Clinician's Guide to Prescribing Depot Antipsychotics
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Original Research Published, Reproduced, Transmitted, Modified, Posted, Sold, Licensed, Or Used for Commercial Purposes
This work may not be copied, distributed, displayed, Original Research published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the Efficacy and Effectiveness of Depot publisher’s Terms & Conditions. Versus Oral Antipsychotics in Schizophrenia: Synthesizing Results Across Different Research Designs Noam Y. Kirson, PhD; Peter J. Weiden, MD; Sander Yermakov, MS; Wayne Huang, MPP; Thomas Samuelson, BA; Steve J. Offord, PhD; Paul E. Greenberg, MS, MA; and Bruce J. O. Wong, MD ABSTRACT he cornerstone of long-term maintenance therapy Objective: Nonadherence is a major challenge in schizophrenia Tof schizophrenia patients is relapse prevention. treatment. While long-acting (depot) antipsychotic medications are Relapse prevention is necessary—albeit not sufficient— often recommended to address adherence problems, evidence on for eventual successful rehabilitation.1 In practice, the the comparative effectiveness of depot versus oral antipsychotics is effectiveness of maintenance antipsychotic treatment is inconsistent. We hypothesize that this inconsistency could be due to often undermined by poor adherence to therapy. Not systematic differences in study design. This review evaluates the effect only is nonadherence the single greatest modifiable of study design on the comparative effectiveness of antipsychotic risk factor for relapse,2,3 it is also often undetected, formulations. The optimal use of different antipsychotic formulations resulting in lost opportunities to employ psychosocial in a general clinical setting depends on better understanding of the underlying reasons for differences in effectiveness across research interventions for adherence, as well as uncertainty as designs. to the relative contribution of lack of efficacy versus Data Sources: A PubMed literature review targeted English-language adherence problems to poor outcomes. -

Treatment of Psychosis: 30 Years of Progress
Journal of Clinical Pharmacy and Therapeutics (2006) 31, 523–534 REVIEW ARTICLE Treatment of psychosis: 30 years of progress I. R. De Oliveira* MD PhD andM.F.Juruena MD *Department of Neuropsychiatry, School of Medicine, Federal University of Bahia, Salvador, BA, Brazil and Department of Psychological Medicine, Institute of Psychiatry, King’s College, University of London, London, UK phrenia. Thirty years ago, psychiatrists had few SUMMARY neuroleptics available to them. These were all Background: Thirty years ago, psychiatrists had compounds, today known as conventional anti- only a few choices of old neuroleptics available to psychotics, and all were liable to cause severe extra them, currently defined as conventional or typical pyramidal side-effects (EPS). Nowadays, new antipsychotics, as a result schizophrenics had to treatments are more ambitious, aiming not only to suffer the severe extra pyramidal side effects. improve psychotic symptoms, but also quality of Nowadays, new treatments are more ambitious, life and social reinsertion. We briefly but critically aiming not only to improve psychotic symptoms, outline the advances in diagnosis and treatment but also quality of life and social reinsertion. Our of schizophrenia, from the mid 1970s up to the objective is to briefly but critically review the present. advances in the treatment of schizophrenia with antipsychotics in the past 30 years. We conclude DIAGNOSIS OF SCHIZOPHRENIA that conventional antipsychotics still have a place when just the cost of treatment, a key factor in Up until the early 1970s, schizophrenia diagnoses poor regions, is considered. The atypical anti- remained debatable. The lack of uniform diagnostic psychotic drugs are a class of agents that have criteria led to relative rates of schizophrenia being become the most widely used to treat a variety of very different, for example, in New York and psychoses because of their superiority with London, as demonstrated in an important study regard to extra pyramidal symptoms. -
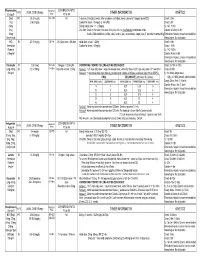
Medication Conversion Chart
Fluphenazine FREQUENCY CONVERSION RATIO ROUTE USUAL DOSE (Range) (Range) OTHER INFORMATION KINETICS Prolixin® PO to IM Oral PO 2.5-20 mg/dy QD - QID NA ↑ dose by 2.5mg/dy Q week. After symptoms controlled, slowly ↓ dose to 1-5mg/dy (dosed QD) Onset: ≤ 1hr 1mg (2-60 mg/dy) Caution for doses > 20mg/dy (↑ risk EPS) Cmax: 0.5hr 2.5mg Elderly: Initial dose = 1 - 2.5mg/dy t½: 14.7-15.3hr 5mg Oral Soln: Dilute in 2oz water, tomato or fruit juice, milk, or uncaffeinated carbonated drinks Duration of Action: 6-8hr 10mg Avoid caffeinated drinks (coffee, cola), tannics (tea), or pectinates (apple juice) 2° possible incompatibilityElimination: Hepatic to inactive metabolites 5mg/ml soln Hemodialysis: Not dialyzable HCl IM 2.5-10 mg/dy Q6-8 hr 1/3-1/2 po dose = IM dose Initial dose (usual): 1.25mg Onset: ≤ 1hr Immediate Caution for doses > 10mg/dy Cmax: 1.5-2hr Release t½: 14.7-15.3hr 2.5mg/ml Duration Action: 6-8hr Elimination: Hepatic to inactive metabolites Hemodialysis: Not dialyzable Decanoate IM 12.5-50mg Q2-3 wks 10mg po = 12.5mg IM CONVERTING FROM PO TO LONG-ACTING DECANOATE: Onset: 24-72hr (4-72hr) Long-Acting SC (12.5-100mg) (1-4 wks) Round to nearest 12.5mg Method 1: 1.25 X po daily dose = equiv decanoate dose; admin Q2-3wks. Cont ½ po daily dose X 1st few mths Cmax: 48-96hr 25mg/ml Method 2: ↑ decanoate dose over 4wks & ↓ po dose over 4-8wks as follows (accelerate taper for sx of EPS): t½: 6.8-9.6dy (single dose) ORAL DECANOATE (Administer Q 2 weeks) 15dy (14-100dy chronic administration) ORAL DOSE (mg/dy) ↓ DOSE OVER (wks) INITIAL DOSE (mg) TARGET DOSE (mg) DOSE OVER (wks) Steady State: 2mth (1.5-3mth) 5 4 6.25 6.25 0 Duration Action: 2wk (1-6wk) Elimination: Hepatic to inactive metabolites 10 4 6.25 12.5 4 Hemodialysis: Not dialyzable 20 8 6.25 12.5 4 30 8 6.25 25 4 40 8 6.25 25 4 Method 3: Admin equivalent decanoate dose Q2-3wks. -

