and POTEE specifcally associated with reduced HGSC OS. Consistent with tumors, EOC cell lines had signifcantly elevated Pan-POTE compared to OSE and FTE cells. Notably, Group 1 & 2 POTEs (POTEs A/B/B2/C/D), Group 3 POTE-actin genes (POTEs E/F/I/J/KP), and other Group 3 POTEs (POTEs G/H/M) show within-group correlated expression, and pan-cancer analyses of tumors and cell lines confrmed this relationship. Based on their restricted expression in normal tissues and increased expression and association with poor prognosis in ovarian cancer, POTEs are potential oncogenes and therapeutic targets in this malignancy. -

Molecular Basis, Diagnosis and Clinical Management Of
cardio_special issue 2013_biomarin:Hrev_master 05/03/13 09.19 Pagina 2 Cardiogenetics 2013; 3(s1):exxxx3(s1):e2 Molecular basis, diagnosis storage of glycosaminoglycans (GAGs), previ- ously called mucopolysaccharides.1 The defi- Correspondence: Rossella Parini, Department of and clinical management of ciency of one of the enzymes participating in Pediatrics, San Gerardo Hospital, Via Pergolesi mucopolysaccharidoses the GAGs degradation pathway causes progres- 33, 20900 Monza, Italy. sive storage in the lysosomes and consequently Tel. +39.039.2333286 - Fax: +39.039.2334364. E-mail: [email protected] Rossella Parini,1 Francesca Bertola,2 in the cells and results in tissues and organs 3 Pierluigi Russo dysfunction. The damage is both direct or by Key words: mucopolysaccharidoses (MPS), heart, 1UOS Malattie Metaboliche Rare, activation of secondary and tertiary pathways heart and MPS, genetics and MPS. 2 Department of Pediatrics, Fondazione among which a role is played by inflammation. The incidence of MPSs as a group is reported Acknowledgments: RP and PR acknowledge MBBM, Azienda Ospedaliera San between 1:25000 and 1:45000.3 At present 11 dif- patients and their families and Drs. Andrea Gerardo, University of Milano-Bicocca; ferent enzyme deficiencies are involved in Imperatori and Lucia Boffi who collaborated in 2Consortium for Human Molecular the clinical cardiological evaluation of MPSs MPSs producing 7 distinct clinical phenotypes1 Genetics, University of Milano-Bicocca; patients. RP thanks Fondazione Pierfranco e (Table 1).4 Depending on the enzyme deficien- 3UO Department of Cardiology, Azienda Luisa Mariani of Milano for providing financial cy, the catabolism of dermatan sulphate, support for clinical assistance to metabolic Ospedaliera San Gerardo, Monza, Italy heparan sulphate, keratan sulphate, chon- patients and Mrs. -

Identification of Key Pathways and Genes in Dementia Via Integrated Bioinformatics Analysis
bioRxiv preprint doi: https://doi.org/10.1101/2021.04.18.440371; this version posted July 19, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Identification of Key Pathways and Genes in Dementia via Integrated Bioinformatics Analysis Basavaraj Vastrad1, Chanabasayya Vastrad*2 1. Department of Biochemistry, Basaveshwar College of Pharmacy, Gadag, Karnataka 582103, India. 2. Biostatistics and Bioinformatics, Chanabasava Nilaya, Bharthinagar, Dharwad 580001, Karnataka, India. * Chanabasayya Vastrad [email protected] Ph: +919480073398 Chanabasava Nilaya, Bharthinagar, Dharwad 580001 , Karanataka, India bioRxiv preprint doi: https://doi.org/10.1101/2021.04.18.440371; this version posted July 19, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract To provide a better understanding of dementia at the molecular level, this study aimed to identify the genes and key pathways associated with dementia by using integrated bioinformatics analysis. Based on the expression profiling by high throughput sequencing dataset GSE153960 derived from the Gene Expression Omnibus (GEO), the differentially expressed genes (DEGs) between patients with dementia and healthy controls were identified. With DEGs, we performed a series of functional enrichment analyses. Then, a protein–protein interaction (PPI) network, modules, miRNA-hub gene regulatory network and TF-hub gene regulatory network was constructed, analyzed and visualized, with which the hub genes miRNAs and TFs nodes were screened out. Finally, validation of hub genes was performed by using receiver operating characteristic curve (ROC) analysis. -

