Introduction to Alkanes Alkanes Do Not React with Most Reagents for Two
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Safety Data Sheet Acc
Page 1/8 Safety Data Sheet acc. to OSHA HCS Printing date 03/18/2019 Reviewed on 03/18/2019 1 Identification · Product identifier · Product Name: EVEN ALK (C16-C36) · Part Number: ENC-EVEN-1K · Application of the substance / the mixture Certified Reference Material · Details of the supplier of the safety data sheet · Manufacturer/Supplier: SPEX CertiPrep, LLC. 203 Norcross Ave, Metuchen, NJ 08840 USA · Information department: product safety department · Emergency telephone number: Emergency Phone Number (24 hours) CHEMTREC (800-424-9300) Outside US: 703-527-3887 2 Hazard(s) identification · Classification of the substance or mixture GHS02 Flame Flam. Liq. 2 H225 Highly flammable liquid and vapor. GHS08 Health hazard Carc. 2 H351 Suspected of causing cancer. GHS07 Acute Tox. 4 H302 Harmful if swallowed. · Label elements · GHS label elements The product is classified and labeled according to the Globally Harmonized System (GHS). · Hazard pictograms GHS02 GHS07 GHS08 · Signal word Danger · Hazard-determining components of labeling: dichloromethane · Hazard statements H225 Highly flammable liquid and vapor. H302 Harmful if swallowed. H351 Suspected of causing cancer. · Precautionary statements Keep away from heat/sparks/open flames/hot surfaces. - No smoking. Use explosion-proof electrical/ventilating/lighting/equipment. Wear protective gloves/protective clothing/eye protection/face protection. If on skin (or hair): Take off immediately all contaminated clothing. Rinse skin with water/shower. Store locked up. Dispose of contents/container in accordance with local/regional/national/international regulations. · Classification system: · NFPA ratings (scale 0 - 4) Health = 1 3 Fire = 3 1 0 Reactivity = 0 (Contd. on page 2) US 48.1.17.1 Page 2/8 Safety Data Sheet acc. -

Managing Health Risks of Solvent
BEST PRACTICE GUIDELINES Managing the Health Risks of Solvent Exposure Managing the Health Risks of Solvent Exposure September 2015 CONTENTS 1 Introduction 5 1.1 SCOPE AND OBJECTIVES 5 2 The regulatory landscape 6 2.1 CAD 6 2.2 REACH 6 2.3 CLP 8 3 Health effects and properties of solvents 9 3.1 EFFECTS OF SOLVENTS VIA SKIN AND EYE CONTACT 9 3.2 EFFECTS VIA INHALATION 10 3.3 CLASSIFICATION AND LABELLING OF SOLVENTS 10 3.4 HAZARD AND RISK 11 4 Exposure scenarios (es) - Basic principles 12 4.1 DEVELOPING AN ES 13 4.2 SOLVENTS MANUFACTURERS MEET THE CHALLENGE OF DEVELOPING ES 13 5 The solvents industry’s approach to REACH 14 5.1 NAMING CONVENTION FOR HYDROCARBON SOLVENTS 14 5.2 DEVELOPMENT OF GENERIC EXPOSURE SCENARIOS (GES) 14 5.2.1 THE USE DESCRIPTOR SYSTEM (UDS) (REF 15) 15 5.2.2 THE ESIG GES LIBRARY 15 6 Exposure limit values for solvents 17 6.1 OELs AND DNELs 17 6.2 INDUSTRY-BASED OELs FOR HYDROCARBON SOLVENTS 18 ManagingFlammability: the Health Risks A safety of Solvent guide Exposurefor users 7 Responsibilities of solvent users 19 8 Role of solvent vapour monitoring 23 9 Key messages 24 10 List of acronyms 25 11 References 28 APPENDIX 1 Overview of control approaches for solvents required by CAD 29 APPENDIX 2 CLP Classification phrases for solvents 30 APPENDIX 3 Hydrocarbon solvents registered under REACH - key data 31 APPENDIX 4 Oxygenated solvents registered under REACH - key data 43 APPENDIX 5 Example ES for a hydrocarbon solvent containing N-Hexane (>5-80%) 50 APPENDIX 6 List of common solvent uses matched to ESIG generic exposure scenario (GES) title with examples of relevant solvent types 53 APPENDIX 7 Mixtures 62 3 Managing the Health Risks of Solvent Exposure DISCLAIMER The information contained in this paper is intended for guidance only and whilst the information is provided in utmost good faith and has been based on the best information currently available, it is to be relied upon at the user’s own risk. -

