Plant Development: Hidden Networks Thomas Berleth
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Introduction to the Cell Cell History Cell Structures and Functions
Introduction to the cell cell history cell structures and functions CK-12 Foundation December 16, 2009 CK-12 Foundation is a non-profit organization with a mission to reduce the cost of textbook materials for the K-12 market both in the U.S. and worldwide. Using an open-content, web-based collaborative model termed the “FlexBook,” CK-12 intends to pioneer the generation and distribution of high quality educational content that will serve both as core text as well as provide an adaptive environment for learning. Copyright ©2009 CK-12 Foundation This work is licensed under the Creative Commons Attribution-Share Alike 3.0 United States License. To view a copy of this license, visit http://creativecommons.org/licenses/by-sa/3.0/us/ or send a letter to Creative Commons, 171 Second Street, Suite 300, San Francisco, California, 94105, USA. Contents 1 Cell structure and function dec 16 5 1.1 Lesson 3.1: Introduction to Cells .................................. 5 3 www.ck12.org www.ck12.org 4 Chapter 1 Cell structure and function dec 16 1.1 Lesson 3.1: Introduction to Cells Lesson Objectives • Identify the scientists that first observed cells. • Outline the importance of microscopes in the discovery of cells. • Summarize what the cell theory proposes. • Identify the limitations on cell size. • Identify the four parts common to all cells. • Compare prokaryotic and eukaryotic cells. Introduction Knowing the make up of cells and how cells work is necessary to all of the biological sciences. Learning about the similarities and differences between cell types is particularly important to the fields of cell biology and molecular biology. -

The Structure, Function, and Biosynthesis of Plant Cell Wall Pectic Polysaccharides
Carbohydrate Research 344 (2009) 1879–1900 Contents lists available at ScienceDirect Carbohydrate Research journal homepage: www.elsevier.com/locate/carres The structure, function, and biosynthesis of plant cell wall pectic polysaccharides Kerry Hosmer Caffall a, Debra Mohnen a,b,* a University of Georgia, Department of Biochemistry and Molecular Biology and Complex Carbohydrate Research Center, 315 Riverbend Road Athens, GA 30602, United States b DOE BioEnergy Science Center (BESC), 315 Riverbend Road Athens, GA 30602, United States article info abstract Article history: Plant cell walls consist of carbohydrate, protein, and aromatic compounds and are essential to the proper Received 18 November 2008 growth and development of plants. The carbohydrate components make up 90% of the primary wall, Received in revised form 4 May 2009 and are critical to wall function. There is a diversity of polysaccharides that make up the wall and that Accepted 6 May 2009 are classified as one of three types: cellulose, hemicellulose, or pectin. The pectins, which are most abun- Available online 2 June 2009 dant in the plant primary cell walls and the middle lamellae, are a class of molecules defined by the pres- ence of galacturonic acid. The pectic polysaccharides include the galacturonans (homogalacturonan, Keywords: substituted galacturonans, and RG-II) and rhamnogalacturonan-I. Galacturonans have a backbone that Cell wall polysaccharides consists of -1,4-linked galacturonic acid. The identification of glycosyltransferases involved in pectin Galacturonan a Glycosyltransferases synthesis is essential to the study of cell wall function in plant growth and development and for maxi- Homogalacturonan mizing the value and use of plant polysaccharides in industry and human health. -

Plant:Animal Cell Comparison
Comparing Plant And Animal Cells http://khanacademy.org/video?v=Hmwvj9X4GNY Plant Cells shape - most plant cells are squarish or rectangular in shape. amyloplast (starch storage organelle)- an organelle in some plant cells that stores starch. Amyloplasts are found in starchy plants like tubers and fruits. cell membrane - the thin layer of protein and fat that surrounds the cell, but is inside the cell wall. The cell membrane is semipermeable, allowing some substances to pass into the cell and blocking others. cell wall - a thick, rigid membrane that surrounds a plant cell. This layer of cellulose fiber gives the cell most of its support and structure. The cell wall also bonds with other cell walls to form the structure of the plant. chloroplast - an elongated or disc-shaped organelle containing chlorophyll. Photosynthesis (in which energy from sunlight is converted into chemical energy - food) takes place in the chloroplasts. chlorophyll - chlorophyll is a molecule that can use light energy from sunlight to turn water and carbon dioxide gas into glucose and oxygen (i.e. photosynthesis). Chlorophyll is green. cytoplasm - the jellylike material outside the cell nucleus in which the organelles are located. Golgi body - (or the golgi apparatus or golgi complex) a flattened, layered, sac-like organelle that looks like a stack of pancakes and is located near the nucleus. The golgi body modifies, processes and packages proteins, lipids and carbohydrates into membrane-bound vesicles for "export" from the cell. lysosome - vesicles containing digestive enzymes. Where the digestion of cell nutrients takes place. mitochondrion - spherical to rod-shaped organelles with a double membrane. -

