Remodeling of Epigenome and Transcriptome Landscapes with Aging in Mice Reveals Widespread Induction of Inflammatory Responses
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

S41467-020-18249-3.Pdf
ARTICLE https://doi.org/10.1038/s41467-020-18249-3 OPEN Pharmacologically reversible zonation-dependent endothelial cell transcriptomic changes with neurodegenerative disease associations in the aged brain Lei Zhao1,2,17, Zhongqi Li 1,2,17, Joaquim S. L. Vong2,3,17, Xinyi Chen1,2, Hei-Ming Lai1,2,4,5,6, Leo Y. C. Yan1,2, Junzhe Huang1,2, Samuel K. H. Sy1,2,7, Xiaoyu Tian 8, Yu Huang 8, Ho Yin Edwin Chan5,9, Hon-Cheong So6,8, ✉ ✉ Wai-Lung Ng 10, Yamei Tang11, Wei-Jye Lin12,13, Vincent C. T. Mok1,5,6,14,15 &HoKo 1,2,4,5,6,8,14,16 1234567890():,; The molecular signatures of cells in the brain have been revealed in unprecedented detail, yet the ageing-associated genome-wide expression changes that may contribute to neurovas- cular dysfunction in neurodegenerative diseases remain elusive. Here, we report zonation- dependent transcriptomic changes in aged mouse brain endothelial cells (ECs), which pro- minently implicate altered immune/cytokine signaling in ECs of all vascular segments, and functional changes impacting the blood–brain barrier (BBB) and glucose/energy metabolism especially in capillary ECs (capECs). An overrepresentation of Alzheimer disease (AD) GWAS genes is evident among the human orthologs of the differentially expressed genes of aged capECs, while comparative analysis revealed a subset of concordantly downregulated, functionally important genes in human AD brains. Treatment with exenatide, a glucagon-like peptide-1 receptor agonist, strongly reverses aged mouse brain EC transcriptomic changes and BBB leakage, with associated attenuation of microglial priming. We thus revealed tran- scriptomic alterations underlying brain EC ageing that are complex yet pharmacologically reversible. -

Wild-Type Bone Marrow Cells ) Mice with −/− in C1q-Deficient (C1qa
Reconstitution of the Complement Function in C1q-Deficient C1qa؊/؊) Mice with Wild-Type Bone Marrow Cells1) Franz Petry,2* Marina Botto,† Rafaela Holtappels,‡ Mark J. Walport,† and Michael Loos* Besides Ab-independent and Ab-dependent activation of the complement classical pathway in host defense, C1q plays a key role in the processing of immune complexes and in the clearance of apoptotic cells. In humans, C1q deficiency leads to systemic lupus erythematosus-like symptoms in over 90% of the cases, thus making this defect a strong disease susceptibility factor. Similarly, -C1q-deficient mice (C1qa؊/؊) develop systemic lupus erythematosus-like symptoms, such as autoantibodies and glomerulone phritis. We have previously provided evidence that C1q is produced by cells of the monocyte-macrophage lineage. In this study, .we have tested whether transplantation of bone marrow cells would be sufficient to reconstitute C1q levels in C1qa؊/؊ mice C1qa؊/؊ mice received a single graft of 107 bone marrow cells from wild-type (wt) donors after irradiation doses of 6, 7, 8, or 9 .Gy. Engraftment was monitored by a Y chromosome-specific PCR and a PCR that differentiated wt from C1qa؊/؊ genotype Serum levels of C1q Ag and C1 function increased rapidly in the recipient mice, and titers reached normal levels within 6 wk after bone marrow transplantation. In wt mice that received C1qa؊/؊ bone marrow, serum levels of C1q decreased constantly over time and became C1q deficient within 55 wk. These data clearly demonstrate that bone marrow-derived cells are the source of serum C1q and are competent to reconstitute normal C1q serum levels in C1q-deficient mice. -

