Antibiotics Against Multidrug-Resistant Gram-Negative Bacteria
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

1: Clinical Pharmacokinetics 1
1: CLINICAL PHARMACOKINETICS 1 General overview: clinical pharmacokinetics, 2 Pharmacokinetics, 4 Drug clearance (CL), 6 Volume of distribution (Vd), 8 The half-life (t½), 10 Oral availability (F), 12 Protein binding (PB), 14 pH and pharmacokinetics, 16 1 Clinical pharmacokinetics General overview General overview: clinical pharmacokinetics 1 The ultimate aim of drug therapy is to achieve effi cacy without toxicity. This involves achieving a plasma concentration (Cp) within the ‘therapeutic window’, i.e. above the min- imal effective concentration (MEC), but below the minimal toxic concentration (MTC). Clinical pharmacokinetics is about all the factors that determine variability in the Cp and its time-course. The various factors are dealt with in subsequent chapters. Ideal therapeutics: effi cacy without toxicity Minimum Toxic Concentration (MTC) Ideal dosing Minimum Effective Concentration (MEC) Drug concentration Time The graph shows a continuous IV infusion at steady state, where the dose-rate is exactly appropriate for the patient’s clearance (CL). Inappropriate dosing Dosing too high in relation to the patient’s CL – toxicity likely Minimum Toxic Concentration (MTC) Minimum Effective Concentration (MEC) Dosing too low in relation to the Drug concentration patient’s CL – drug may be ineffective Time Some reasons for variation in CL Low CL High CL Normal variation Normal variation Renal impairment Increased renal blood fl ow Genetic poor metabolism Genetic hypermetabolism Liver impairment Enzyme induction Enzyme inhibition Old age/neonate 2 General overview Clinical Pharmacokinetics Pharmacokinetic factors determining ideal therapeutics If immediate effect is needed, a loading dose (LD) must be given to achieve a desired 1 concentration. The LD is determined by the volume of distribution (Vd). -

IDSA Guideline Review
1 Infectious Diseases Society of America Antimicrobial Resistant Treatment Guidance: Gram- Negative Bacterial Infections A Focus on Extended-Spectrum β-lactamase Producing Enterobacterales (ESBL-E), Carbapenem- Resistant Enterobacterales (CRE), and Pseudomonas aeruginosa with Difficult-to-Treat Resistance (DTR- P. aeruginosa) Guideline Summary by Kendra Suder, PharmD Candidate General Recommendations Considerations for choosing empiric therapy: o Local susceptibility patterns/ antibiogram o Patient’s previous organisms isolated & susceptibility data in the last 6 months o Antibiotic exposure in the last 30 days What if a patient was empirically placed on an antibiotic regimen that is now considered inactive against the organism based on susceptibility data? o If an inactive agent was started empirically for cystitis and patient improves, no change in antibiotic or extension of therapy is necessary o However, for other types of infections, guidelines recommend a change to an active regimen for a full treatment course (dated from the start of active therapy) Extended-Spectrum β-Lactamase-Producing Enterobacterales (ESBL-E) Extended-spectrum β-lactamase o Enzymes that inactivate most penicillins, cephalosporins, and aztreonam o Most commonly produced by Escherichia coli, Klebsiella pneumoniae, Klebsiella oxytoca, and Proteus mirabilis (guideline recommendations refer to ESBL-E infections caused by these organisms) o Most common type in the US is the CTX-M, especially CTX-M-15 In institutions that do not perform ESBL testing, a lack of -

