The SR Protein Family Peter J Shepard and Klemens J Hertel
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cooperativity of Imprinted Genes Inactivated by Acquired Chromosome 20Q Deletions
Cooperativity of imprinted genes inactivated by acquired chromosome 20q deletions Athar Aziz, … , Anne C. Ferguson-Smith, Anthony R. Green J Clin Invest. 2013;123(5):2169-2182. https://doi.org/10.1172/JCI66113. Research Article Oncology Large regions of recurrent genomic loss are common in cancers; however, with a few well-characterized exceptions, how they contribute to tumor pathogenesis remains largely obscure. Here we identified primate-restricted imprinting of a gene cluster on chromosome 20 in the region commonly deleted in chronic myeloid malignancies. We showed that a single heterozygous 20q deletion consistently resulted in the complete loss of expression of the imprinted genes L3MBTL1 and SGK2, indicative of a pathogenetic role for loss of the active paternally inherited locus. Concomitant loss of both L3MBTL1 and SGK2 dysregulated erythropoiesis and megakaryopoiesis, 2 lineages commonly affected in chronic myeloid malignancies, with distinct consequences in each lineage. We demonstrated that L3MBTL1 and SGK2 collaborated in the transcriptional regulation of MYC by influencing different aspects of chromatin structure. L3MBTL1 is known to regulate nucleosomal compaction, and we here showed that SGK2 inactivated BRG1, a key ATP-dependent helicase within the SWI/SNF complex that regulates nucleosomal positioning. These results demonstrate a link between an imprinted gene cluster and malignancy, reveal a new pathogenetic mechanism associated with acquired regions of genomic loss, and underline the complex molecular and cellular consequences of “simple” cancer-associated chromosome deletions. Find the latest version: https://jci.me/66113/pdf Research article Cooperativity of imprinted genes inactivated by acquired chromosome 20q deletions Athar Aziz,1,2 E. Joanna Baxter,1,2,3 Carol Edwards,4 Clara Yujing Cheong,5 Mitsuteru Ito,4 Anthony Bench,3 Rebecca Kelley,1,2 Yvonne Silber,1,2 Philip A. -

Lupus-Associated Endogenous Retroviral LTR Polymorphism and Epigenetic Imprinting Promote HRES-1/Rab4 Expression and Mtor Activation
Lupus-associated endogenous retroviral LTR polymorphism and epigenetic imprinting promote HRES-1/Rab4 expression and mTOR activation Aparna Godavarthy, … , Katalin Banki, Andras Perl JCI Insight. 2019. https://doi.org/10.1172/jci.insight.134010. Research In-Press Preview Immunology Graphical abstract Find the latest version: https://jci.me/134010/pdf LUPUS-ASSOCIATED ENDOGENOUS RETROVIRAL LTR POLYMORPHISM AND EPIGENETIC IMPRINTING PROMOTE HRES-1/RAB4 EXPRESSION AND MTOR ACTIVATION Aparna Godavarthy*1, Ryan Kelly*1, John Jimah1, Miguel Beckford1, Tiffany Caza1,2, David Fernandez1,2, Nick Huang1,3, Manuel Duarte1,2, Joshua Lewis1,2, Hind J. Fadel4, Eric M. Poeschla4, Katalin Banki5, and Andras Perl1,2,3 * These authors contributed equally to the study. 1, Division of Rheumatology, Department of Medicine; 2, Department of Microbiology and Immunology; 3, Department of Biochemistry and Molecular Biology, and 5, Department of Pathology, State University of New York, Upstate Medical University, College of Medicine, 750 East Adams Street, Syracuse, New York 13210; 4, Department of Molecular Medicine; Mayo Clinic College of Medicine, 200 First Street SW, Rochester 55905, USA; Correspondence: Andras Perl, M.D., Ph.D. , State University of New York, College of Medicine, 750 East Adams Street, Syracuse, New York 13210; Phone: (315) 464-4194; Fax: (315) 464- 4176; E-mail: [email protected] Key Words: Systemic Lupus Erythematosus, T Cells, HRES-1/Rab4. mTOR, Autoimmunity The authors have declared that no conflict of interest exists. Supplementary Materials include Supplemental Methods and Supplementary Figures S1-S26. 