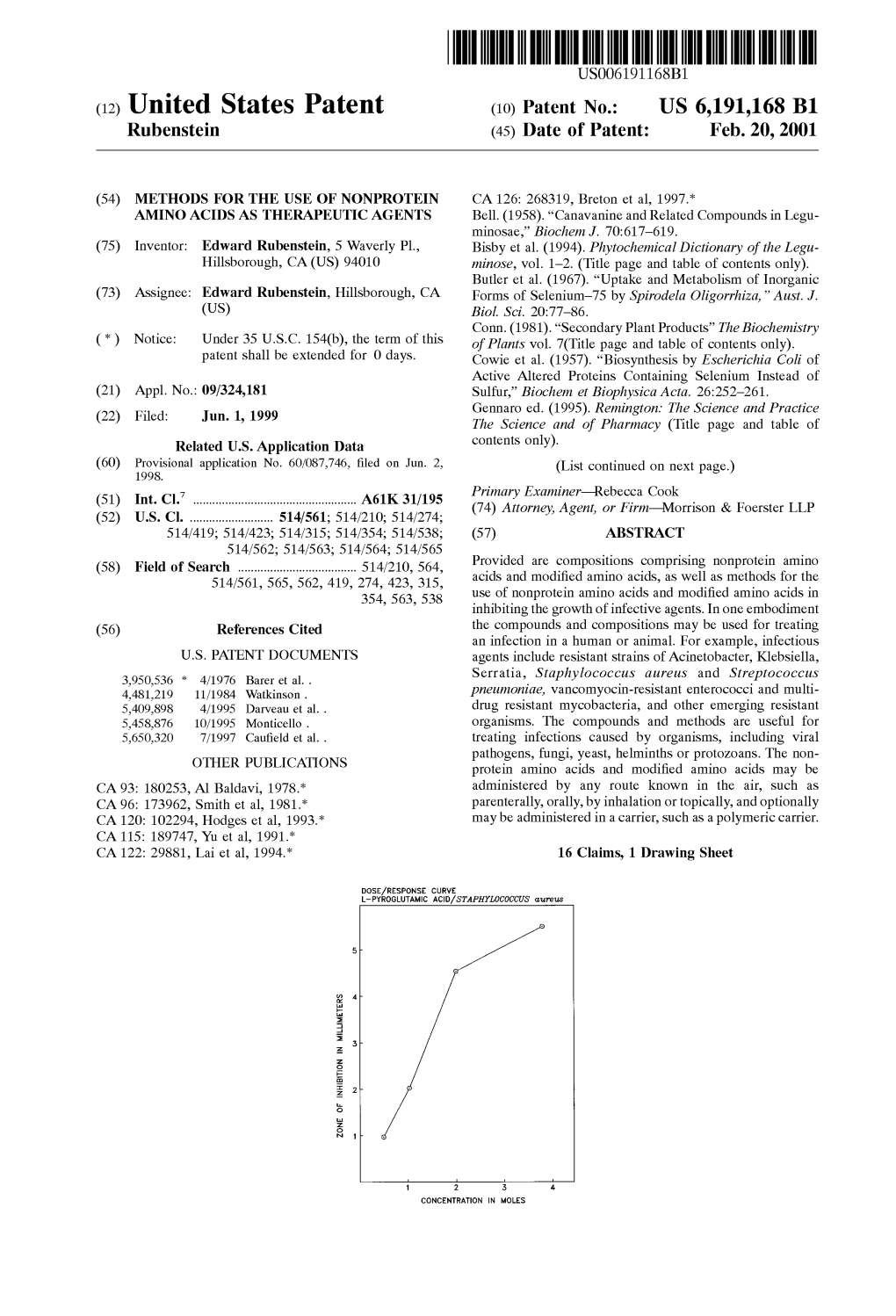
Zone Finhibton Inmillimeters
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Generate Metabolic Map Poster
Authors: Pallavi Subhraveti Ron Caspi Peter Midford Peter D Karp An online version of this diagram is available at BioCyc.org. Biosynthetic pathways are positioned in the left of the cytoplasm, degradative pathways on the right, and reactions not assigned to any pathway are in the far right of the cytoplasm. Transporters and membrane proteins are shown on the membrane. Ingrid Keseler Periplasmic (where appropriate) and extracellular reactions and proteins may also be shown. Pathways are colored according to their cellular function. Gcf_003855395Cyc: Shewanella livingstonensis LMG 19866 Cellular Overview Connections between pathways are omitted for legibility. -