Chloroplasts Are the Food Producers of the Cell. the Organelles Are Only Found in Plant Cells and Some Protists Such As Algae
Name: ___________________________ Cell #2 H.W. due September 22nd, 2016 Period: _________ Chloroplasts are the food producers of the cell. The organelles are only found in plant cells and some protists such as algae. Animal cells do not have chloroplasts. Chloroplasts work to convert light energy of the Sun into sugars that can be used by cells. It is like a solar panel that changes sunlight energy into electric energy. The entire process is called photosynthesis and it all depends on the little green chlorophyll molecules in each chloroplast. In the process of photosynthesis, plants create sugars and release oxygen (O2). The oxygen released by the chloroplasts is the same oxygen you breathe every day. Chloroplasts are found in plant cells, but not in animal cells. The purpose of the chloroplast is to make sugars that feed the cell’s machinery. Photosynthesis is the process of a plant taking energy from the Sun and creating sugars. When the energy from the Sun hits a chloroplast and the chlorophyll molecules, light energy is converted into the chemical energy. Plants use water, carbon dioxide, and sunlight to make sugar and oxygen. During photosynthesis radiant energy or solar energy or light energy is transferred into chemical energy in the form of sugar (glucose). You already know that during photosynthesis plants make their own food. The food that the plant makes is in the form of sugar that is used to provide energy for the plant. The extra sugar that the plant does not use is stored as starch for later use. Mitochondria are known as the powerhouses of the cell. -

Plastid in Human Parasites
SCIENTIFIC CORRESPONDENCE being otherwise homo Plastid in human geneous. The 35-kb genome-containing organ parasites elle identified here did not escape the attention of early Sm - The discovery in malarial and toxo electron microscopists who plasmodial parasites of genes normally - not expecting the pres occurring in the photosynthetic organelle ence of a plastid in a proto of plants and algae has prompted specula zoan parasite like tion that these protozoans might harbour Toxoplasma - ascribed to it a vestigial plastid1• The plastid-like para various names, including site genes occur on an extrachromosomal, 'Hohlzylinder' (hollow cylin maternally inherited2, 35-kilobase DNA der), 'Golgi adjunct' and circle with an architecture reminiscent of 'grof3e Tilkuole mit kriiftiger that of plastid genomes3•4• Although the Wandung' (large vacuole 35-kb genome is distinct from the 6-7-kb with stout surrounds) ( see linear mitochondrial genome3-6, it is not refs cited in ref. 9). Our pre known where in the parasite cells the plas liminary experiments with tid-like genome resides. Plasmodium falciparum, the To determine whether a plastid is pre causative agent of the most sent, we used high-resolution in situ lethal form of malaria, hybridization7 to localize transcripts of a identify an organelle (not plastid-like 16S ribosomal RNA gene shown) which appears sim from Toxoplasma gondii8, the causative ilar to the T. gondii plastid. agent of toxoplasmosis. Transcripts accu The number of surrounding mulate in a small, ovoid organelle located membranes in the P. anterior to the nucleus in the mid-region falciparum plastid, and its of the cell (a, b in the figure). -

Lesson 1: Plant Cells
LESSON 1: PLANT CELLS LEVEL 1 What is a plant? A quick answer might be “something that is green and has leaves.” But are all plants green? There’s a type of maple tree that has purplish-red leaves. Obviously it is a plant because it is a tree. So being green can’t be a requirement for being a plant, though most plants are indeed green. What about leaves? Do all plants have leaves? Think about a cactus. Do those sharp needles count as leaves? Or what about the “stone plant”? It looks like a rock. (No kidding--it really does!) What makes a plant a plant? The answer is... a plant is a plant because it can make its own food using a process called photosynthesis. Plants can use the energy from sunlight to turn water and carbon dioxide into sugar. (“Photo” means “light,” and “synthesis” means “make.”) Wouldn’t it be nice if you could make your own food from sunlight? No more going to the grocery store or planting a garden. You could just stand in the sunshine, take a deep breath, drink a glass of water, and make your own food. Sounds funny, but that’s exactly what plants do. They take water from the ground, carbon dioxide from the air, and energy from light and turn them into food. Photosynthesis is a very complicated chemical process. The exact details of how a plant takes apart the molecules of water and carbon dioxide and turns them into sugar is so complicated that you need a college degree in chemistry to really understand it. -

