Synaptic Pharmacology of the Superior Olivary Complex Studied in Mouse Brain Slice
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Neurotransmitters-Drugs Andbrain Function.Pdf
Neurotransmitters, Drugs and Brain Function. Edited by Roy Webster Copyright & 2001 John Wiley & Sons Ltd ISBN: Hardback 0-471-97819-1 Paperback 0-471-98586-4 Electronic 0-470-84657-7 Neurotransmitters, Drugs and Brain Function Neurotransmitters, Drugs and Brain Function. Edited by Roy Webster Copyright & 2001 John Wiley & Sons Ltd ISBN: Hardback 0-471-97819-1 Paperback 0-471-98586-4 Electronic 0-470-84657-7 Neurotransmitters, Drugs and Brain Function Edited by R. A. Webster Department of Pharmacology, University College London, UK JOHN WILEY & SONS, LTD Chichester Á New York Á Weinheim Á Brisbane Á Singapore Á Toronto Neurotransmitters, Drugs and Brain Function. Edited by Roy Webster Copyright & 2001 John Wiley & Sons Ltd ISBN: Hardback 0-471-97819-1 Paperback 0-471-98586-4 Electronic 0-470-84657-7 Copyright # 2001 by John Wiley & Sons Ltd. Bans Lane, Chichester, West Sussex PO19 1UD, UK National 01243 779777 International ++44) 1243 779777 e-mail +for orders and customer service enquiries): [email protected] Visit our Home Page on: http://www.wiley.co.uk or http://www.wiley.com All Rights Reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted, in any form or by any means, electronic, mechanical, photocopying, recording, scanning or otherwise, except under the terms of the Copyright, Designs and Patents Act 1988 or under the terms of a licence issued by the Copyright Licensing Agency Ltd, 90 Tottenham Court Road, London W1P0LP,UK, without the permission in writing of the publisher. Other Wiley Editorial Oces John Wiley & Sons, Inc., 605 Third Avenue, New York, NY 10158-0012, USA WILEY-VCH Verlag GmbH, Pappelallee 3, D-69469 Weinheim, Germany John Wiley & Sons Australia, Ltd. -

Auditory and Vestibular Systems Objective • to Learn the Functional
Auditory and Vestibular Systems Objective • To learn the functional organization of the auditory and vestibular systems • To understand how one can use changes in auditory function following injury to localize the site of a lesion • To begin to learn the vestibular pathways, as a prelude to studying motor pathways controlling balance in a later lab. Ch 7 Key Figs: 7-1; 7-2; 7-4; 7-5 Clinical Case #2 Hearing loss and dizziness; CC4-1 Self evaluation • Be able to identify all structures listed in key terms and describe briefly their principal functions • Use neuroanatomy on the web to test your understanding ************************************************************************************** List of media F-5 Vestibular efferent connections The first order neurons of the vestibular system are bipolar cells whose cell bodies are located in the vestibular ganglion in the internal ear (NTA Fig. 7-3). The distal processes of these cells contact the receptor hair cells located within the ampulae of the semicircular canals and the utricle and saccule. The central processes of the bipolar cells constitute the vestibular portion of the vestibulocochlear (VIIIth cranial) nerve. Most of these primary vestibular afferents enter the ipsilateral brain stem inferior to the inferior cerebellar peduncle to terminate in the vestibular nuclear complex, which is located in the medulla and caudal pons. The vestibular nuclear complex (NTA Figs, 7-2, 7-3), which lies in the floor of the fourth ventricle, contains four nuclei: 1) the superior vestibular nucleus; 2) the inferior vestibular nucleus; 3) the lateral vestibular nucleus; and 4) the medial vestibular nucleus. Vestibular nuclei give rise to secondary fibers that project to the cerebellum, certain motor cranial nerve nuclei, the reticular formation, all spinal levels, and the thalamus. -