Antiviral Compounds Antivirale Verbindungen Composes Antiviraux
(19) TZZ_Z _T (11) EP 1 778 702 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: C07F 9/141 (2006.01) C07D 207/08 (2006.01) 13.07.2011 Bulletin 2011/28 A61K 31/662 (2006.01) A61P 31/12 (2006.01) (21) Application number: 05791144.8 (86) International application number: PCT/US2005/025503 (22) Date of filing: 18.07.2005 (87) International publication number: WO 2006/020276 (23.02.2006 Gazette 2006/08) (54) ANTIVIRAL COMPOUNDS ANTIVIRALE VERBINDUNGEN COMPOSES ANTIVIRAUX (84) Designated Contracting States: (74) Representative: Reitstötter - Kinzebach AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Patentanwälte HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI Sternwartstrasse 4 SK TR 81679 München (DE) Designated Extension States: AL BA HR MK YU (56) References cited: EP-A- 1 337 550 WO-A-00/09543 (30) Priority: 16.07.2004 US 588633 P WO-A-00/59929 WO-A-98/17679 27.07.2004 US 591635 P WO-A-99/07733 WO-A-02/060926 WO-A-03/053349 WO-A-03/064416 (43) Date of publication of application: WO-A-03/064455 WO-A-03/064456 02.05.2007 Bulletin 2007/18 WO-A-03/066103 WO-A-03/099274 WO-A-03/099316 US-A1- 2003 186 895 (60) Divisional application: US-A1- 2003 224 977 US-A1- 2004 048 802 10178084.9 / 2 316 839 US-B1- 6 608 027 (73) Proprietor: GILEAD SCIENCES, INC. -

Purification and Characterization of the Higher Plant Enzyme L-Canaline Reductase (L-Canavanine Catabolism/Plant Nitrogen Metabolism/Leguminosae) GERALD A
Proc. Natd. Acad. Sci. USA Vol. 89, pp. 1780-1784, March 1992 Biochemistry Purification and characterization of the higher plant enzyme L-canaline reductase (L-canavanine catabolism/plant nitrogen metabolism/Leguminosae) GERALD A. ROSENTHAL T. H. Morgan School of Biological Sciences, University of Kentucky, Lexington, KY 40506 Communicated by John S. Boyer, December 3, 1991 (receivedfor review April 20, 1991) ABSTRACT A newly discovered enzyme, L-canaline re- oglutaric acid to generate stoichiometrically a canaline-2- ductase (NADPH:L-canaline oxidoreductase, EC 1.6.6.-), has oxoglutaric acid oxime (5). Canaline also reacts readily with been isolated and purified from 10-day-old leaves of the jack the pyridoxal phosphate moiety of vitamin B6-containing bean Canavalia ensiformis (Leguminosae). This higher plant is enzymes to form a stable, covalently linked complex (6, 7). representative of a large number of legumes that synthesize In vitro analysis of canaline interaction with homogeneous L-canavanine, an important nitrogen-storing nonprotein amino ornithine aminotransferase (ornithine-oxo-acid aminotrans- acid. Canavanine-storing legumes contain arginase, which ferase; L-ornithine:2-oxo-acid aminotransferase, EC hydrolyzes L-canavanine to form the toxic metabolite L-cana- 2.6.1.13) reveals its marked ability to form an oxime complex line. Canaline reductase, having a mass of =167 kDa and with and thereby inhibit this pyridoxal phosphate-dependent composed of 82-kDa dimers, catalyzes a NADPH-dependent enzyme (8). As little as 10 nM canaline causes a significant reductive cleavage of L-canaline to L-homoserine and ammo- reduction in ornithine aminotransferase activity (9). nia. This is the only enzyme known to use reduced NADP to Canavanine-storing legumes can accumulate high levels of cleave an O-N bond. -