Adsorption of Light Alkanes on the Surface of Substrates with Varying Symmetry and Composition
University of Tennessee, Knoxville TRACE: Tennessee Research and Creative Exchange Doctoral Dissertations Graduate School 5-2018 Adsorption of Light Alkanes on the Surface of Substrates with Varying Symmetry and Composition Nicholas Allan Strange University of Tennessee Follow this and additional works at: https://trace.tennessee.edu/utk_graddiss Recommended Citation Strange, Nicholas Allan, "Adsorption of Light Alkanes on the Surface of Substrates with Varying Symmetry and Composition. " PhD diss., University of Tennessee, 2018. https://trace.tennessee.edu/utk_graddiss/4967 This Dissertation is brought to you for free and open access by the Graduate School at TRACE: Tennessee Research and Creative Exchange. It has been accepted for inclusion in Doctoral Dissertations by an authorized administrator of TRACE: Tennessee Research and Creative Exchange. For more information, please contact [email protected]. To the Graduate Council: I am submitting herewith a dissertation written by Nicholas Allan Strange entitled "Adsorption of Light Alkanes on the Surface of Substrates with Varying Symmetry and Composition." I have examined the final electronic copy of this dissertation for form and content and recommend that it be accepted in partial fulfillment of the equirr ements for the degree of Doctor of Philosophy, with a major in Chemistry. John Z. Larese, Major Professor We have read this dissertation and recommend its acceptance: Takeshi Egami, Jeffrey D. Kovac, Brian K. Long Accepted for the Council: Dixie L. Thompson Vice Provost and Dean of the Graduate School (Original signatures are on file with official studentecor r ds.) Adsorption of Light Alkanes on the Surface of Substrates with Varying Symmetry and Composition A Dissertation Presented for the Doctor of Philosophy Degree The University of Tennessee, Knoxville Nicholas Allan Strange May 2018 Copyright © 2018 by Nicholas A. -
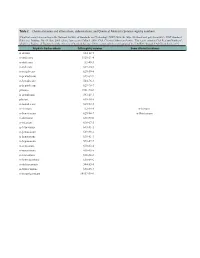
Table 2. Chemical Names and Alternatives, Abbreviations, and Chemical Abstracts Service Registry Numbers
Table 2. Chemical names and alternatives, abbreviations, and Chemical Abstracts Service registry numbers. [Final list compiled according to the National Institute of Standards and Technology (NIST) Web site (http://webbook.nist.gov/chemistry/); NIST Standard Reference Database No. 69, June 2005 release, last accessed May 9, 2008. CAS, Chemical Abstracts Service. This report contains CAS Registry Numbers®, which is a Registered Trademark of the American Chemical Society. CAS recommends the verification of the CASRNs through CAS Client ServicesSM] Aliphatic hydrocarbons CAS registry number Some alternative names n-decane 124-18-5 n-undecane 1120-21-4 n-dodecane 112-40-3 n-tridecane 629-50-5 n-tetradecane 629-59-4 n-pentadecane 629-62-9 n-hexadecane 544-76-3 n-heptadecane 629-78-7 pristane 1921-70-6 n-octadecane 593-45-3 phytane 638-36-8 n-nonadecane 629-92-5 n-eicosane 112-95-8 n-Icosane n-heneicosane 629-94-7 n-Henicosane n-docosane 629-97-0 n-tricosane 638-67-5 n-tetracosane 643-31-1 n-pentacosane 629-99-2 n-hexacosane 630-01-3 n-heptacosane 593-49-7 n-octacosane 630-02-4 n-nonacosane 630-03-5 n-triacontane 638-68-6 n-hentriacontane 630-04-6 n-dotriacontane 544-85-4 n-tritriacontane 630-05-7 n-tetratriacontane 14167-59-0 Table 2. Chemical names and alternatives, abbreviations, and Chemical Abstracts Service registry numbers.—Continued [Final list compiled according to the National Institute of Standards and Technology (NIST) Web site (http://webbook.nist.gov/chemistry/); NIST Standard Reference Database No. -