Anatomy of the Superior Olivary Complex.Pdf
Douglas Oliver University of Connecticut Health Center SUPERIOR OLIVE Auditory Pathways Auditory CORTEX GLUT Cortex GABA GLY Medial Geniculate MGB Body Inferior IC Colliculus DLL DLL COCHLEA VLL VLL DCN VCN SOC Auditory Pathways IC Organization of Superior Olivary Complex . Subdivisions and Cytoarchitecture . Neuron types . Inputs . Outputs . Synapses . Basic Circuit Cytoarchitecture of Superior Olivary Complex LSO LSO MSO MSO MNTB D MNTB M (somata & dendrites) (axons & endings) Tsuchitani, 1978, Fig. 10 Comparative anatomy of SOC Tetsufumi Ito & Shig Kuwada Binaural Basic Circuits 8 ‐ 9 Brodal Fig MSO: medial superior olive; LSO: lateral superior olive NTB: nucleus of trapezoid body; IC: inferior colliculus MSO Principle glutamate Cells . Fusiform . Bipolar . Disc‐shaped . Each dendrite innervated by a different side MSO‐In situ hybridization RPO MSO MNTB SPO LSO VGLUT1 VGLUT2 VIAAT NISSL MSO Inputs and Synapses H=high frequency EI - ILD L=low frequency EE - ITD LSO MSO L L B H B B H G LNTB TO LSO MNTB E=Excitation (glutamate) ‐‐‐ I=Inhibition (glycine) ITD CODING Unlike retinal targets, the cochlear nuclei contain maps of frequency, not location. So how does the auditory system know ‘where’ a sound is coming from? T + ITD T By comparing the interaural time differences (ITD) between the ears How is this accomplished?... LSO MSO Right Input A Right Input B C Time Code Time Code E E A A B B C C D D E E Output Output abcde Place Code abcde Place Code Excitation MSO creates a response to Left Input Left Input Inhibition interaural time differences I Time Code E Time Code DEMSO "peak" unit LSO "trough" unit ITD ITD Figure 14.2 Binaural Responses in MSO MSO Summary . -

The Superior Olivary Complex +
Excitatory and inhibitory transmission in the superior olivary complex. Ian D. Forsythe, Matt Barker, Margaret Barnes-Davies, Brian Billups, Paul Dodson, Fatima Osmani, Steven Owens and Adrian Wong. Department of Cell Physiology and Pharmacology, University of Leicester, Leicester LE1 9HN. UK. The timing and pattern of action potentials propagating into the brainstem from both cochleae contain information about the azimuth location of that sound in auditory space. This binaural information is integrated in the superior olivary complex. This part of the auditory pathway is adapted for fast conduction speeds and the preservation of timing information with several complimentary mechanisms (see Oertel, 1999; Trussell, 1999). There are large diameter axons terminating in giant somatic synapses that activate receptor ion channels with fast kinetics. The resultant postsynaptic potentials generated in the receiving neuron are integrated with a suite of voltage-gated ion channels that determine the action potential threshold, duration and repetitive firing properties. We have studied presynaptic and postsynaptic mechanisms that regulate efficacy, timing and integration of synaptic responses in the medial nucleus of the trapezoid body and the medial and lateral superior olives. Presynaptic calcium currents in the calyx of Held. The calyx of Held is a giant synaptic terminal that forms around the soma of principal cells in the Medial Nucleus of the Trapezoid Body (MNTB) (Forsythe, 1994). Each MNTB neuron receives a single calyx. Action potentials propagating into the synaptic terminal trigger the opening of P-type calcium channels (Forsythe et al. 1998) which in turn trigger the release of glutamate into the synaptic cleft (Borst et al., 1995). -