(12) United States Patent (10) Patent No.: US 7,776,844 B2 Yu Et Al
USOO777684.4B2 (12) United States Patent (10) Patent No.: US 7,776,844 B2 Yu et al. (45) Date of Patent: Aug. 17, 2010 (54) N-(PHOSPHONOALKYL)-AMINO ACIDS, 7,429,575 B2 9/2008 Yu et al. DERVATIVES THEREOF AND 2005, 016491.6 A1 7/2005 Leadbetter et al. COMPOSITIONS AND METHODS OF USE OTHER PUBLICATIONS (76) Inventors: Ruey J. Yu, 655 Stump Rd., Chalfont, Struninet al., 1989, CAS. 111:233525.* PA (US)18914; Eugene J. Van Scott, 3 Shi et al., 2004, CAS;142. 231739.* Hidden La., Abington, PA (US) 19001 Li et al., 2005, CAS; 143: 158351.* Sandeman et al., 2002, CAS: 137:274431.* (*) Notice: Subject to any disclaimer, the term of this Single co's i. 164: patent is extended or adjusted under 35 Stahl e et et al., al., 1995, CAS:si. 124:563.10.* U.S.C. 154(b) by 0 days. Jezowska-Bojczuk et al., 1994, CAS: 120:28.1638.* Strumin et al., 1989, CAS: 111:194894.* (21) Appl. No.: 12/428,906 Balthazor et al., 1987, CAS: 106:50457.* The Merck Index. "An Encyclopedia of Chemicals, Drugs, and (22) Filed: Apr. 23, 2009 Biologicals.” (2001) p. 1768, (2 pgs.), 13th Edition, O'Neil et al. (Ed.), Merck & Co., Inc., Whitehouse Station, N.J. (65) Prior Publication Data Wester et al., 1991, CAS: 114:242443. US 2009/0208499 A1 Aug. 20, 2009 * cited by examiner Related U.S. Application Data Primary Examiner Rei-tsang Shiao - - - (74) Attorney, Agent, or Firm Panitch Schwarze Belisario & (62) Division of application No. 12/194203, filed on Aug. -

| Mo Naman Attituunika Mitatti
|MO NAMAN ATTITUUNIKAUS009962450B2 MITATTI (12 ) United States Patent (10 ) Patent No. : US 9 , 962 , 450 B2 Kraynov et al. ( 45) Date of Patent : May 8 , 2018 ( 54 ) METHOD OF TREATING HEART FAILURE 4 ,659 ,839 A 4 / 1987 Nicolotti et al . WITH MODIFIED RELAXIN POLYPEPTIDES 4 ,670 ,417 A 6 / 1987 Iwasaki et al. 4 ,671 , 958 A 6 / 1987 Rodwell et al. 4 ,680 , 338 A 7 / 1987 Sundoro ( 71) Applicant : AMBRX , INC . , La Jolla , CA (US ) 4 ,689 ,406 A 8 / 1987 Banks et al . 4 ,699 ,784 A 10 / 1987 Shih et al . ( 72 ) Inventors : Vadim Kraynov , San Diego , CA (US ) ; 4 ,738 , 921 A 4 / 1988 Belagaje et al. Nick Knudsen , San Diego , CA (US ) ; 4 ,755 ,465 A 7 / 1988 Gray et al. 4 ,837 , 148 A 6 / 1989 Cregg Amha Hewet, Chula Vista , CA (US ) ; 4 ,859 ,600 A 8 / 1989 Gray et al . Kristine De Dios, San Diego , CA (US ) ; 4 , 876 , 197 A 10 / 1989 Burke et al. Jason Pinkstaff , Encinitas, CA (US ) ; 4 , 880 ,734 A 11/ 1989 Burke et al . Lorraine Sullivan , San Diego , CA 4 , 902 ,502 A 2 / 1990 Nitecki et al . 4 , 904 , 584 A 2 / 1990 Shaw ( US ) 4 ,929 , 555 A 5 / 1990 Cregg et al. 5 ,021 , 234 A 6 / 1991 Ehrenfeld (73 ) Assignee : AMBRX , INC. , La Jolla , CA (US ) 5 ,089 ,398 A 2 / 1992 Rosenberg et al . 5 , 122 ,614 A 6 / 1992 Zalipsky ( * ) Notice : Subject to any disclaimer , the term of this 5 , 145 , 962 A 9 / 1992 Hudson et al. -