Section 2. Hazards Identification OSHA/HCS Status : This Material Is Considered Hazardous by the OSHA Hazard Communication Standard (29 CFR 1910.1200)
SAFETY DATA SHEET Flammable Liquid Mixture: Docosane / Dodecane / Eicosane / Hexadecane / Hexane / N-Decane / N-Heptane / N-Nonane / N-Octadecane / N-Octane / N-Tridecane / Tetracosane / Tetradecane / Undecane Section 1. Identification GHS product identifier : Flammable Liquid Mixture: Docosane / Dodecane / Eicosane / Hexadecane / Hexane / N-Decane / N-Heptane / N-Nonane / N-Octadecane / N-Octane / N-Tridecane / Tetracosane / Tetradecane / Undecane Other means of : Not available. identification Product use : Synthetic/Analytical chemistry. SDS # : 018777 Supplier's details : Airgas USA, LLC and its affiliates 259 North Radnor-Chester Road Suite 100 Radnor, PA 19087-5283 1-610-687-5253 24-hour telephone : 1-866-734-3438 Section 2. Hazards identification OSHA/HCS status : This material is considered hazardous by the OSHA Hazard Communication Standard (29 CFR 1910.1200). Classification of the : FLAMMABLE LIQUIDS - Category 1 substance or mixture SKIN CORROSION/IRRITATION - Category 2 SERIOUS EYE DAMAGE/ EYE IRRITATION - Category 2A TOXIC TO REPRODUCTION (Fertility) - Category 2 TOXIC TO REPRODUCTION (Unborn child) - Category 2 SPECIFIC TARGET ORGAN TOXICITY (SINGLE EXPOSURE) (Respiratory tract irritation) - Category 3 SPECIFIC TARGET ORGAN TOXICITY (SINGLE EXPOSURE) (Narcotic effects) - Category 3 SPECIFIC TARGET ORGAN TOXICITY (REPEATED EXPOSURE) - Category 2 AQUATIC HAZARD (ACUTE) - Category 2 AQUATIC HAZARD (LONG-TERM) - Category 1 GHS label elements Hazard pictograms : Signal word : Danger Hazard statements : Extremely flammable liquid and vapor. May form explosive mixtures in Air. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. Suspected of damaging fertility or the unborn child. May cause drowsiness and dizziness. May cause damage to organs through prolonged or repeated exposure. Very toxic to aquatic life with long lasting effects. -

Crude Oil and Fuels
Name Class Teacher Co-op Academy Leeds Science homework booklet. C14 – Crude oil and fuels Help and support: BBC bitesize for help with this booklet. Or type in the address below: https://www.bbc.co.uk/bitesize/guides/zshvw6f/revision/1 Free GCSE Science lessons: Videos 1 – 5 only: https://www.youtube.com/watch?v=CX2IYWggEBc&list=PL9IouNCPbCxVDcgWiviYYWj0xKMPX Td8s&index=1 Oakwood Academy: https://classroom.thenational.academy/units/organic-chemistry-7c58 Revision guides pages 146 - 148 Spec Summary of the specification content Learning outcomes ref. What most candidates should be able to do 4.7.1. ● Crude oil is a finite resource found in rocks. Crude oil is ● Be able to recognise substances as alkanes 1 the remains of an ancient biomass consisting mainly of given their formulae in these forms. plankton that was buried in mud. ● Students do not need to know the names of ● Crude oil is a mixture of a very large number of specific alkanes other than methane, ethane, compounds. Most of the compounds in crude oil are propane and butane. hydrocarbons, which are molecules made up of hydrogen and carbon atoms only. ● Most of the hydrocarbons in crude oil are hydrocarbons called alkanes. The general formula for the homologous series of alkanes is CnH2n+2 ● The first four members of the alkanes are methane, ethane, propane and butane. ● Alkane molecules can be represented in the following forms: ● C2H6 or 4.7.1. ● The many hydrocarbons in crude oil may be separated ● Explain how fractional distillation works in 2 into fractions, each of which contains molecules with a terms of evaporation and condensation. -