Evolution of the Life Cycle in Land Plants
Journal of Systematics and Evolution 50 (3): 171–194 (2012) doi: 10.1111/j.1759-6831.2012.00188.x Review Evolution of the life cycle in land plants ∗ 1Yin-Long QIU 1Alexander B. TAYLOR 2Hilary A. McMANUS 1(Department of Ecology and Evolutionary Biology, University of Michigan, Ann Arbor, MI 48109, USA) 2(Department of Biological Sciences, Le Moyne College, Syracuse, NY 13214, USA) Abstract All sexually reproducing eukaryotes have a life cycle consisting of a haploid and a diploid phase, marked by meiosis and syngamy (fertilization). Each phase is adapted to certain environmental conditions. In land plants, the recently reconstructed phylogeny indicates that the life cycle has evolved from a condition with a dominant free-living haploid gametophyte to one with a dominant free-living diploid sporophyte. The latter condition allows plants to produce more genotypic diversity by harnessing the diversity-generating power of meiosis and fertilization, and is selectively favored as more solar energy is fixed and fed into the biosystem on earth and the environment becomes more heterogeneous entropically. Liverworts occupy an important position for understanding the origin of the diploid generation in the life cycle of land plants. Hornworts and lycophytes represent critical extant transitional groups in the change from the gametophyte to the sporophyte as the independent free-living generation. Seed plants, with the most elaborate sporophyte and the most reduced gametophyte (except the megagametophyte in many gymnosperms), have the best developed sexual reproduction system that can be matched only by mammals among eukaryotes: an ancient and stable sex determination mechanism (heterospory) that enhances outcrossing, a highly bimodal and skewed distribution of sperm and egg numbers, a male-driven mutation system, female specialization in mutation selection and nourishment of the offspring, and well developed internal fertilization. -

Phytoliths of Pteridophytes
South African Journal of Botany 77 (2011) 10–19 Minireview Phytoliths of pteridophytes J. Mazumdar UGC Centre for Advanced Study, Department of Botany, The University of Burdwan, Burdwan-713104, India Received 3 June 2010; received in revised form 14 July 2010; accepted 28 July 2010 Abstract Study of phytoliths of pteridophytes is an emerging area of research. Literature on this aspect is limited but increasing. Some recent findings have shown that phytoliths may have systematic and phylogenetic utility in pteridophytes. Phytoliths are functionally significant for the development and survival of pteridophytes. Experiments with some pteridophytes have revealed various aspects of silica uptake, deposition and biological effects. © 2010 SAAB. Published by Elsevier B.V. All rights reserved. Keywords: Biogenic silica; Ferns; Pteridophytes; Phytolith; Silicification 1. Introduction In spite of environmental effects on silica uptake, ongoing investigations indicate that phytolith formation is primarily Many plants deposit silica as solid hydrated Silicone dioxide under genetic control (Piperno, 2006). Phytoliths have been (SiO2,nH2O) in the cell lumen or in intercellular spaces, where used successfully as taxonomic tools in angiosperms, especially it is known as “phytoliths” or “plant stones” (Greek, phyto = in monocots (Piperno, 1988; Tubb et al., 1993). Less plant, lithos = stone) or “silicophytoliths” or “opal phytoliths” or information is available about pteridophytic phytoliths. The “plant opal” or “opaline silica” or “biogenic silica” or “bioliths”. objective of this review is to evaluate the present status and The term “phytolith” may also be applied to other mineral future prospects of phytolith research in pteridophytes. structures of plant origin, including calcium oxalate crystals, but is more usually restricted to silica particles (Prychid et al., 2004). -