1 ABSTRACT Overexpression and long terminal repeat (LTR) polymorphism of the HRES-1/Rab4 human endogenous retrovirus locus have been associated with T-cell activation and disease manifestations in systemic lupus erythematosus (SLE). -

Topoisomerase 1 Suppresses Replication Stress and Genomic Instability by Preventing Interference Between Replication and Transcription
Topoisomerase 1 suppresses replication stress and genomic instability by preventing interference between replication and transcription. Sandie Tuduri, Laure Crabbé, Chiara Conti, Hélène Tourrière, Heidi Holtgreve-Grez, Anna Jauch, Véronique Pantesco, John de Vos, Aubin Thomas, Charles Theillet, et al. To cite this version: Sandie Tuduri, Laure Crabbé, Chiara Conti, Hélène Tourrière, Heidi Holtgreve-Grez, et al.. Topoi- somerase 1 suppresses replication stress and genomic instability by preventing interference between replication and transcription.. Nature Cell Biology, Nature Publishing Group, 2009, 11 (11), pp.1315- 1324. 10.1038/ncb1984. hal-00430775 HAL Id: hal-00430775 https://hal.archives-ouvertes.fr/hal-00430775 Submitted on 14 Jun 2010 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Topoisomerase I suppresses genomic instability by preventing interference between replication and transcription Sandie Tuduri 1,2, Laure Crabbé 1, Chiara Conti 3, Hélène Tourrière 1, Heidi Holtgreve-Grez 4, Anna Jauch 4, Véronique Pantesco 5, John De Vos 5, Aubin -

Serine Arginine-Rich Protein-Dependent Suppression Of
Serine͞arginine-rich protein-dependent suppression of exon skipping by exonic splicing enhancers El Che´ rif Ibrahim*, Thomas D. Schaal†, Klemens J. Hertel‡, Robin Reed*, and Tom Maniatis†§ *Department of Cell Biology, Harvard Medical School, 240 Longwood Avenue, Boston, MA 02115; †Department of Molecular and Cellular Biology, Harvard University, 7 Divinity Avenue, Cambridge, MA 02138; and ‡Department of Microbiology and Molecular Genetics, University of California, Irvine, CA 92697-4025 Contributed by Tom Maniatis, January 28, 2005 The 5 and 3 splice sites within an intron can, in principle, be joined mechanisms by which exon skipping is prevented. Our data to those within any other intron during pre-mRNA splicing. How- reveal that splicing to the distal 3Ј splice site is suppressed by ever, exons are joined in a strict 5 to 3 linear order in constitu- proximal exonic sequences and that SR proteins are required for tively spliced pre-mRNAs. Thus, specific mechanisms must exist to this suppression. Thus, SR protein͞exonic enhancer complexes prevent the random joining of exons. Here we report that insertion not only function in exon and splice-site recognition but also play of exon sequences into an intron can inhibit splicing to the a role in ensuring that 5Ј and 3Ј splice sites within the same intron .downstream 3 splice site and that this inhibition is independent of are used, thus suppressing exon skipping intron size. The exon sequences required for splicing inhibition were found to be exonic enhancer elements, and their inhibitory Materials and Methods activity requires the binding of serine͞arginine-rich splicing fac- Construction of Plasmids. -