Adiponectin and Related C1q/TNF-Related Proteins Bind Selectively to Anionic Phospholipids and Sphingolipids
Adiponectin and related C1q/TNF-related proteins bind selectively to anionic phospholipids and sphingolipids Jessica J. Yea,b, Xin Bianc,d, Jaechul Lima,b, and Ruslan Medzhitova,b,1 aHHMI, Yale University, New Haven, CT 06520; bDepartment of Immunobiology, Yale University School of Medicine, New Haven, CT 06520; cDepartment of Cell Biology, Yale University School of Medicine, New Haven, CT 06520; and dDepartment of Neuroscience, Yale University School of Medicine, New Haven, CT 06520 Contributed by Ruslan Medzhitov, April 14, 2020 (sent for review December 20, 2019; reviewed by Ido Amit and G. William Wong) Adiponectin (Acrp30) is an adipokine associated with protection 5-mers of trimers, respectively referred to as low molecular weight from cardiovascular disease, insulin resistance, and inflammation. (LMW), medium molecular weight (MMW), and high molecular Although its effects are conventionally attributed to binding weight (HMW) complexes. Adiponectin can also be cleaved by Adipor1/2 and T-cadherin, its abundance in circulation, role in serum elastases and thrombin between the globular C1q-like head ceramide metabolism, and homology to C1q suggest an overlooked domain and the collagenous tail domain, forming globular adi- role as a lipid-binding protein, possibly generalizable to other C1q/ ponectin (gAd). While gAd appears to bind AdipoR1/2 and ac- TNF-related proteins (CTRPs) and C1q family members. To investi- tivate AMPK more strongly than full-length protein (19, 20), levels gate this, adiponectin, representative family members, and variants of HMW multimers are more strongly associated with insulin were expressed in Expi293 cells and tested for binding to lipids in sensitivity (21, 22), have a longer half-life in circulation (18), and liposomes using density centrifugation. -
Supplementary Figures and Tables
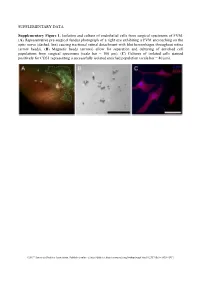
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

Differential and Altered Spatial Distribution of Complement Expression in Age-Related Macular Degeneration
Physiology and Pharmacology Differential and Altered Spatial Distribution of Complement Expression in Age-Related Macular Degeneration John T. Demirs, Junzheng Yang, Maura A. Crowley, Michael Twarog, Omar Delgado, Yubin Qiu, Stephen Poor, Dennis S. Rice, Thaddeus P. Dryja,* Karen Anderson,** and Sha-Mei Liao Department of Ophthalmology, Novartis Institutes for Biomedical Research, Cambridge, Massachusetts, United States Correspondence: Sha-Mei Liao, 22 PURPOSE. Dysregulation of the alternative complement pathway is a major pathogenic Windsor Street, Cambridge, MA mechanism in age-related macular degeneration. We investigated whether locally synthe- 02139, USA; sized complement components contribute to AMD by profiling complement expression [email protected]. in postmortem eyes with and without AMD. Current affiliation: *Cogan Eye METHODS. AMD severity grade 1 to 4 was determined by analysis of postmortem acquired Pathology Laboratory, Massachusetts fundus images and hematoxylin and eosin stained histological sections. TaqMan (donor Eye and Ear, Boston, Massachusetts, eyes n = 39) and RNAscope/in situ hybridization (n = 10) were performed to detect United States. = ** Biogen, Cambridge, complement mRNA. Meso scale discovery assay and Western blot (n 31) were used to Massachusetts, United States. measure complement protein levels. Received: September 2, 2020 RESULTS. The levels of complement mRNA and protein expression were approximately Accepted: May 19, 2021 15- to 100-fold (P < 0.0001–0.001) higher in macular retinal pigment epithelium Published: June 23, 2021 (RPE)/choroid tissue than in neural retina, regardless of AMD grade status. Complement Citation: Demirs JT, Yang J, Crowley mRNA and protein levels were modestly elevated in vitreous and the macular neural MA, et al. -