Download English-US Transcript (PDF)
MITOCW | MIT3_091SCF10lec27_iPOD The following content is provided under a creative commons license. Your support will help MIT OpenCourseWare continue to offer high-quality educational resources for free. To make a donation or view additional materials from hundreds of MIT courses, visit MIT OpenCourseWare at ocw.mit.edu. PROFESSOR: OK. Couple of announcements. First of all, the Wolf Lecture is tomorrow, right here in 10-250 at 4:00. As also, there will be the weekly quiz, the mini celebration. But there's going to be, a week from today, celebration part trois. Yes, third celebration of learning. And remember, what we're shooting for is the smiley face. None of this. And those of you who are over here on the grade point distribution curve, we want to move you to the right. So I'll say a little bit more later in the week about what the coverage will be. I'll be available for office hours later, and I thought to try to cheer you up, and remind you that studying is not something you do only the night before the exam. You have to be studying on a regular basis. All right? So from the iconic film Ghostbusters, there's this one scene. [FILM PLAYBACK] For a moment, pretend that I don't know anything about metallurgy, engineering, or physics, and just tell me what the hell is going on. You never studied. [END FILM PLAYBACK] You don't want to be a character in that scene. So what are you going to do about it? You're going to study. -

HSPA Substance CAS No. Relationship Compilation
Hydrocarbon Solvents REACH REGISTRATIONS CAS - EC number relationship This documents is for refence only. It has no legal nor regulator value and does not claim to be complete. version October2018 Reach HSPA Category HSPA Substance Name Registration Related CAS No.(s) Related CAS No.(s) Substance Name provisional EC No. Solvent naphtha (petroleum), light C9 Aromatics Hydrocarbons, C9, aromatics 918-668-5 64742-95-6 aromatic Hydrocarbons, C10, Solvent naphtha (petroleum), heavy C10-12 Aromatics 919-284-0 64742-94-5 aromatics, >1% naphthalene aromatic Hydrocarbons, C10, Solvent naphtha (petroleum), heavy C10-12 Aromatics 918-811-1 64742-94-5 aromatics, <1% naphthalene aromatic Hydrocarbons, C10-C13, Solvent naphtha (petroleum), heavy C10-12 Aromatics 922-153-0 64742-94-5 aromatics, <1% naphthalene aromatic Hydrocarbons, C10-C13, Solvent naphtha (petroleum), heavy C10-12 Aromatics 926-273-4 64742-94-5 aromatics, >1% naphthalene aromatic Hydrocarbons, C9-C10, n- C9-14 Aliphatics (2-25% Naphtha (petroleum), alkanes, isoalkanes, cyclics, 927-344-2 64742-82-1 aromatics) hydrodesulfurized heavy aromatics (2- 25%) Hydrocarbons, C10-C13, n- C9-14 Aliphatics (2-25% Naphtha (petroleum), alkanes, isoalkanes, cyclics, 919-164-8 64742-82-1 aromatics) hydrodesulfurized heavy aromatics (2- 25%) Naphtha (petroleum), Hydrocarbons, C8-12, n- C9-14 Aliphatics (2-25% hydrodesulfurized heavy; Low alkanes, isoalkanes, cyclics, 928-136-4 64742-82-1 aromatics) boiling point hydrogen treated aromatics (2- 25%) naphtha Naphtha (petroleum), Hydrocarbons, C9-C12, -

CAS # IUPAC Name/Chemical Name