Mechanism of Action of Ethylenediamine On
Stone, T.W., Lui, C. and Addae, J.I. (2010) Effects of ethylenediamine – a putative GABA-releasing agent – on rat hippocampal slices and neocortical activity in vivo. European Journal of Pharmacology, 650 (2-3). pp. 568-578. ISSN 0014-2999. http://eprints.gla.ac.uk/46340/ Deposited on: 22 November 2010 Enlighten – Research publications by members of the University of Glasgow http://eprints.gla.ac.uk Effects of ethylenediamine – a putative GABA-releasing agent – on rat hippocampal slices and neocortical activity in vivo Trevor W Stone1 , Caleb Lui1 and Jonas I. Addae2 1 Neuroscience and Molecular Pharmacology, Faculty of Biomedical and Life Sciences, University of Glasgow, Glasgow G12 8QQ, U.K. 2 Dept of Preclinical Sciences, St. Augustine Campus University of the West Indies, Trinidad and Tobago. Correspondence: Prof T. W. Stone, West Medical Building, University of Glasgow, Glasgow G12 8QQ, UK. Tel - +44 141 330 4481; fax - +44 141 330 5481; e-mail - [email protected] 1 ABSTRACT The simple diamine diaminoethane (ethylenediamine, EDA) has been shown to activate GABA receptors in the central and peripheral nervous systems, partly by a direct action and partly by releasing endogenous GABA. These effects have been shown to be produced by the complexation of EDA with bicarbonate to form a carbamate. The present work has compared EDA, GABA and β-alanine responses in rat CA1 neurons using extracellular and intracellular recordings, as well as neocortical evoked potentials in vivo. Superfusion of GABA onto hippocampal slices produced depolarisation and a decrease of field epsps, both effects fading rapidly, but showing sensitivity to blockade by bicuculline. -
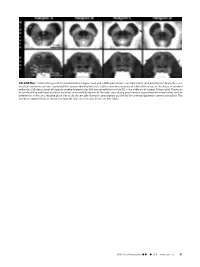
ON-LINE FIG 1. Selected Images of the Caudal Midbrain (Upper Row
ON-LINE FIG 1. Selected images of the caudal midbrain (upper row) and middle pons (lower row) from 4 of 13 total postmortem brains illustrate excellent anatomic contrast reproducibility across individual datasets. Subtle variations are present. Note differences in the shape of cerebral peduncles (24), decussation of superior cerebellar peduncles (25), and spinothalamic tract (12) in the midbrain of subject D (top right). These can be attributed to individual anatomic variation, some mild distortion of the brain stem during procurement at postmortem examination, and/or differences in the axial imaging plane not easily discernable during its prescription parallel to the anterior/posterior commissure plane. The numbers in parentheses in the on-line legends refer to structures in the On-line Table. AJNR Am J Neuroradiol ●:●●2019 www.ajnr.org E1 ON-LINE FIG 3. Demonstration of the dentatorubrothalamic tract within the superior cerebellar peduncle (asterisk) and rostral brain stem. A, Axial caudal midbrain image angled 10° anterosuperior to posteroinferior relative to the ACPC plane demonstrates the tract traveling the midbrain to reach the decussation (25). B, Coronal oblique image that is perpendicular to the long axis of the hippocam- pus (structure not shown) at the level of the ventral superior cerebel- lar decussation shows a component of the dentatorubrothalamic tract arising from the cerebellar dentate nucleus (63), ascending via the superior cerebellar peduncle to the decussation (25), and then enveloping the contralateral red nucleus (3). C, Parasagittal image shows the relatively long anteroposterior dimension of this tract, which becomes less compact and distinct as it ascends toward the thalamus. ON-LINE FIG 2. -

Interactions Between Ethanol and the Endocannabinoid System at Gabaergic Synapses on Basolateral Amygdala Principal Neurons
Alcohol 49 (2015) 781e794 Contents lists available at ScienceDirect Alcohol journal homepage: http://www.alcoholjournal.org/ Interactions between ethanol and the endocannabinoid system at GABAergic synapses on basolateral amygdala principal neurons Giuseppe Talani a,b, David M. Lovinger a,* a Section on Synaptic Pharmacology, Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health, Bethesda, MD 20892, USA b Institute of Neuroscience, National Research Council, Monserrato, Cagliari, Italy article info abstract Article history: The basolateral amygdala (BLA) plays crucial roles in stimulus value coding, as well as drug and alcohol Received 14 April 2015 dependence. Ethanol alters synaptic transmission in the BLA, while endocannabinoids (eCBs) produce Received in revised form presynaptic depression at BLA synapses. Recent studies suggest interactions between ethanol and eCBs 11 August 2015 that have important consequences for alcohol drinking behavior. To determine how ethanol and eCBs Accepted 25 August 2015 interact in the BLA, we examined the physiology and pharmacology of GABAergic synapses onto BLA pyramidal neurons in neurons from young rats. Application of ethanol at concentrations relevant to intoxication increased, in both young and adult animals, the frequency of spontaneous and miniature Keywords: Alcohol GABAergic inhibitory postsynaptic currents, indicating a presynaptic site of ethanol action. Ethanol did CB1 receptor not potentiate sIPSCs during inhibition of adenylyl cyclase while still exerting its effect during inhibition Synapse of protein kinase A. Activation of type 1 cannabinoid receptors (CB1) in the BLA inhibited GABAergic Inhibition transmission via an apparent presynaptic mechanism, and prevented ethanol potentiation. Surprisingly, Arachidonoyl ethanolamide ethanol potentiation was also prevented by CB1 antagonists/inverse agonists. -