Exome Sequencing Revealed C1Q Homozygous Mutation in Pediatric
Allergol Immunopathol (Madr). 2018;46(6):594---598 Allergologia et immunopathologia Sociedad Espa ˜nola de Inmunolog´ıa Cl´ınica, Alergolog´ıa y Asma Pedi ´atrica www.elsevier.es/ai ORIGINAL ARTICLE Exome sequencing revealed C1Q homozygous mutation in Pediatric Systemic Lupus Erythematosus a b,c d,e d,e d,e S. Zoghi , V. Ziaee , T. Hirschmugl , R. Jimenez-Heredia , A. Krolo , d,e,f,g a,h,i,∗ K. Boztug , N. Rezaei a Department of Immunology, School of Medicine, Tehran University of Medical Sciences, Tehran, Iran b Division of Pediatric Rheumatology, Children’s Medical Center, Pediatrics Center of Excellence, Tehran University of Medical Sciences, Tehran, Iran c Pediatric Rheumatology Research Group, Rheumatology Research Center, Tehran University of Medical Sciences, Tehran, Iran d CeMM Research Center of Molecular Medicine, Austrian Academy of Sciences, and Division of Neonatal Medicine and Intensive Care, Department of Pediatrics and Adolescent Medicine, Medical University Vienna, Vienna, Austria e Ludwig Boltzmann Institute for Rare and Undiagnosed Diseases, Vienna, Austria f Department of Pediatrics and Adolescent Medicine, Medical University of Vienna, Vienna, Austria g St Anna Kinderspital and Children’s Cancer Research Institute, Department of Pediatrics, Medical University of Vienna, Vienna, Austria h Research Center for Immunodeficiencies, Pediatrics Center of Excellence, Children’s Medical Center, Tehran University of Medical Sciences, Tehran, Iran i Network of Immunity in Infection, Malignancy and Autoimmunity (NIIMA), Universal Scientific Education and Research Network (USERN), Tehran, Iran Received 27 December 2017; accepted 9 February 2018 Available online 5 May 2018 KEYWORDS Abstract Pediatric Systemic Introduction and objectives: Pediatric Systemic Lupus Erythematosus (pSLE) is an autoimmune Lupus Erythematosus; disorder of children. -

Single-Cell Transcriptomes Reveal a Complex Cellular Landscape in the Middle Ear and Differential Capacities for Acute Response to Infection
fgene-11-00358 April 9, 2020 Time: 15:55 # 1 ORIGINAL RESEARCH published: 15 April 2020 doi: 10.3389/fgene.2020.00358 Single-Cell Transcriptomes Reveal a Complex Cellular Landscape in the Middle Ear and Differential Capacities for Acute Response to Infection Allen F. Ryan1*, Chanond A. Nasamran2, Kwang Pak1, Clara Draf1, Kathleen M. Fisch2, Nicholas Webster3 and Arwa Kurabi1 1 Departments of Surgery/Otolaryngology, UC San Diego School of Medicine, VA Medical Center, La Jolla, CA, United States, 2 Medicine/Center for Computational Biology & Bioinformatics, UC San Diego School of Medicine, VA Medical Center, La Jolla, CA, United States, 3 Medicine/Endocrinology, UC San Diego School of Medicine, VA Medical Center, La Jolla, CA, United States Single-cell transcriptomics was used to profile cells of the normal murine middle ear. Clustering analysis of 6770 transcriptomes identified 17 cell clusters corresponding to distinct cell types: five epithelial, three stromal, three lymphocyte, two monocyte, Edited by: two endothelial, one pericyte and one melanocyte cluster. Within some clusters, Amélie Bonnefond, Institut National de la Santé et de la cell subtypes were identified. While many corresponded to those cell types known Recherche Médicale (INSERM), from prior studies, several novel types or subtypes were noted. The results indicate France unexpected cellular diversity within the resting middle ear mucosa. The resolution of Reviewed by: Fabien Delahaye, uncomplicated, acute, otitis media is too rapid for cognate immunity to play a major Institut Pasteur de Lille, France role. Thus innate immunity is likely responsible for normal recovery from middle ear Nelson L. S. Tang, infection. The need for rapid response to pathogens suggests that innate immune The Chinese University of Hong Kong, China genes may be constitutively expressed by middle ear cells. -