Schizophrenia Care Guide
August 2015 CCHCS/DHCS Care Guide: Schizophrenia SUMMARY DECISION SUPPORT PATIENT EDUCATION/SELF MANAGEMENT GOALS ALERTS Minimize frequency and severity of psychotic episodes Suicidal ideation or gestures Encourage medication adherence Abnormal movements Manage medication side effects Delusions Monitor as clinically appropriate Neuroleptic Malignant Syndrome Danger to self or others DIAGNOSTIC CRITERIA/EVALUATION (PER DSM V) 1. Rule out delirium or other medical illnesses mimicking schizophrenia (see page 5), medications or drugs of abuse causing psychosis (see page 6), other mental illness causes of psychosis, e.g., Bipolar Mania or Depression, Major Depression, PTSD, borderline personality disorder (see page 4). Ideas in patients (even odd ideas) that we disagree with can be learned and are therefore not necessarily signs of schizophrenia. Schizophrenia is a world-wide phenomenon that can occur in cultures with widely differing ideas. 2. Diagnosis is made based on the following: (Criteria A and B must be met) A. Two of the following symptoms/signs must be present over much of at least one month (unless treated), with a significant impact on social or occupational functioning, over at least a 6-month period of time: Delusions, Hallucinations, Disorganized Speech, Negative symptoms (social withdrawal, poverty of thought, etc.), severely disorganized or catatonic behavior. B. At least one of the symptoms/signs should be Delusions, Hallucinations, or Disorganized Speech. TREATMENT OPTIONS MEDICATIONS Informed consent for psychotropic -