CAS # IUPAC Name/Chemical Name/Essenal Oil CAS, Common Name 57-10-3 Hexadecanoic acid Palmi&c acid, natural 57-55-6 Propane-1,2-diol Propylene glycol 59-02-9 (2R)-2,5,7,8-Tetramethyl-2-[(4R,8R)-4,8,12-trimethyltridecyl]-3,4-dihydrochromen-6-ol α-Tocopherol 60-12-8 2-Phenylethanol Phenylethyl alcohol 64-17-5 Ethanol Ethyl alcohol 64-18-6 Methanoic acid Formic acid 64-19-7 Ethanoic acid Ace&c acid 65-85-0 Benzoic acid Benzoic acid 66-25-1 Hexanal C-6 Aldehyde 67-63-0 Propan-2-ol 2-Propanol 67-64-1 Propan-2-one 2-Propanone 75-07-0 Acetaldehyde Acetaldehyde 75-18-3 Methylsulfanylmethane Dimethyl sulfide 75-65-0 2-Methyl-propan-2-ol 2-Methyl-2-propanol 76-22-2 1,7,7-Trimethylbicyclo[2.2.1]heptan-2-one Camphor 77-53-2 (1S,2R,5S,7R,8R)-2,6,6,8-Tetramethyltricyclo[5.3.1.0(1,5)]undecan-8-ol Cedrol 77-54-3 (1S,2R,5S,8R)-2,6,6,8-Tetramethyltricyclo[5.3.1.0(1,5)]undecan-8-yl acetate Cedarwood oil acetylated 77-83-8 Ethyl 3-methyl-3-phenyloxirane-2-carboxylate Ethyl methylphenylglycidate 77-90-7 1,2,3-Tributyl 2-(acetyloxy)propane-1,2,3-tricarboxylate Tributyl o-acetylcitrate 77-92-9 3-Carboxy-3-hydroxypentanedioic acid Citric acid; 2-Hydroxypropane-1,2,3-tricarboxylic acid 77-93-0 1,2,3-Triethyl 2-hydroxypropane-1,2,3-tricarboxylate Triethyl citrate 78-35-3 3,7-Dimethylocta-1,6-dien-3-yl 2-methylpropanoate Linalyl isobutyrate 78-36-4 3,7-Dimethylocta-1,6-dien-3-yl butanoate 1-Ethenyl-1,5-dimethyl-4-hexen-1-yl butanoate 78-37-5 3,7-Dimethylocta-1,6-dien-3-yl (E)-3-phenylprop-2-enoate Linalyl cinnamate 78-69-3 3,7-Dimethyloctan-3-ol Tetrahydrolinalool -

Extended Hard-Sphere Model for Predicting the Viscosity of Long
Extended Hard-Sphere Model for Predicting the Viscosity of long- chain n-alkanes Nicolas Riesco,1,2 and Velisa Vesovic*,1 1. Department of Earth Science and Engineering, Imperial College London, South Kensington Campus, London SW7 2AZ, United Kingdom. 2 Qatar Carbonates and Carbon Storage Research Centre (QCCSRC), Imperial College London, London SW7 2AZ, UK * To whom correspondence should be addressed. E-mail: [email protected] 1 Abstract An extended hard-sphere model is presented that can accurately and reliably predict the viscosity of long chain n-alkanes. The method is based on the hard-sphere model of Dymond and Assael, that makes use of an universal function relating reduced viscosity to reduced volume. The existing expression for the molar core volume is extrapolated to long chain n-alkanes, while the roughness factor is determined from experimental data. A new correlation for roughness factor is developed that allows the extended model to reproduce the available experimental viscosity data on long chain n-alkanes up to tetracontane (n- C40H82) within ±5 %, at pressure up to 30 MPa. In the dilute gas limit a physically realistic model, based on Lennard-Jones effective potential, is proposed and used to evaluate the zero-density viscosity of n-alkanes to within ±2.4 %, that is better than currently available. Keywords Alkanes; Correlation; Hard-Sphere Theory; High Pressure; Liquid; Vapour; Viscosity. 2 1. Introduction Normal, paraffinic alkanes (n-alkanes) of generic chemical formula CnH2n+2 are an important constituent of oil and are ubiquitous in petroleum and chemical processes. In numerous industrial applications that involve the flow of fluids, knowledge of the viscosity of n-alkanes and their mixtures is an essential pre-requisite for good design and optimal operations [1-2]. -

Short Summary of IUPAC Nomenclature of Organic Compounds