Complement C1q Expression in Erythema Nodosum Leprosum
RESEARCH ARTICLE Complement C1q expression in Erythema nodosum leprosum Edessa Negera1,2*, Stephen L. Walker1, Tsehaynesh Lema2, Abraham Aseffa2, Diana N. Lockwood1, Hazel M. Dockrell1 1 London School of Hygiene and Tropical Medicine, Faculty of Infectious Tropical Diseases, London, United Kingdom, 2 Armauer Hansen Research Institute, Addis Ababa, Ethiopia * [email protected] Abstract a1111111111 a1111111111 Complement C1q is a soluble protein capable of initiating components of the classical path- a1111111111 a1111111111 way in host defence system. In earlier qualitative studies, C1q has been implicated in the a1111111111 pathogenesis of Erythema Nodosum Leprosum (ENL). However, little is known about the role of this complement in ENL reaction. In the present study we described the protein level of C1q production and its gene expression in the peripheral blood and skin biopsies in patients with ENL reaction and lepromatous leprosy (LL) patient controls before and after treatment. Thirty untreated patients with ENL reaction and 30 non-reactional LL patient con- OPEN ACCESS trols were recruited at ALERT Hospital, Ethiopia. Peripheral blood and skin biopsies were Citation: Negera E, Walker SL, Lema T, Aseffa A, Lockwood DN, Dockrell HM (2018) Complement obtained from each patient before and after treatment. The level of circulating C1q in the C1q expression in Erythema nodosum leprosum. plasma was determined by enzyme-linked immunosorbent assay. The mRNA expression of PLoS Negl Trop Dis 12(3): e0006321. https://doi. the three C1q components, C1qA, C1qB, and C1qC in the peripheral blood and skin biopsies org/10.1371/journal.pntd.0006321 was determined by qPCR. Circulating C1q in the peripheral blood of untreated ENL patients Editor: Richard Odame Phillips, Kwame Nkrumah was significantly decreased compared to LL patient controls. -

Systems Genetic Discovery of Host-Microbiome Interactions
bioRxiv preprint doi: https://doi.org/10.1101/349605; this version posted June 18, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. 1 Systems genetic discovery of host-microbiome interactions 2 reveals mechanisms of microbial involvement in disease 3 4 5 Running Title: Host genomics, microbial abundance and disease. 6 7 Jason A. Bubier1, Vivek M. Philip1,2, Christopher Quince3, James Campbell4,5, Yanjiao Zhou1 8 Tatiana Vishnivetskaya2, Suman Duvvuru2, Rachel Hageman Blair6, Juliet Ndukum1, Kevin D. 9 Donohue7,8,Charles Phillips9, Carmen M. Foster4, David J. Mellert1, George Weinstock1, 10 Cymbeline T. Culiat2, Erich J. Baker10, Michael A. Langston9, Bruce O’Hara7,11, Anthony V. 11 Palumbo4, Mircea Podar4 and Elissa J. Chesler1,2,4 12 13 14 1The Jackson Laboratory 15 2Genome Science and Technology Program, University of Tennessee and Oak Ridge National 16 Laboratory 17 3School of Engineering, University of Glasgow, Glasgow G12 8LT, UK. 18 4Biosciences Division, Oak Ridge National Laboratory 19 5Department of Natural Sciences, Northwest Missouri State University, Maryville, MO 64468 20 6 Department of Biostatistics, State University of New York at Buffalo, Buffalo, NY 21 7Signal Solutions, LLC, Lexington, KY; 22 8 Electrical and Computer Engineering Department, University of Kentucky, Lexington, KY 23 9 Electrical Engineering and Computer Science, University of Tennessee 24 10School of Engineering & Computer Science, Baylor University, Waco, TX 76798 25 11 Department of Biology, University of Kentucky, Lexington, KY 26 27 Corresponding Author: 28 29 Elissa J. -

Anti-C1QA Antibody (ARG58282)
Product datasheet [email protected] ARG58282 Package: 100 μl anti-C1QA antibody Store at: -20°C Summary Product Description Rabbit Polyclonal antibody recognizes C1QA Tested Reactivity Hu Tested Application WB Host Rabbit Clonality Polyclonal Isotype IgG Target Name C1QA Antigen Species Human Immunogen KLH-conjugated synthetic peptide corresponding to aa. 89-103 (Center) of Human C1QA. Conjugation Un-conjugated Alternate Names Complement C1q subcomponent subunit A Application Instructions Application table Application Dilution WB 1:1000 Application Note * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. Positive Control Human blood plasma Calculated Mw 26 kDa Properties Form Liquid Purification Purification with Protein A and immunogen peptide. Buffer PBS and 0.09% (W/V) Sodium azide. Preservative 0.09% (W/V) Sodium azide. Storage instruction For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C or below. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. Note For laboratory research only, not for drug, diagnostic or other use. Bioinformation www.arigobio.com 1/2 Gene Symbol C1QA Gene Full Name complement component 1, q subcomponent, A chain Background This gene encodes a major constituent of the human complement subcomponent C1q. C1q associates with C1r and C1s in order to yield the first component of the serum complement system. Deficiency of C1q has been associated with lupus erythematosus and glomerulonephritis. -