Cefoxitin Versus Piperacillin– Tazobactam As Surgical Antibiotic Prophylaxis in Patients Undergoing Pancreatoduodenectomy: Protocol for a Randomised Controlled Trial
Open access Protocol BMJ Open: first published as 10.1136/bmjopen-2020-048398 on 4 March 2021. Downloaded from Cefoxitin versus piperacillin– tazobactam as surgical antibiotic prophylaxis in patients undergoing pancreatoduodenectomy: protocol for a randomised controlled trial Nicole M Nevarez ,1 Brian C Brajcich,2 Jason Liu,2,3 Ryan Ellis,2 Clifford Y Ko,2 Henry A Pitt,4 Michael I D'Angelica,5 Adam C Yopp1 To cite: Nevarez NM, ABSTRACT Strengths and limitations of this study Brajcich BC, Liu J, et al. Introduction Although antibiotic prophylaxis is Cefoxitin versus piperacillin– established in reducing postoperative surgical site tazobactam as surgical ► A major strength of this study is the multi- infections (SSIs), the optimal antibiotic for prophylaxis in antibiotic prophylaxis institutional, double- arm, randomised controlled in patients undergoing pancreatoduodenectomy (PD) remains unclear. The study trial design. objective is to evaluate if administration of piperacillin– pancreatoduodenectomy: ► A limitation of this study is that all perioperative care protocol for a randomised tazobactam as antibiotic prophylaxis results in decreased is at the discretion of the operating surgeon and is controlled trial. BMJ Open 30- day SSI rate compared with cefoxitin in patients not standardised. 2021;11:e048398. doi:10.1136/ undergoing elective PD. ► All data will be collected through the American bmjopen-2020-048398 Methods and analysis This study will be a multi- College of Surgeons National Surgical Quality ► Prepublication history for institution, double- arm, non- blinded randomised controlled Improvement Program, which is a strength for its this paper is available online. superiority trial. Adults ≥18 years consented to undergo PD ease of use but a limitation due to the variety of data To view these files, please visit for all indications who present to institutions participating included. -

Activity of Aztreonam in Combination with Ceftazidime-Avibactam Against
Diagnostic Microbiology and Infectious Disease 99 (2021) 115227 Contents lists available at ScienceDirect Diagnostic Microbiology and Infectious Disease journal homepage: www.elsevier.com/locate/diagmicrobio Activity of aztreonam in combination with ceftazidime–avibactam against serine- and metallo-β-lactamase–producing ☆ ☆☆ ★ Pseudomonas aeruginosa , , Michelle Lee a, Taylor Abbey a, Mark Biagi b, Eric Wenzler a,⁎ a College of Pharmacy, University of Illinois at Chicago, Chicago, IL, USA b College of Pharmacy, University of Illinois at Chicago, Rockford, IL, USA article info abstract Article history: Existing data support the combination of aztreonam and ceftazidime–avibactam against serine-β-lactamase Received 29 June 2020 (SBL)– and metallo-β-lactamase (MBL)–producing Enterobacterales, although there is a paucity of data against Received in revised form 15 September 2020 SBL- and MBL-producing Pseudomonas aeruginosa. In this study, 5 SBL- and MBL-producing P. aeruginosa (1 Accepted 19 September 2020 IMP, 4 VIM) were evaluated against aztreonam and ceftazidime–avibactam alone and in combination via broth Available online xxxx microdilution and time-kill analyses. All 5 isolates were nonsusceptible to aztreonam, aztreonam–avibactam, – – Keywords: and ceftazidime avibactam. Combining aztreonam with ceftazidime avibactam at subinhibitory concentrations Metallo-β-lactamase produced synergy and restored bactericidal activity in 4/5 (80%) isolates tested. These results suggest that the VIM combination of aztreonam and ceftazidime–avibactam may be a viable treatment option against SBL- and IMP MBL-producing P. aeruginosa. Aztreonam © 2020 Elsevier Inc. All rights reserved. Ceftazidime–avibactam Synergy 1. Introduction of SBLs and MBLs harbored by P. aeruginosa are fundamentally different than those in Enterobacterales (Alm et al., 2015; Kazmierczak et al., The rapid global dissemination of Ambler class B metallo-β- 2016; Periasamy et al., 2020; Poirel et al., 2000; Watanabe et al., lactamase (MBL) enzymes along with their increasing diversity across 1991). -