Evolution of Mammalian Sound Localization Circuits: a Developmental Perspective
Progress in Neurobiology 141 (2016) 1–24 Contents lists available at ScienceDirect Progress in Neurobiology journal homepage: www.elsevier.com/locate/pneurobio Review article Evolution of mammalian sound localization circuits: A developmental perspective a,b, Hans Gerd Nothwang * a Neurogenetics group, Center of Excellence Hearing4All, School of Medicine and Health Sciences, Carl von Ossietzky University Oldenburg, 26111 Oldenburg, Germany b Research Center for Neurosensory Science, Carl von Ossietzky University Oldenburg, 26111 Oldenburg, Germany A R T I C L E I N F O A B S T R A C T Article history: Localization of sound sources is a central aspect of auditory processing. A unique feature of mammals is Received 29 May 2015 the smooth, tonotopically organized extension of the hearing range to high frequencies (HF) above Received in revised form 27 February 2016 10 kHz, which likely induced positive selection for novel mechanisms of sound localization. How this Accepted 27 February 2016 change in the auditory periphery is accompanied by changes in the central auditory system is unresolved. Available online 28 March 2016 + I will argue that the major VGlut2 excitatory projection neurons of sound localization circuits (dorsal cochlear nucleus (DCN), lateral and medial superior olive (LSO and MSO)) represent serial homologs with Keywords: modifications, thus being paramorphs. This assumption is based on common embryonic origin from an Brainstem + + Atoh1 /Wnt1 cell lineage in the rhombic lip of r5, same cell birth, a fusiform cell morphology, shared Evolution Homology genetic components such as Lhx2 and Lhx9 transcription factors, and similar projection patterns. Such a Innovation parsimonious evolutionary mechanism likely accelerated the emergence of neurons for sound Rhombomere localization in all three dimensions. -

Acoustically Responsive Fibers in the Vestibular Nerve of the Cat
The Journal of Neuroscience, October 1994, 74(10): 6056-6070 Acoustically Responsive Fibers in the Vestibular Nerve of the Cat Michael P. McCue1v2*a and John J. Guinan, Jr.r.2.3-4 ‘Eaton-Peabody Laboratory of Auditory Physiology, Department of Otolaryngology, Massachusetts Eye and Ear Infirmary, Boston, Massachusetts 02114, 2Harvard-MIT Division of Health Science and Technology and Research Laboratory of Electronics, and 3Department of Electrical Engineering and Computer Science, Massachusetts Institute of Technology, Cambridge, Massachusetts 02139, and 4Department of Otology and Laryngology, Harvard Medical School, Boston, Massachusetts 02115 Recordings were made from single afferent fibers in the and levels within the normal range of human hearing. We inferior vestibular nerve, which innervates the saccule and suggest a number of auditory roles that these fibers may posterior semicircular canal. A substantial portion of the fi- play in the everyday life of mammals. bers with irregular background activity increased their firing [Key words: saccule, otoliths, auditory system, mamma- in response to moderately intense clicks and tones. lian sound reception, middle-ear muscles, cochlear nucleus] In responsive fibers, acoustic clicks evoked action poten- tials with minimum latencies of I 1 .O msec. Fibers fell into The vertebrate inner ear contains several senseorgans involved two classes, with the shortest latency either to condensation in the maintenance of equilibrium and the detection of vibra- clicks (PUSH fibers) or to rarefaction clicks (PULL fibers). tion. The precise sensory role assumedby homologous organs Low-frequency (800 Hz) tone bursts at moderately high sound varies among species.For example, the sacculeis thought to act levels (>80 dB SPL) caused synchronization of spikes to asa linear accelerometerin mammals(Fernindez and Goldberg, preferred phases of the tone cycle. -

At the Gabaa Receptor
THE EFFECTS OF CHRONIC ETHANOL INTAKE ON THE ALLOSTERIC INTERACTION BE T WEEN GABA AND BENZODIAZEPINE AT THE GABAA RECEPTOR THESIS Presented to the Graduate Council of the University of North Texas in Partial Fulfillment of the Requirements For the Degree of MASTER OF SCIENCE By Jianping Chen, B.S., M.S. Denton, Texas May, 1992 Chen, Jianping, The Effects of Chronic Ethanol Intake on the Allsteric Interaction Between GABA and BenzodiazeDine at the GABAA Receptor. Master of Science (Biomedical Sciences/Pharmacology), May, 1992, 133 pp., 4 tables, 3.0 figures, references, 103 titles. This study examined the effects of chronic ethanol intake on the density, affinity, and allosteric modulation of rat brain GABAA receptor subtypes. In the presence of GABA, the apparent affinity for the benzodiazepine agonist flunitrazepam was increased and for the inverse agonist R015-4513 was decreased. No alteration in the capacity of GABA to modulate flunitrazepam and R015-4513 binding was observed in membranes prepared from cortex, hippocampus or cerebellum following chronic ethanol intake or withdrawal. The results also demonstrate two different binding sites for [3H]RO 15-4513 in rat cerebellum that differ in their affinities for diazepam. Chronic ethanol treatment and withdrawal did not significantly change the apparent affinity or density of these two receptor subtypes. ACKNOWLEDGEMENT I would like to express my sincere thanks to my major professor, Dr. Michael W. Martin. .I deeply appreciate his guidance and direction which initiated this study, and his kindness in sharing his laboratory facilities with me. His suggestions, patience, encouragement and support in the laboratory have contributed significantly to my understanding of the receptor mechanism of drug action. -