Management of Side Effects of Antipsychotics
Management of side effects of antipsychotics Oliver Freudenreich, MD, FACLP Co-Director, MGH Schizophrenia Program www.mghcme.org Disclosures I have the following relevant financial relationship with a commercial interest to disclose (recipient SELF; content SCHIZOPHRENIA): • Alkermes – Consultant honoraria (Advisory Board) • Avanir – Research grant (to institution) • Janssen – Research grant (to institution), consultant honoraria (Advisory Board) • Neurocrine – Consultant honoraria (Advisory Board) • Novartis – Consultant honoraria • Otsuka – Research grant (to institution) • Roche – Consultant honoraria • Saladax – Research grant (to institution) • Elsevier – Honoraria (medical editing) • Global Medical Education – Honoraria (CME speaker and content developer) • Medscape – Honoraria (CME speaker) • Wolters-Kluwer – Royalties (content developer) • UpToDate – Royalties, honoraria (content developer and editor) • American Psychiatric Association – Consultant honoraria (SMI Adviser) www.mghcme.org Outline • Antipsychotic side effect summary • Critical side effect management – NMS – Cardiac side effects – Gastrointestinal side effects – Clozapine black box warnings • Routine side effect management – Metabolic side effects – Motor side effects – Prolactin elevation • The man-in-the-arena algorithm www.mghcme.org Receptor profile and side effects • Alpha-1 – Hypotension: slow titration • Dopamine-2 – Dystonia: prophylactic anticholinergic – Akathisia, parkinsonism, tardive dyskinesia – Hyperprolactinemia • Histamine-1 – Sedation – Weight gain -

Sex-Specific Cannabidiol- and Iloperidone-Induced Neuronal Activity Changes in an in Vitro MAM Model System of Schizophrenia
International Journal of Molecular Sciences Article Sex-Specific Cannabidiol- and Iloperidone-Induced Neuronal Activity Changes in an In Vitro MAM Model System of Schizophrenia Rachel-Karson Thériault 1,2,†, Myles St-Denis 1,†, Tristen Hewitt 1,2, Jibran Y. Khokhar 2,3 , Jasmin Lalonde 1,2 and Melissa L. Perreault 2,3,* 1 Department of Molecular and Cellular Biology, University of Guelph, Guelph, ON N1G 2W1, Canada; [email protected] (R.-K.T.); [email protected] (M.S.-D.); [email protected] (T.H.); [email protected] (J.L.) 2 Collaborative Program in Neuroscience, University of Guelph, Guelph, ON N1G 2W1, Canada; [email protected] 3 Department of Biomedical Sciences, University of Guelph, Guelph, ON N1G 2W1, Canada * Correspondence: [email protected]; Tel.: +1-(519)-824-4120 (ext. 52013) † These authors contributed equally to this work. Abstract: Cortical circuit dysfunction is thought to be an underlying mechanism of schizophrenia (SZ) pathophysiology with normalization of aberrant circuit activity proposed as a biomarker for antipsychotic efficacy. Cannabidiol (CBD) shows potential as an adjunctive antipsychotic therapy; however, potential sex effects in these drug interactions remain unknown. In the present study, we sought to elucidate sex effects of CBD coadministration with the atypical antipsychotic iloperidone (ILO) on the activity of primary cortical neuron cultures derived from the rat methylazoxymethanol Citation: Thériault, R.-K.; St-Denis, acetate (MAM) model used for the study of SZ. Spontaneous network activity measurements were M.; Hewitt, T.; Khokhar, J.Y.; Lalonde, J.; Perreault, M.L. Sex-Specific obtained using a multielectrode array at baseline and following administration of CBD or ILO Cannabidiol- and Iloperidone- alone, or combined. -

Revision of Precautions Asenapine Maleate, Aripiprazole, Olanzapine
Published by Translated by Ministry of Health, Labour and Welfare Pharmaceuticals and Medical Devices Agency This English version is intended to be a reference material to provide convenience for users. In the event of inconsistency between the Japanese original and this English translation, the former shall prevail. Revision of Precautions Asenapine maleate, aripiprazole, olanzapine, quetiapine fumarate, clocapramine hydrochloride hydrate, chlorpromazine hydrochloride, chlorpromazine hydrochloride/promethazine hydrochloride/phenobarbital, chlorpromazine phenolphthalinate, spiperone, zotepine, timiperone, haloperidol, paliperidone, pipamperone hydrochloride, fluphenazine decanoate, fluphenazine maleate, brexpiprazole, prochlorperazine maleate, prochlorperazine mesilate, Pharmaceuticals and Medical Devices Agency Office of Safety I 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013 Japan E-mail: [email protected] Published by Translated by Ministry of Health, Labour and Welfare Pharmaceuticals and Medical Devices Agency This English version is intended to be a reference material to provide convenience for users. In the event of inconsistency between the Japanese original and this English translation, the former shall prevail. propericiazine, bromperidol, perphenazine, perphenazine hydrochloride, perphenazine fendizoate, perphenazine maleate, perospirone hydrochloride hydrate, mosapramine hydrochloride, risperidone (oral drug), levomepromazine hydrochloride, levomepromazine maleate March 27, 2018 Non-proprietary name Asenapine maleate, -