Genetic Deletions in Sputum As Diagnostic Markers for Early Detection of Stage I Non ^ Small Cell Lung Cancer Ruiyun Li,1Nevins W
Imaging, Diagnosis, Prognosis Genetic Deletions in Sputum as Diagnostic Markers for Early Detection of Stage I Non ^ Small Cell Lung Cancer Ruiyun Li,1Nevins W. Todd,2 Qi Qiu,3 Ta o F a n , 3 Richard Y. Zhao,3 William H. Rodgers,3 Hong-Bin Fang,4 Ruth L. Katz,5 Sanford A. Stass,3 and Feng Jiang3 Abstract Purpose: Analysis of molecular genetic markers in biological fluids has been proposed as a powerful tool for cancer diagnosis. We have characterized in detail the genetic signatures in primary non ^ small cell lung cancer, which provided potential diagnostic biomarkers for lung cancer. The aim of this study was to determine whether the genetic changes can be used as markers in sputum specimen for the early detection of lung cancer. Experimental Design: Genetic aberrations in the genes HYAL2, FHIT,andSFTPC were evalu- ated in paired tumors and sputum samples from 38 patients with stage I non ^ small cell lung cancer and in sputum samples from 36 cancer-free smokers and 28 healthy nonsmokers by using fluorescence in situ hybridization. Results: HYAL2 and FHIT weredeletedin84%and79%tumorsandin45%and40%paired sputum, respectively. SFTPC was deleted exclusively in tumor tissues (71%).There was concor- dance of HYAL2 or FHIT deletions in matched sputum and tumor tissues from lung cancer patients (r =0.82,P =0.04;r =0.84,P = 0.03), suggesting that the genetic changes in sputum might indicate the presence of the same genetic aberrations in lung tumors. Furthermore, abnor- mal cells were found in 76% sputum by detecting combined HYAL2 and FHIT deletions whereas in 47% sputum by cytology, of the cancer cases, implying that detecting the combination of HYAL2 and FHIT deletions had higher sensitivity than that of sputum cytology for lung cancer diagnosis. -

Role and Regulation of the P53-Homolog P73 in the Transformation of Normal Human Fibroblasts
Role and regulation of the p53-homolog p73 in the transformation of normal human fibroblasts Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen Julius-Maximilians-Universität Würzburg vorgelegt von Lars Hofmann aus Aschaffenburg Würzburg 2007 Eingereicht am Mitglieder der Promotionskommission: Vorsitzender: Prof. Dr. Dr. Martin J. Müller Gutachter: Prof. Dr. Michael P. Schön Gutachter : Prof. Dr. Georg Krohne Tag des Promotionskolloquiums: Doktorurkunde ausgehändigt am Erklärung Hiermit erkläre ich, dass ich die vorliegende Arbeit selbständig angefertigt und keine anderen als die angegebenen Hilfsmittel und Quellen verwendet habe. Diese Arbeit wurde weder in gleicher noch in ähnlicher Form in einem anderen Prüfungsverfahren vorgelegt. Ich habe früher, außer den mit dem Zulassungsgesuch urkundlichen Graden, keine weiteren akademischen Grade erworben und zu erwerben gesucht. Würzburg, Lars Hofmann Content SUMMARY ................................................................................................................ IV ZUSAMMENFASSUNG ............................................................................................. V 1. INTRODUCTION ................................................................................................. 1 1.1. Molecular basics of cancer .......................................................................................... 1 1.2. Early research on tumorigenesis ................................................................................. 3 1.3. Developing -