Signals That Control Plant Vascular Cell Differentiation
REVIEWS SIGNALS THAT CONTROL PLANT VASCULAR CELL DIFFERENTIATION Hiroo Fukuda Plant vascular cells originate from procambial cells, which are vascular stem cells. Recent studies with Zinnia elegans cell culture and Arabidopsis thaliana mutants indicate that intercellular- signalling molecules such as auxin, cytokinin, brassinosteroids and xylogen regulate the maintenance or differentiation of procambial cells through distinct intracellular-signal transduction and gene-expression machineries. This intercellular- and intracellular-signalling system might be involved in determining the continuity and pattern formation of vascular tissues. PLANT CELL BIOLOGY VASCULAR CELLS The body plan of plants is controlled by a combination such as PROCAMBIUM and VASCULAR CAMBIUM.But MONO- Cells that form the plant of clonal fate and positional information that is pro- COTYLEDONOUS PLANTS and ferns, in which secondary vascular tissues, including vided by local signals, as is commonly seen in multicel- thickening growth does not occur, lack cambium. procambial cells, cambial cells, lular organisms. Recent advances in molecular genetics Individual species of vascular plants form distinct radial sieve elements, companion cells, fibre cells, xylem and phloem and genome biology have uncovered unique mecha- patterns of vascular bundles in each organ, which have parenchyma cells, and tracheary nisms of plant-body formation. Vascular cells are been categorized as collateral, bicollateral, amphivasal elements. formed under a well-defined plant-differentiation pro- or amphicribral patterns (FIG. 1a, b). gramme. Although vascular cells usually differentiate at Procambial cells are vascular stem cells that are APICAL MERISTEM predicted positions and at a predicted time to form a derived from the apical meristem and give rise to xylem The meristematic tissue that is located at the tip of the shoot specific vascular pattern, the arrangement of the vascular and phloem precursor cells. -
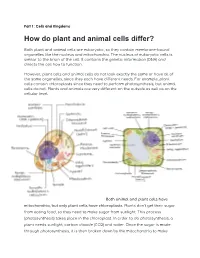
How Do Plant and Animal Cells Differ?
Part 1: Cells and Kingdoms How do plant and animal cells differ? Both plant and animal cells are eukaryotic, so they contain membrane-bound organelles like the nucleus and mitochondria. The nucleus of eukaryotic cells is similar to the brain of the cell. It contains the genetic information (DNA) and directs the cell how to function. However, plant cells and animal cells do not look exactly the same or have all of the same organelles, since they each have different needs. For example, plant cells contain chloroplasts since they need to perform photosynthesis, but animal cells do not. Plants and animals are very different on the outside as well as on the cellular level. Both animal and plant cells have mitochondria, but only plant cells have chloroplasts. Plants don’t get their sugar from eating food, so they need to make sugar from sunlight. This process (photosynthesis) takes place in the chloroplast. In order to do photosynthesis, a plant needs sunlight, carbon dioxide (CO2) and water. Once the sugar is made through photosynthesis, it is then broken down by the mitochondria to make energy for the cell. Because animals get sugar from the food they eat, they do not need chloroplasts: just mitochondria. Both plant and animal cells have vacuoles. A plant cell contains a large, singular vacuole that is used for storage of water and nutrients. It also helps maintain the shape of the cell. In contrast, animal cells have many, smaller vacuoles, which also are used for storage of water and nutrients. Plant cells have a cell wall, as well as a cell membrane. -

History of Plant Tissue Culture
HISTORY OF PLANT TISSUE CULTURE Programme: B.Sc (H) Botany Course Title: Plant Biotechnology Course code: BOTY 3014 Prof. Shahana Majumder Department of Botany Mahatma Gandhi Central University, Motihari Disclaimer • These materials are taken/modified/compiled from various sources like research articles and freely available internet websites, and are meant to be used solely for the teaching purpose in a public university, and solely for the use of UG students. • Plant tissue culture is the aseptic culture of cells, tissues, organs and their components under defined physical and chemical conditions in vitro. • The theoretical basis for plant tissue culture was proposed by Gottlieb Haberlandt in his address to the German Academy of Science in 1902 on his experiments on the culture of single cells •Historically, Henri-Louis Duhamel du Monceau (1756) pioneered the experiments on wound healing in plants through spontaneous callus (unorganised mass of cells) formation on decorticated region of elm plants. Elm tree • Vochting (1878) suggested the presence of polarity as a key feature that guide the development of plant fragments. • He observed that the upper portion of a piece of a stem always produced buds and the basal region produced callus or roots. • In 1902, a German Botanist Gottlieb Haberlandt developed the concept of culture of isolated cells of Tradescantia in artificial condition. Though his experiment failed to induce the cells to divide. Tradescantia sp • He did not succeed because by that time even auxin was not discovered. But he lent a foundation to plant physiology. • He described the cultivation of mesophyll cells of Lamium purpureum and Eichhornia crassipes, epidermal cells of Ornithogalum and hair cells of Pumonaria Eichhornia crassipes, Ornithogalum • Cell survived for 3-4 weeks. -

Cell Theory and Cell Organelles By: Christopher Meisler Science Methods
Cell Theory and Cell Organelles By: Christopher Meisler Science Methods This is a 6 day unit plan covering: First Observation of cells, Cell Theory and the organelles of both Plant and Animal Cells. Some ideas I have come up with for grading are: a long term 3D model of cell, multiple worksheets, a rap on Cell Theory, an egg- speriment, and a seeing is believing project. I went away form old school tes and used more project and worksheets. Day 1: First Observation of the Cell and Introduction of the 3D Cell project Here is where you would start the unit. The first day would be introducing all the basics and all material that is going to be covered. To help the students understand that cells are not easily seen I've included a project called: Is Seeing Believing? Here also is a good time to introduce the long term 3D project that the students would be making. For this make sure the students understand that they may not understand all of it right away but they will know enough to start. In a day or 2 the students will have all the information needed to do the project. Title: Is Seeing Believing? Grade level/ Subject: 6 - 10 Life Science Overview: Students will be cutting out a small section of a picture that is in black and white. Then from here the students will write down what thev see. Then usine" a hand held magnifying class the students will then write down in detail what they see. From hear the students will take turns at a microscope and look at there piece and once again.