Washington State Health Care Authority
Washington State Health Care Authority Prescription Drug Program 626 8th Ave SE, Olympia, WA 98501 ● 206-521-2029 https://www.hca.wa.gov/about-hca/prescription-drug-program February 17, 2021 Dear Interested Party, Based on recommendations by the Washington State Pharmacy and Therapeutics Committee, the Health Care Authority, Uniform Medical Plan (UMP), and the Department of Labor & Industries (L&I) have named the following drugs as preferred in their respective therapeutic classes on the Washington State Preferred Drug List (PDL), effective immediately: Antiplatelet reviewed 8/15/2018 Agency Coverage Ingredient Name Label Name of Preferred Product L&I UMP clopidogrel bisulfate clopidogrel tablet Not participating Yes The effect of this recommendation is no change to the Washington PDL. Second Generation Antipsychotics reviewed 12/18/2019 Agency Coverage Ingredient Name Label Name of Preferred Products L&I UMP aripiprazole Abilify Maintena® suspension reconstituted No Yes aripiprazole solution Yes Yes aripiprazole tablet Yes Yes aripiprazole ODT tablet dispersible Yes Yes aripiprazole lauroxil Aristada® injectable No Yes Aristada Initio® injectable No Yes asenapine maleate Saphris® sublingual Yes Yes brexpiprazole Rexulti® tablet Yes Yes cariprazine HCL Vraylar® capsule Yes Yes clozapine clozapine tablet Yes Yes clozapine ODT tablet dispersible Yes Yes iloperidone Fanapt® tablet Yes Yes Fanapt Titration Pack® tablet Yes Yes lurasidone HCL Latuda® tablet Yes Yes olanzapine olanzapine injectable No Yes olanzapine tablet Yes Yes olanzapine -

Is Aristada (Aripiprazole Lauroxil) a Safe and Effective Treatment for Schizophrenia in Adult Patients? Kyle J
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Student Dissertations, Theses and Papers Scholarship 2017 Is Aristada (Aripiprazole Lauroxil) a Safe and Effective Treatment For Schizophrenia In Adult Patients? Kyle J. Knowles Philadelphia College of Osteopathic Medicine Follow this and additional works at: https://digitalcommons.pcom.edu/pa_systematic_reviews Part of the Psychiatry Commons Recommended Citation Knowles, Kyle J., "Is Aristada (Aripiprazole Lauroxil) a Safe and Effective Treatment For Schizophrenia In Adult Patients?" (2017). PCOM Physician Assistant Studies Student Scholarship. 381. https://digitalcommons.pcom.edu/pa_systematic_reviews/381 This Selective Evidence-Based Medicine Review is brought to you for free and open access by the Student Dissertations, Theses and Papers at DigitalCommons@PCOM. It has been accepted for inclusion in PCOM Physician Assistant Studies Student Scholarship by an authorized administrator of DigitalCommons@PCOM. For more information, please contact [email protected]. Is Aristada (Aripiprazole Lauroxil) a Safe and Effective Treatment For Schizophrenia In Adult Patients? Kyle J. Knowles, PA-S A SELECTIVE EVIDENCE BASED MEDICINE REVIEW In Partial Fulfillment of the Requirements For The Degree of Master of Science In Health Sciences- Physician Assistant Department of Physician Assistant Studies Philadelphia College of Osteopathic Medicine Philadelphia, Pennsylvania December 16, 2016 ABSTRACT OBJECTIVE: The objective of this selective EBM review is to determine whether or not “Is Aristada (aripiprazole lauroxil) a safe and effective treatment for schizophrenia in adult patients?” STUDY DESIGN: Review of three randomized controlled studies. All three trials were conducted between 2014 and 2015. DATA SOURCES: One randomized, controlled trial and two randomized, controlled, double- blind trials found via Cochrane Library and PubMed. -