Short Summary of IUPAC Nomenclature of Organic Compounds Introduction The purpose of the IUPAC system of nomenclature is to establish an international standard of naming compounds to facilitate communication. The goal of the system is to give each structure a unique and unambiguous name, and to correlate each name with a unique and unambiguous structure. I. Fundamental Principle IUPAC nomenclature is based on naming a molecule’s longest chain of carbons connected by single bonds, whether in a continuous chain or in a ring. All deviations, either multiple bonds or atoms other than carbon and hydrogen, are indicated by prefixes or suffixes according to a specific set of priorities. II. Alkanes and Cycloalkanes Alkanes are the family of saturated hydrocarbons, that is, molecules containing carbon and hydrogen connected by single bonds only. These molecules can be in continuous chains (called linear or acyclic), or in rings (called cyclic or alicyclic). The names of alkanes and cycloalkanes are the root names of organic compounds. Beginning with the five-carbon alkane, the number of carbons in the chain is indicated by the Greek or Latin prefix. Rings are designated by the prefix “cyclo”. (In the geometrical symbols for rings, each apex represents a carbon with the number of hydrogens required to fill its valence.) CH4 methane CH3[CH2]10CH3 dodecane CH3CH3 ethane CH3[CH2]11CH3 tridecane CH3CH2CH3 propane CH3[CH2]12CH3 tetradecane CH3[CH2]2CH3 butane CH3[CH2]18CH3 icosane CH3[CH2]3CH3 pentane CH3[CH2]19CH3 henicosane CH3[CH2]4CH3 hexane CH3[CH2]20CH3 docosane CH3[CH2]5CH3 heptane CH3[CH2]21CH3 tricosane CH3[CH2]6CH3 octane CH3[CH2]28CH3 triacontane CH3[CH2]7CH3 nonane CH3[CH2]29CH3 hentriacontane CH3[CH2]8CH3 decane CH3[CH2]38CH3 tetracontane CH3[CH2]9CH3 undecane CH3[CH2]48CH3 pentacontane H H C H H C C cyclopropane H H cyclobutane cyclopentane cyclohexane cycloheptane cyclooctane Short Summary of IUPAC Nomenclature, p. -

Correlation of Surface and Interfacial Tension of Light Hydrocarbons in the Critical Region
Correlation of Surface and Interfacial Tension of Light Hydrocarbons in the Critical Region E.W.HOUGH* THE U. OF TEXAS MEMBER AIME AUSTIN. TEX. G. L. STEGEMEIER SHELL DEVELOPMENT CO. JUNIOR MEMBER AIME HOUSTON. TEX. Downloaded from http://onepetro.org/spejournal/article-pdf/1/04/259/2152696/spe-197-pa.pdf by guest on 28 September 2021 ABSTRACT Interfacial tension is defined as the specific Empirical equations for surface tension of propane surface-free energy between two phases of unlike and normal butane as functions of reduced temper fractional composition, while surface tension is ature are obtained from experimental data. Another defined as the specific surface-free energy between correlation relating surface tension to enthalpy of two phases of the same fractional composition. The vaporization is given for these two compounds. In usual definitions relating interfacial tension to a addition, new parachor numbers are calculated for liquid-liquid interface and surface tension to a the normal paraffin hydrocarbons. These numbers gas-liquid interface are not clearly defined when are utilized for the calculation of interfacial tension the critical regi~n is included, and there is no sharp of two-component systems as functions of pressure distinction between a gas and a liquid phase. and Jemperature, using a modified form of Weinaug Interfacial tension is probably the most important Katz equation. The experimental data for two binary single force that makes one-half to one-third of the systems are approximated by the correlation. From total oil actually in place in a reservoir rock un these results it is found that the interfacial tension recoverable by conventional gas-drive or waterflood in the high-pressure region remains extremely low methods.