The Solution Structure of Dead End Bound to AU-Rich RNA Reveals an Unprecedented Mode of Tandem RRM-RNA Recognition Required for Mrna Repression
bioRxiv preprint doi: https://doi.org/10.1101/572156; this version posted March 9, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. The solution structure of Dead End bound to AU-rich RNA reveals an unprecedented mode of tandem RRM-RNA recognition required for mRNA repression Authors Malgorzata M. Duszczyk1*, Harry Wischnewski2, Tamara Kazeeva1, Fionna E. Loughlin1,4, Christine von Schroetter1, Ugo Pradère3, Jonathan Hall3, Constance Ciaudo2*, Frédéric H.-T. Allain1,5* Affiliations 1Institute of Molecular Biology and Biophysics and 2Institute of Molecular Health Sciences, Department of Biology, 3Institute of Pharmaceutical Sciences, Department of Chemistry and Applied Biosciences, ETH Zürich, 8093, Zürich, Switzerland 4Present Address: Monash Biomedicine Discovery Institute, Department of Biochemistry and Molecular Biology, Monash University, Clayton, 3800 VIC, Australia 5Lead Contact Contact Information *To whom correspondence should be addressed. E-mail: [email protected] (M.M.D.), [email protected] (C.C.), [email protected] (F.H.- T.A.) Abstract Dead End (DND1) is an RNA-binding protein essential for germline development through its role in the clearance of AU-rich mRNAs. Here, we present the solution structure of its tandem RNA Recognition Motifs (RRMs) bound to AU-rich RNA. The structure reveals how an NYAYUNN element is recognized in agreement with recent genome-wide studies. RRM1 acts as a main binding platform, including unusual helical and -hairpin extensions to the canonical RRM fold. -
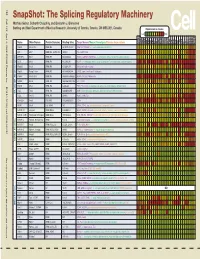
Snapshot: the Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J
192 Cell SnapShot: The Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J. Blencowe 133 Banting and Best Department of Medical Research, University of Toronto, Toronto, ON M5S 3E1, Canada Expression in mouse , April4, 2008©2008Elsevier Inc. Low High Name Other Names Protein Domains Binding Sites Target Genes/Mouse Phenotypes/Disease Associations Amy Ceb Hip Hyp OB Eye SC BM Bo Ht SM Epd Kd Liv Lu Pan Pla Pro Sto Spl Thy Thd Te Ut Ov E6.5 E8.5 E10.5 SRp20 Sfrs3, X16 RRM, RS GCUCCUCUUC SRp20, CT/CGRP; −/− early embryonic lethal E3.5 9G8 Sfrs7 RRM, RS, C2HC Znf (GAC)n Tau, GnRH, 9G8 ASF/SF2 Sfrs1 RRM, RS RGAAGAAC HipK3, CaMKIIδ, HIV RNAs; −/− embryonic lethal, cond. KO cardiomyopathy SC35 Sfrs2 RRM, RS UGCUGUU AChE; −/− embryonic lethal, cond. KO deficient T-cell maturation, cardiomyopathy; LS SRp30c Sfrs9 RRM, RS CUGGAUU Glucocorticoid receptor SRp38 Fusip1, Nssr RRM, RS ACAAAGACAA CREB, type II and type XI collagens SRp40 Sfrs5, HRS RRM, RS AGGAGAAGGGA HipK3, PKCβ-II, Fibronectin SRp55 Sfrs6 RRM, RS GGCAGCACCUG cTnT, CD44 DOI 10.1016/j.cell.2008.03.010 SRp75 Sfrs4 RRM, RS GAAGGA FN1, E1A, CD45; overexpression enhances chondrogenic differentiation Tra2α Tra2a RRM, RS GAAARGARR GnRH; overexpression promotes RA-induced neural differentiation SR and SR-Related Proteins Tra2β Sfrs10 RRM, RS (GAA)n HipK3, SMN, Tau SRm160 Srrm1 RS, PWI AUGAAGAGGA CD44 SWAP Sfrs8 RS, SWAP ND SWAP, CD45, Tau; possible asthma susceptibility gene hnRNP A1 Hnrnpa1 RRM, RGG UAGGGA/U HipK3, SMN2, c-H-ras; rheumatoid arthritis, systemic lupus -

1 Inventory of Supplemental Information Presented in Relation To