Pharmacodynamic Evaluation of Suppression of in Vitro Resistance In
www.nature.com/scientificreports OPEN Pharmacodynamic evaluation of suppression of in vitro resistance in Acinetobacter baumannii strains using polymyxin B‑based combination therapy Nayara Helisandra Fedrigo1, Danielle Rosani Shinohara1, Josmar Mazucheli2, Sheila Alexandra Belini Nishiyama1, Floristher Elaine Carrara‑Marroni3, Frederico Severino Martins4, Peijuan Zhu5, Mingming Yu6, Sherwin Kenneth B. Sy2 & Maria Cristina Bronharo Tognim1* The emergence of polymyxin resistance in Gram‑negative bacteria infections has motivated the use of combination therapy. This study determined the mutant selection window (MSW) of polymyxin B alone and in combination with meropenem and fosfomycin against A. baumannii strains belonging to clonal lineages I and III. To evaluate the inhibition of in vitro drug resistance, we investigate the MSW‑derived pharmacodynamic indices associated with resistance to polymyxin B administrated regimens as monotherapy and combination therapy, such as the percentage of each dosage interval that free plasma concentration was within the MSW (%TMSW) and the percentage of each dosage interval that free plasma concentration exceeded the mutant prevention concentration (%T>MPC). The MSW of polymyxin B varied between 1 and 16 µg/mL for polymyxin B‑susceptible strains. The triple combination of polymyxin B with meropenem and fosfomycin inhibited the polymyxin B‑resistant subpopulation in meropenem‑resistant isolates and polymyxin B plus meropenem as a double combination sufciently inhibited meropenem‑intermediate, and susceptible strains. T>MPC 90% was reached for polymyxin B in these combinations, while %TMSW was 0 against all strains. TMSW for meropenem and fosfomycin were also reduced. Efective antimicrobial combinations signifcantly reduced MSW. The MSW‑derived pharmacodynamic indices can be used for the selection of efective combination regimen to combat the polymyxin B‑resistant strain. -

FDA Drug Safety Communication: FDA Cautions About Dose Confusion and Medication Error with Antibacterial Drug Avycaz (Ceftazidime and Avibactam)
FDA Drug Safety Communication: FDA cautions about dose confusion and medication error with antibacterial drug Avycaz (ceftazidime and avibactam) Safety Announcement [09-22-2015] The U.S. Food and Drug Administration (FDA) is warning health care professionals about the risk for dosing errors with the intravenous antibacterial drug Avycaz (ceftazidime and avibactam) due to confusion about the drug strength displayed on the vial and carton labels. Avycaz was initially approved with the vial and carton labels displaying the individual strengths of the two active ingredients (i.e., 2 gram/0.5 gram); however, the product is dosed based on the sum of the active ingredients (i.e., 2.5 gram). To prevent medication errors, we have revised the labels to indicate that each vial contains Avycaz 2.5 gram, equivalent to ceftazidime 2 gram and avibactam 0.5 gram (see Photos). Avycaz is approved for intravenous administration to treat complicated infections in the urinary tract, or in combination with the antibacterial drug metronidazole to treat complicated infections in the abdomen in patients with limited or no alternative treatment options. Antibacterial drugs work by killing or stopping the growth of bacteria that can cause illness. Since Avycaz’s approval in February 2015, we have received reports of three medication error cases related to confusion on how the strength was displayed on the Avycaz vial and carton labels. Two cases stated that the errors occurred during preparation of the dose in the pharmacy. The third case described concern about the potential for confusion because the strength displayed for Avycaz differs from how the strength is displayed for other beta-lactam/beta-lactamase antibacterial drugs. -