Paliperidone Long Acting Injection Patient Information
Paliperidone (pala-pera-done) long acting injection Patient Information - Hillmorton Hospital Pharmacy www.healthinfo.org.nz Why have I been prescribed paliperidone? Paliperidone is used to treat schizophrenia, psychosis and similar conditions. It can also be used to treat mania. A person diagnosed with schizophrenia may hear voices talking to them or about them. They may also become suspicious or paranoid. Some people also have problems with their thinking and feel that other people can read their thoughts. These are called “positive Paliperidone Injection Long Acting symptoms”. Paliperidone can help to relieve these symptoms. Many people with schizophrenia also experience “negative symptoms”. They feel tired and lacking in energy and may become quite inactive and withdrawn. Paliperidone may help relieve these symptoms as well. Paliperidone may also be prescribed for people who have had side effects such as strange movements and shaking, with older types of antipsychotics. Paliperidone is a close relative of the antipsychotic risperidone. It’s side effect profile is similar but has the advantage of being able to be given as a once a month injection. What exactly is paliperidone? Is paliperidone safe for me? It is usually safe to take paliperidone Paliperidone is one of a group of regularly as prescribed by your doctor, but medicines used to treat schizophrenia it doesn’t suit everyone. Let your doctor and similar disorders. These illnesses are know if any of the following apply to you, sometimes referred to as psychoses, as extra care may be needed: hence the name given to this group of medicines, which is the “antipsychotics”. -

A Brief Overview of Psychiatric Pharmacotherapy
A Brief Overview of Psychiatric Pharmacotherapy Joel V. Oberstar, M.D. Chief Executive Officer Disclosures • Some medications discussed are not approved by the FDA for use in the population discussed/described. • Some medications discussed are not approved by the FDA for use in the manner discussed/described. • Co-Owner: – PrairieCare and PrairieCare Medical Group – Catch LLC Disclaimer The contents of this handout are for informational purposes only and are not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified healthcare provider with any questions you may have regarding a medical or psychiatric condition. Never disregard professional/medical advice or delay in seeking it because of something you have read in this handout. Material in this handout may be copyrighted by the author or by third parties; reasonable efforts have been made to give attribution where appropriate. Caveat Regarding the Role of Medication… Neuroscience Overview Mind Over Matter, National Institute on Drug Abuse, National Institutes of Health. Available at: http://teens.drugabuse.gov/mom/index.asp. http://medicineworld.org/images/news-blogs/brain-700997.jpg Neuroscience Overview Mind Over Matter, National Institute on Drug Abuse, National Institutes of Health. Available at: http://teens.drugabuse.gov/mom/index.asp. Neurotransmitter Receptor Source: National Institute on Drug Abuse Common Diagnoses and Associated Medications • Psychotic Disorders – Antipsychotics • Bipolar Disorders – Mood Stabilizers, Antipsychotics, & Antidepressants • Depressive Disorders – Antidepressants • Anxiety Disorders – Antidepressants & Anxiolytics • Attention Deficit Hyperactivity Disorder – Stimulants, Antidepressants, 2-Adrenergic Agents, & Strattera Classes of Medications • Anti-depressants • Stimulants and non-stimulant alternatives • Anti-psychotics (a.k.a. -

Psychotropic Medication Concerns When Treating Individuals with Developmental Disabilities
Tuesday, 2:30 – 4:00, C2 Psychotropic Medication Concerns when Treating Individuals with Developmental Disabilities Richard Berchou 248-613-6716 [email protected] Objective: Identify effective methods for the practical application of concepts related to improving the delivery of services for persons with developmental disabilities Notes: Medication Assistance On-Line Resources OBTAINING MEDICATION: • Needy Meds o Needymeds.com • Partnership for Prescriptions Assistance o Pparx.org • Patient Assistance Program Center o Rxassist.org • Insurance coverage & Prior authorization forms for most drug plans o Covermymeds.com REMINDERS TO TAKE MEDICATION: • Medication reminder by Email, Phone call, or Text message o Sugaredspoon.com ANSWER MOST QUESTIONS ABOUT MEDICATIONS: • Univ. of Michigan/West Virginia Schools of Pharmacy o Justaskblue.com • Interactions between medications, over-the-counter (OTC) products and some foods; also has a pictorial Pill Identifier: May input an entire list of medications o Drugs.com OTHER TRUSTED SITES: • Patient friendly information about disease and diagnoses o Mayoclinic.com, familydoctor.org • Package inserts, boxed warnings, “Dear Doctor” letters (can sign up to receive e- mail alerts) o Dailymed.nlm.nih.gov • Communications about drug safety o www.Fda.gov/cder/drug/drugsafety/drugindex.htm • Purchasing medications on-line o Pharnacychecker.com Updated 2013 Psychotropic Medication for Persons with Developmental Disabilities April 23, 2013 Richard Berchou, Pharm. D. Assoc. Clinical Prof., Dept. Psychiatry & Behavioral