Inventory of Supplemental Information presented in relation to each of the main figures in the manuscript 1. Figure S1. Related to Figure 1. 2. Figure S2. Related to Figure 2. 3. Figure S3. Related to Figure 3. 4. Figure S4. Related to Figure 4. 5. Figure S5. Related to Figure 5. 6. Figure S6. Related to Figure 6. 7. Figure S7. Related to Figure 7. 8. Table S1. Related to Figures 1 and S1. 9. Table S2. Related to Figures 3, 4, 6 and 7. 10. Table S3. Related to Figure 3. Supplemental Experimental Procedures 1. Patients and samples 2. Cell culture 3. Long term culture-initiating cells (LTC-IC) assay 4. Differentiation of CD34+ cord blood cells 5. Bi-phasic erythroid differentiation assay 6. Clonal assay 7. Mutation and sequencing analysis 8. Bisulphite sequencing and quantitative pyrosequencing 9. Human SNP genotyping 10. Strand-specific cDNA synthesis and PCR 11. Chromatin Immuno-precipitation (ChIP) 12. Immunoprecipitation and western blot 13. Colony genotyping 14. shRNA generation and viral infection 15. Retrovirus generation and transduction 16. Study approval 17. Statistical Methods 18. Primers list Supplementary References (19) 1 Additional SGK2 families 2 3 4 ♂ ♀ ♂ ♀ ♂ ♀ C/G C/C C/C C/G C/C C/G ♀ ♀ ♂ ♀ C/G C/G C/G C/G T T C T G C T A G A T T C T C C T A G A T T C T C C T A G A T T C T C C T A G A T-Cells T-Cells T-Cells T-Cells Additional GDAP1L1 familiy 2 ♂ ♀ G/G A/A ♀ G/A A C A C G G T G Erythroblasts Figure S1 - . -

Gene and Protein Expression Profiling of Human Ovarian Cancer Cells Treated with the Heat Shock Protein 90 Inhibitor 17-Allylamino-17-Demethoxygeldanamycin
Research Article Gene and Protein Expression Profiling of Human Ovarian Cancer Cells Treated with the Heat Shock Protein 90 Inhibitor 17-Allylamino-17-Demethoxygeldanamycin Alison Maloney,1 Paul A. Clarke,1 Soren Naaby-Hansen,3,4 Rob Stein,3,5 Jens-Oliver Koopman,3,4 Akunna Akpan,3,4 Alice Yang,3,4 Marketa Zvelebil,3,4 Rainer Cramer,3,4 Lindsay Stimson,1 Wynne Aherne,1 Udai Banerji,1,2 Ian Judson,1,2 Swee Sharp,1 Marissa Powers,1 Emmanuel deBilly,1 Joanne Salmons,1 Michael Walton,1 Al Burlingame,3,4 Michael Waterfield,3,4 and Paul Workman1 1Haddow Laboratories, Cancer Research UK Centre for Cancer Therapeutics, The Institute of Cancer Research; 2Royal Marsden NHS Foundation Trust, Sutton, Surrey, United Kingdom; 3Ludwig Institute for Cancer Research and Departments of 4Biochemistry and Molecular Biology and 5Oncology, University College London, London, United Kingdom Abstract anticancer agents and provide a means of obtaining a detailed The promising antitumor activity of 17-allylamino-17-deme- molecular signature of drug action (1, 2). In addition, these thoxygeldanamycin (17AAG) results from inhibition of the methods may identify pharmacodynamic markers that can be used molecular chaperone heat shock protein 90(HSP90)and to evaluate drugs in clinical trials. Gene expression microarrays are subsequent degradation of multiple oncogenic client proteins. increasingly used to investigate the molecular responses to cancer Gene expression microarray and proteomic analysis were used drugs in tumor cells (1). Although valuable, analysis of gene to profile molecular changes in the A2780human ovarian expression at the mRNA level alone cannot adequately predict cancer cell line treated with 17AAG. -

Identification of the RNA Recognition Element of the RBPMS Family of RNA-Binding Proteins and Their Transcriptome-Wide Mrna Targets