Multidrug-Resistant Gram-Negative Bacteria
Multidrug-Resistant Gram-Negative Bacteria: Trends, Risk Factors, and Treatments A worldwide public health problem, antibiotic resistance leads to treatment-resistant infections associated with prolonged hospitalizations, increased cost, and greater risk for morbidity. Lee S. Engel MD, PhD CASE INTRODUCTION An 80-year-old female nursing home resident with Antibiotics have saved the lives of millions of people a history of dementia, anemia, atrial fibrillation, and have contributed to the major gains in life ex- hypertension, incontinence, and recurrent urinary pectancy over the last century. In US hospitals, 190 tract infections (UTIs), as well as a long-term Foley million doses of antibiotics are administered each catheter, is admitted because she was found to be day.1 Furthermore, more than 133 million courses febrile and less responsive than normal. The patient of antibiotics are prescribed each year for outpatients. has no drug allergies. Upon admission, she has a However, antibiotic use has also resulted in a major temperature of 38.9ºC, heart rate of 92 beats/min, health care challenge—the development and spread and blood pressure of 106/64 mm Hg. The patient of resistant bacteria. Worldwide, antimicrobial resis- opens her eyes to stimuli but does not speak. Aside tance is most evident in diarrheal diseases, respira- from poor dentition and an irregular heart rate, tory tract infections, meningitis, sexually transmitted findings on physical examination, which includes infections, and hospital and health care–acquired in- a pulmonary and abdominal examination, are nor- fections.2 Vancomycin-resistant enterococci, methi- mal. Serum chemistries demonstrate a white blood cillin-resistant Staphylococcus aureus, multidrug-resis- cell (WBC) count of 11,300 cells/mm3. -

General Items
Essential Medicines List (EML) 2019 Application for the inclusion of imipenem/cilastatin, meropenem and amoxicillin/clavulanic acid in the WHO Model List of Essential Medicines, as reserve second-line drugs for the treatment of multidrug-resistant tuberculosis (complementary lists of anti-tuberculosis drugs for use in adults and children) General items 1. Summary statement of the proposal for inclusion, change or deletion This application concerns the updating of the forthcoming WHO Model List of Essential Medicines (EML) and WHO Model List of Essential Medicines for Children (EMLc) to include the following medicines: 1) Imipenem/cilastatin (Imp-Cln) to the main list but NOT the children’s list (it is already mentioned on both lists as an option in section 6.2.1 Beta Lactam medicines) 2) Meropenem (Mpm) to both the main and the children’s lists (it is already on the list as treatment for meningitis in section 6.2.1 Beta Lactam medicines) 3) Clavulanic acid to both the main and the children’s lists (it is already listed as amoxicillin/clavulanic acid (Amx-Clv), the only commercially available preparation of clavulanic acid, in section 6.2.1 Beta Lactam medicines) This application makes reference to amendments recommended in particular to section 6.2.4 Antituberculosis medicines in the latest editions of both the main EML (20th list) and the EMLc (6th list) released in 2017 (1),(2). On the basis of the most recent Guideline Development Group advising WHO on the revision of its guidelines for the treatment of multidrug- or rifampicin-resistant (MDR/RR-TB)(3), the applicant considers that the three agents concerned be viewed as essential medicines for these forms of TB in countries. -