Different Species Common Arthritis
High Resolution Mapping of Cia3: A Common Arthritis Quantitative Trait Loci in Different Species This information is current as Xinhua Yu, Haidong Teng, Andreia Marques, Farahnaz of September 27, 2021. Ashgari and Saleh M. Ibrahim J Immunol 2009; 182:3016-3023; ; doi: 10.4049/jimmunol.0803005 http://www.jimmunol.org/content/182/5/3016 Downloaded from Supplementary http://www.jimmunol.org/content/suppl/2009/02/18/182.5.3016.DC1 Material References This article cites 36 articles, 9 of which you can access for free at: http://www.jimmunol.org/ http://www.jimmunol.org/content/182/5/3016.full#ref-list-1 Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists by guest on September 27, 2021 • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2009 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology High Resolution Mapping of Cia3: A Common Arthritis Quantitative Trait Loci in Different Species1 Xinhua Yu, Haidong Teng, Andreia Marques, Farahnaz Ashgari, and Saleh M. -

Experimental Eye Research 129 (2014) 93E106
Experimental Eye Research 129 (2014) 93e106 Contents lists available at ScienceDirect Experimental Eye Research journal homepage: www.elsevier.com/locate/yexer Transcriptomic analysis across nasal, temporal, and macular regions of human neural retina and RPE/choroid by RNA-Seq S. Scott Whitmore a, b, Alex H. Wagner a, c, Adam P. DeLuca a, b, Arlene V. Drack a, b, Edwin M. Stone a, b, Budd A. Tucker a, b, Shemin Zeng a, b, Terry A. Braun a, b, c, * Robert F. Mullins a, b, Todd E. Scheetz a, b, c, a Stephen A. Wynn Institute for Vision Research, The University of Iowa, Iowa City, IA, USA b Department of Ophthalmology and Visual Sciences, Carver College of Medicine, The University of Iowa, Iowa City, IA, USA c Department of Biomedical Engineering, College of Engineering, The University of Iowa, Iowa City, IA, USA article info abstract Article history: Proper spatial differentiation of retinal cell types is necessary for normal human vision. Many retinal Received 14 September 2014 diseases, such as Best disease and male germ cell associated kinase (MAK)-associated retinitis pigmen- Received in revised form tosa, preferentially affect distinct topographic regions of the retina. While much is known about the 31 October 2014 distribution of cell types in the retina, the distribution of molecular components across the posterior pole Accepted in revised form 4 November 2014 of the eye has not been well-studied. To investigate regional difference in molecular composition of Available online 5 November 2014 ocular tissues, we assessed differential gene expression across the temporal, macular, and nasal retina and retinal pigment epithelium (RPE)/choroid of human eyes using RNA-Seq. -

Supplementary Information.Pdf
Supplementary Information Whole transcriptome profiling reveals major cell types in the cellular immune response against acute and chronic active Epstein‐Barr virus infection Huaqing Zhong1, Xinran Hu2, Andrew B. Janowski2, Gregory A. Storch2, Liyun Su1, Lingfeng Cao1, Jinsheng Yu3, and Jin Xu1 Department of Clinical Laboratory1, Children's Hospital of Fudan University, Minhang District, Shanghai 201102, China; Departments of Pediatrics2 and Genetics3, Washington University School of Medicine, Saint Louis, Missouri 63110, United States. Supplementary information includes the following: 1. Supplementary Figure S1: Fold‐change and correlation data for hyperactive and hypoactive genes. 2. Supplementary Table S1: Clinical data and EBV lab results for 110 study subjects. 3. Supplementary Table S2: Differentially expressed genes between AIM vs. Healthy controls. 4. Supplementary Table S3: Differentially expressed genes between CAEBV vs. Healthy controls. 5. Supplementary Table S4: Fold‐change data for 303 immune mediators. 6. Supplementary Table S5: Primers used in qPCR assays. Supplementary Figure S1. Fold‐change (a) and Pearson correlation data (b) for 10 cell markers and 61 hypoactive and hyperactive genes identified in subjects with acute EBV infection (AIM) in the primary cohort. Note: 23 up‐regulated hyperactive genes were highly correlated positively with cytotoxic T cell (Tc) marker CD8A and NK cell marker CD94 (KLRD1), and 38 down‐regulated hypoactive genes were highly correlated positively with B cell, conventional dendritic cell