Downloaded from rnajournal.cshlp.org on October 1, 2021 - Published by Cold Spring Harbor Laboratory Press Identification of the RNA recognition element of the RBPMS family of RNA-binding proteins and their transcriptome-wide mRNA targets THALIA A. FARAZI,1,5 CARL S. LEONHARDT,1,5 NEELANJAN MUKHERJEE,2 ALEKSANDRA MIHAILOVIC,1 SONG LI,3 KLAAS E.A. MAX,1 CINDY MEYER,1 MASASHI YAMAJI,1 PAVOL CEKAN,1 NICHOLAS C. JACOBS,2 STEFANIE GERSTBERGER,1 CLAUDIA BOGNANNI,1 ERIK LARSSON,4 UWE OHLER,2 and THOMAS TUSCHL1,6 1Laboratory of RNA Molecular Biology, Howard Hughes Medical Institute, The Rockefeller University, New York, New York 10065, USA 2Berlin Institute for Medical Systems Biology, Max Delbrück Center for Molecular Medicine, 13125 Berlin, Germany 3Biology Department, Duke University, Durham, North Carolina 27708, USA 4Institute of Biomedicine, The Sahlgrenska Academy, University of Gothenburg, Gothenburg, SE-405 30, Sweden ABSTRACT Recent studies implicated the RNA-binding protein with multiple splicing (RBPMS) family of proteins in oocyte, retinal ganglion cell, heart, and gastrointestinal smooth muscle development. These RNA-binding proteins contain a single RNA recognition motif (RRM), and their targets and molecular function have not yet been identified. We defined transcriptome-wide RNA targets using photoactivatable-ribonucleoside-enhanced crosslinking and immunoprecipitation (PAR-CLIP) in HEK293 cells, revealing exonic mature and intronic pre-mRNA binding sites, in agreement with the nuclear and cytoplasmic localization of the proteins. Computational and biochemical approaches defined the RNA recognition element (RRE) as a tandem CAC trinucleotide motif separated by a variable spacer region. Similar to other mRNA-binding proteins, RBPMS family of proteins relocalized to cytoplasmic stress granules under oxidative stress conditions suggestive of a support function for mRNA localization in large and/or multinucleated cells where it is preferentially expressed. -

Aneuploidy: Using Genetic Instability to Preserve a Haploid Genome?
Health Science Campus FINAL APPROVAL OF DISSERTATION Doctor of Philosophy in Biomedical Science (Cancer Biology) Aneuploidy: Using genetic instability to preserve a haploid genome? Submitted by: Ramona Ramdath In partial fulfillment of the requirements for the degree of Doctor of Philosophy in Biomedical Science Examination Committee Signature/Date Major Advisor: David Allison, M.D., Ph.D. Academic James Trempe, Ph.D. Advisory Committee: David Giovanucci, Ph.D. Randall Ruch, Ph.D. Ronald Mellgren, Ph.D. Senior Associate Dean College of Graduate Studies Michael S. Bisesi, Ph.D. Date of Defense: April 10, 2009 Aneuploidy: Using genetic instability to preserve a haploid genome? Ramona Ramdath University of Toledo, Health Science Campus 2009 Dedication I dedicate this dissertation to my grandfather who died of lung cancer two years ago, but who always instilled in us the value and importance of education. And to my mom and sister, both of whom have been pillars of support and stimulating conversations. To my sister, Rehanna, especially- I hope this inspires you to achieve all that you want to in life, academically and otherwise. ii Acknowledgements As we go through these academic journeys, there are so many along the way that make an impact not only on our work, but on our lives as well, and I would like to say a heartfelt thank you to all of those people: My Committee members- Dr. James Trempe, Dr. David Giovanucchi, Dr. Ronald Mellgren and Dr. Randall Ruch for their guidance, suggestions, support and confidence in me. My major advisor- Dr. David Allison, for his constructive criticism and positive reinforcement. -