Subretinal Macrophages Produce Classical Complement Activator C1q
Jiao et al. Molecular Neurodegeneration (2018) 13:45 https://doi.org/10.1186/s13024-018-0278-0 RESEARCH ARTICLE Open Access Subretinal macrophages produce classical complement activator C1q leading to the progression of focal retinal degeneration Haihan Jiao1, Matt Rutar1,2, Nilisha Fernando1, Ted Yednock3, Sethu Sankaranarayanan3, Riemke Aggio-Bruce1, Jan Provis1,4 and Riccardo Natoli1,4* Abstract Background: The role of the alternative complement pathway and its mediation by retinal microglia and macrophages, is well-established in the pathogenesis of Age-Related Macular Degeneration (AMD). However, the contribution of the classical complement pathway towards the progression of retinal degenerations is not fully understood, including the role of complement component 1q (C1q) as a critical activator molecule of the classical pathway. Here, we investigated the contribution of C1q to progressive photoreceptor loss and neuroinflammation in retinal degenerations. Methods: Wild-type (WT), C1qa knockout (C1qa−/−) and mice treated with a C1q inhibitor (ANX-M1; Annexon Biosciences), were exposed to photo-oxidative damage (PD) and were observed for progressive lesion development. Retinal function was assessed by electroretinography, followed by histological analyses to assess photoreceptor degeneration. Retinal inflammation was investigated through complement activation, macrophage recruitment and inflammasome expression using western blotting, qPCR and immunofluorescence. C1q was localised in human AMD donor retinas using immunohistochemistry. Results: PD mice had increased levels of C1qa which correlated with increasing photoreceptor cell death and macrophage recruitment. C1qa−/− mice did not show any differences in photoreceptor loss or inflammation at 7 days compared to WT, however at 14 days after the onset of damage, C1qa−/− retinas displayed less photoreceptor cell death, reduced microglia/macrophage recruitment to the photoreceptor lesion, and higher visual function. -

Complement C1q Subcomponent Subunit A
UCSD MOLECULE PAGES doi:10.6072/H0.MP.A004228.01 Volume 1, Issue 2, 2012 Copyright UC Press, All rights reserved. Review Article Open Access Complement C1q subcomponent subunit A Anjana Chandrasekhar1, Ashok Reddy Dinasarapu1, Andrea J Tenner2, Shankar Subramaniam3 Complement C1q subcomponent subunit A (C1qA) is one of the three components of C1q molecule. Functional C1q is composed of eighteen polypeptide chains: six C1qA chains, six C1qB chains, and six C1qC chains, which are arranged as six heterotrimers of ABC: (ABC)6. Each of the individual C1q polypeptide chain consists of a N-terminal region and a C-terminal globular region (gC1q), of ~135 residues. Each N-terminal consists of 2-11 amino acid segments containing a half-cysteine residue that is involved in formation of inter-chain disulphide bonds, followed by a collagen-like region (CLR) consisting of ~81 residues. The collagen-like regions in A, B and C chains of each heterotrimer come together to form a triple helical collagen like structure. Further, A and B chains in each heterotrimer are bound by a disulphide bond, while C chain forms a disulphide bond with a C chain from the adjoining heterotrimer. Therefore the eighteen subunits come together to form six globular heads (gC1q), which are clusters of 3 independently folded C-terminal domains of the A, B and C chain. These globular domains recognize an array of self, non-self and altered-self ligands. C1q associates with the proenzymes C1r and C1s (2 molecules of each, in the molar ratio of 1:2:2 in a calcium dependent manner) to yield an active C1 complex, the first component of the serum complement system.