12. What's Really New in Antibiotic Therapy Print
What’s really new in antibiotic therapy? Martin J. Hug Freiburg University Medical Center EAHP Academy Seminars 20-21 September 2019 Newsweek, May 24-31 2019 Disclosures There are no conflicts of interest to declare EAHP Academy Seminars 20-21 September 2019 Antiinfectives and Resistance EAHP Academy Seminars 20-21 September 2019 Resistance of Klebsiella pneumoniae to Pip.-Taz. olates) EAHP Academy Seminars 20-21 September 2019 https://resistancemap.cddep.org/AntibioticResistance.php Multiresistant Pseudomonas Aeruginosa Combined resistance against at least three different types of antibiotics, 2017 EAHP Academy Seminars 20-21 September 2019 https://atlas.ecdc.europa.eu/public/index.aspx Distribution of ESBL producing Enterobacteriaceae EAHP Academy Seminars 20-21 September 2019 Rossolini GM. Global threat of Gram-negative antimicrobial resistance. 27th ECCMID, Vienna, 2017, IS07 Priority Pathogens Defined by the World Health Organisation Critical Priority High Priority Medium Priority Acinetobacter baumanii Enterococcus faecium Streptococcus pneumoniae carbapenem-resistant vancomycin-resistant penicillin-non-susceptible Pseudomonas aeruginosa Helicobacter pylori Haemophilus influenzae carbapenem-resistant clarithromycin-resistant ampicillin-resistant Enterobacteriaceae Salmonella species Shigella species carbapenem-resistant fluoroquinolone-resistant fluoroquinolone-resistant Staphylococcus aureus vancomycin or methicillin -resistant Campylobacter species fluoroquinolone-resistant Neisseria gonorrhoae 3rd gen. cephalosporin-resistant -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -
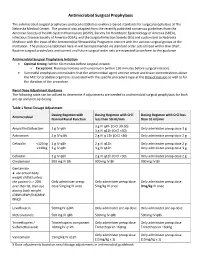
Antimicrobial Surgical Prophylaxis
Antimicrobial Surgical Prophylaxis The antimicrobial surgical prophylaxis protocol establishes evidence-based standards for surgical prophylaxis at The Nebraska Medical Center. The protocol was adapted from the recently published consensus guidelines from the American Society of Health-System Pharmacists (ASHP), Society for Healthcare Epidemiology of America (SHEA), Infectious Disease Society of America (IDSA), and the Surgical Infection Society (SIS) and customized to Nebraska Medicine with the input of the Antimicrobial Stewardship Program in concert with the various surgical groups at the institution. The protocol established here-in will be implemented via standard order sets utilized within One Chart. Routine surgical prophylaxis and current and future surgical order sets are expected to conform to this guidance. Antimicrobial Surgical Prophylaxis Initiation Optimal timing: Within 60 minutes before surgical incision o Exceptions: Fluoroquinolones and vancomycin (within 120 minutes before surgical incision) Successful prophylaxis necessitates that the antimicrobial agent achieve serum and tissue concentrations above the MIC for probable organisms associated with the specific procedure type at the time of incision as well as for the duration of the procedure. Renal Dose Adjustment Guidance The following table can be utilized to determine if adjustments are needed to antimicrobial surgical prophylaxis for both pre-op and post-op dosing. Table 1 Renal Dosage Adjustment Dosing Regimen with Dosing Regimen with CrCl Dosing Regimen with -

Penicillin Skin Testing
Penicillin Skin Testing Penicillin Skin Testing: Frequently Asked Questions Benzylpenicilloyl Polylysine (Pre-Pen®) and Diluted Penicillin G June 2012 VA Pharmacy Benefits Management Services, Medical Advisory Panel and VISN Pharmacist Executives Benzylpenicilloyl polylysine (Pre-Pen®) was FDA approved in 2009 for the assessment of sensitization to penicillin in those patients suspected, based upon previous experience, of having a clinical hypersensitivity to penicillin. Penicillin skin testing is the most reliable way to evaluate patients for IgE-mediated penicillin allergy. Skin testing is conducted using benzylpenicilloyl polylysine (major determinant), penicillin G diluted with normal saline to 10,000 units/ml (minor determinant), a positive and negative control. Testing with both the major and minor determinant of penicillin can identify up to 97% of patients with an immediate hypersensitivity to penicillin. The intent of this document is to help guide appropriate use of penicillin skin testing. 1. WHEN IS PENICILLIN SKIN TESTING INDICATED? Penicillin skin testing can be considered in those patients with a prior history of hypersensitivity to penicillin or allergy to penicillin in situations where the provider considers penicillin the drug of choice or prefers treatment with penicillin. If skin testing is indicated, the patient should be referred to a VA Allergy Specialist or other appropriately trained physician who is experienced in the application and interpretation of PCN skin testing, as locally designated. Although approximately 10% of patients will remain allergic to penicillin their entire lives, a large majority will stop expressing penicillin-specific IgE-mediated antibodies and can be safely treated with penicillin. Skin testing with benzylpenicilloyl polylysine is contraindicated in patients who are known to be extremely hypersensitive to penicillin and in those patients who have experienced a systemic or marked local reaction to prior administration of benzylpenicilloyl polylysine.