A Novel Resveratrol Analog: Its Cell Cycle Inhibitory, Pro-Apoptotic and Anti-Inflammatory Activities on Human Tumor Cells
A NOVEL RESVERATROL ANALOG : ITS CELL CYCLE INHIBITORY, PRO-APOPTOTIC AND ANTI-INFLAMMATORY ACTIVITIES ON HUMAN TUMOR CELLS A dissertation submitted to Kent State University in partial fulfillment of the requirements for the degree of Doctor of Philosophy by Boren Lin May 2006 Dissertation written by Boren Lin B.S., Tunghai University, 1996 M.S., Kent State University, 2003 Ph. D., Kent State University, 2006 Approved by Dr. Chun-che Tsai , Chair, Doctoral Dissertation Committee Dr. Bryan R. G. Williams , Co-chair, Doctoral Dissertation Committee Dr. Johnnie W. Baker , Members, Doctoral Dissertation Committee Dr. James L. Blank , Dr. Bansidhar Datta , Dr. Gail C. Fraizer , Accepted by Dr. Robert V. Dorman , Director, School of Biomedical Sciences Dr. John R. Stalvey , Dean, College of Arts and Sciences ii TABLE OF CONTENTS LIST OF FIGURES……………………………………………………………….………v LIST OF TABLES……………………………………………………………………….vii ACKNOWLEDGEMENTS….………………………………………………………….viii I INTRODUCTION….………………………………………………….1 Background and Significance……………………………………………………..1 Specific Aims………………………………………………………………………12 II MATERIALS AND METHODS.…………………………………………….16 Cell Culture and Compounds…….……………….…………………………….….16 MTT Cell Viability Assay………………………………………………………….16 Trypan Blue Exclusive Assay……………………………………………………...18 Flow Cytometry for Cell Cycle Analysis……………..……………....……………19 DNA Fragmentation Assay……………………………………………...…………23 Caspase-3 Activity Assay………………………………...……….….…….………24 Annexin V-FITC Staining Assay…………………………………..…...….………28 NF-kappa B p65 Activity Assay……………………………………..………….…29 -

Binding Specificities of Human RNA Binding Proteins Towards Structured
bioRxiv preprint doi: https://doi.org/10.1101/317909; this version posted March 1, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 1 Binding specificities of human RNA binding proteins towards structured and linear 2 RNA sequences 3 4 Arttu Jolma1,#, Jilin Zhang1,#, Estefania Mondragón4,#, Teemu Kivioja2, Yimeng Yin1, 5 Fangjie Zhu1, Quaid Morris5,6,7,8, Timothy R. Hughes5,6, Louis James Maher III4 and Jussi 6 Taipale1,2,3,* 7 8 9 AUTHOR AFFILIATIONS 10 11 1Department of Medical Biochemistry and Biophysics, Karolinska Institutet, Solna, Sweden 12 2Genome-Scale Biology Program, University of Helsinki, Helsinki, Finland 13 3Department of Biochemistry, University of Cambridge, Cambridge, United Kingdom 14 4Department of Biochemistry and Molecular Biology and Mayo Clinic Graduate School of 15 Biomedical Sciences, Mayo Clinic College of Medicine and Science, Rochester, USA 16 5Department of Molecular Genetics, University of Toronto, Toronto, Canada 17 6Donnelly Centre, University of Toronto, Toronto, Canada 18 7Edward S Rogers Sr Department of Electrical and Computer Engineering, University of 19 Toronto, Toronto, Canada 20 8Department of Computer Science, University of Toronto, Toronto, Canada 21 #Authors contributed equally 22 *Correspondence: [email protected] 23 24 25 SUMMARY 26 27 Sequence specific RNA-binding proteins (RBPs) control many important 28 processes affecting gene expression. They regulate RNA metabolism at multiple 29 levels, by affecting splicing of nascent transcripts, RNA folding, base modification, 30 transport, localization, translation and stability. Despite their central role in most 31 aspects of RNA metabolism and function, most RBP binding specificities remain 32 unknown or incompletely defined.