Processing in the Cochlear Nucleus
Processing in The Cochlear Nucleus Alan R. Palmer Medical Research Council Institute of Hearing Research University Park Nottingham NG7 2RD, UK The Auditory Nervous System Cortex Cortex MGB Medial Geniculate Body Excitatory GABAergic IC Inferior Colliculus Glycinergic DNLL Nuclei of the Lateral Lemniscus Lateral Lemniscus Cochlear Nucleus DCN PVCN MSO Lateral Superior Olive AVCN Medial Superior Olive Cochlea MNTB Medial Nucleus of the Trapezoid Body Superior Olive The cochlear nucleus is the site of termination of fibres of the auditory nerve Cochlear Nucleus Auditory Nerve Cochlea 1 Frequency Tonotopicity Basilar membrane Inner hair cell Auditory nerve Fibre To the brain Each auditory-nerve fibre responds only to a narrow range of frequencies Tuning curve Action potential Evans 1975 2 Palmer and Evans 1975 There are many overlapping single-fibre tuning curves in the auditory nerve Audiogram Palmer and Evans 1975 Tonotopic Organisation Lorente - 1933 3 Tonotopic Organisation Base Anterior Cochlea Characteristic Basilar Membrane Frequency Hair Cells Auditory Nerve Apex Cochlear Nucleus Spiral Ganglion Posterior Tonotopic projection of auditory-nerve fibers into the cochlear nucleus Ryugo and Parks, 2003 The cochlear nucleus: the first auditory nucleus in the CNS Best frequency Position along electrode track (mm) Evans 1975 4 stellate (DCN) Inhibitory Synapse Excitatory Synapse DAS to inferior colliculus cartwheel fusiform SUPERIOR OLIVARY giant COMPLEX INFERIOR COLLICULUS granule vertical vertical OCB AANN to CN & IC via TB golgi DORSAL -

Universität Leipzig Fakultät Für Biowissenschaften, Pharmazie Und Psychologie
Universität Leipzig Fakultät für Biowissenschaften, Pharmazie und Psychologie IMPRS NeuroCom" Marc Schönwiesner" " " Auditory and vestibular system Michael Hawke MD / CC-BY-SA-4.0 The middle ear The middle ear Bekesy’s fluid model The traveling wave Problem: traveling waves have very broad peaks – frequency discrimination can not be based on the location of the peak vibration! Masking masker Sound intensity Sound frequency inaudible audible Masking and mp3 encoding Idea: signals that are inaudible because of masking can be removed from the file to save space. The organ of Corti Hair cells Outer hair cell motility The active cochlea model (video by J. Ashmore 1987) Connections from outer and inner hair cells Microtubule inside of a cilium 9 doublet microtubules + 2 central microtubule Dynein arms http://scienceblogs.com/transcript/upload/2006/08/axoneme.gif /www.uni-mainz.de/FB/Medizin/Anatomie Tilting of stereocilia bundles cause graded de- or hyperpolarization in hair cells (and modulation of action potential rates in afferent nerve fibers) Intracellular recording Mechanical stimulation Extracellular recording from afferent nerve fiber The lateral line system Adequate stimulus: Tilting of stereovilli by water movement Lateral line Afferent and efferent innervation Dudel, Menzel, Schmidt Hair cell: One type of sensory cell, different functions through specific accessory structures A. Perception and integration of water-flow pattern at the body surface Vestibular system Vestibular system The vestibular labyrinth answers two basic questions: Where am I going? Which way is up? The vestibular labyrinth answers the two questions by sensing: Head angular acceleration (semicircular canals) = Head rotation Head linear acceleration (saccule and utricle)" = Translational motion.