Download Product Insert (PDF)
PRODUCT INFORMATION L-Canaline Item No. 9002357 CAS Registry No.: 496-93-5 Formal Name: O-amino-L-homoserine O MF: C H N O 4 10 2 3 O FW: 134.1 HO NH2 Purity: ≥95% NH Stability: ≥2 years at -20°C 2 Supplied as: A crystalline solid Laboratory Procedures For long term storage, we suggest that L-canaline be stored as supplied at -20°C. It should be stable for at least two years. L-Canaline is supplied as a crystalline solid. A stock solution may be made by dissolving the L-canaline in the solvent of choice. L-Canaline is soluble in organic solvents such as DMSO which should be purged with an inert gas. The solubility of L-canaline in this solvent is approximately 1 mg/ml. Further dilutions of the stock solution into aqueous buffers or isotonic saline should be made prior to performing biological experiments. Ensure that the residual amount of organic solvent is insignificant, since organic solvents may have physiological effects at low concentrations. Organic solvent-free aqueous solutions of L-canaline can be prepared by directly dissolving the crystalline solid in aqueous buffers. The solubility of L-canaline in PBS, pH 7.2, is approximately 10 mg/ml. We do not recommend storing the aqueous solution for more than one day. Description L-Canaline is an aminooxy analog of ornithine that irreversibly inhibits aminotransferases (transaminases), 1-3 including ornithine aminotransferase (Ki = 2 µM). It forms oximes with α-keto acids and aldehydes, most notably with pyridoxal phosphate, an essential cofactor of aminotransferases.3 L-Canaline is naturally found in plants, including legumes, and is involved in the metabolism of L-canavanine, an aminooxy analog of arginine.4 It is cytotoxic to a range of organisms, including bacteria, insects, and parasites.2,4,5 References 1. -

(12) United States Patent (10) Patent No.: US 9,452.222 B2 Kraynov Et Al
USOO9452222B2 (12) United States Patent (10) Patent No.: US 9,452.222 B2 Kraynov et al. (45) Date of Patent: Sep. 27, 2016 (54) NUCLEIC ACIDS ENCODING MODIFIED 4,680,338 A 7, 1987 Sundoro RELAXN POLYPEPTIDES 4,689,406 A 8, 1987 Banks et al. 4,699,784. A 10, 1987 Shih et al. 4,738,921 A 4, 1988 Belagaje et al. (71) Applicant: Ambrx, Inc., La Jolla, CA (US) 4,755.465 A 7/1988 Gray et al. 4,837,148 A 6/1989 Cregg (72) Inventors: Vadim Kraynov, San Diego, CA (US); 4,859,600 A 8/1989 Gray et al. Nick Knudsen, San Diego, CA (US); 4,876, 197 A 10, 1989 Burke et al. 4,880,734 A 11/1989 Burke et al. Ahma Hewet, Chula Vista, CA (US); 4.902,502 A 2f1990 Nitecki et al. Kristine De Dios, San Diego, CA (US); 4,904,584 A 2, 1990 Shaw Jason Pinkstaff, Encinitas, CA (US); 4,929,555 A 5/1990 Cregg et al. Lorraine Sullivan, San Diego, CA 5,021,234 A 6, 1991 Ehrenfeld (US) 5,089,398 A 2/1992 Rosenberg et al. 5,122,614 A 6/1992 Zalipsky 5,145,962 A 9, 1992 Hudson et al. (73) Assignee: AMBRX, INC., La Jolla, CA (US) 5,162,601 A 11/1992 Slightom 5,219,564 A 6/1993 Zalipsky et al. (*) Notice: Subject to any disclaimer, the term of this 5,229,490 A 7, 1993 Tam patent is extended or adjusted under 35 5,231,178 A 7, 1993 Holtz et al. -

United States Patent Office Patented Nov
3,704,246 United States Patent Office Patented Nov. 28, 1972 2 (I) R 3,704,246 AMNO ACD DERVATIVES ( CH).--N Miklos Bodanszky, Shaker Heights, Ohio, assignor to R2-C YoH-R E. R. Squibb & Sons, Inc., New York, N.Y. No Drawing. Continuation-in-part of application Ser. No. R-i-o-c6 798,790, Feb. 12, 1969, which is a continuation-in-part wherein R is the residue of an a-amino acid, R1 is hydro of abandoned application Ser. No. 451,609, Apr. 28, gen, lower alkyl, hydroxy-substituted lower alkyl, or 1965. This application Dec. 16, 1970, Ser. No. 98,924 lower alkyl lower alkoxy; R and R3 each is hydrogen, int. C. C07d 87/54 lower alkyl, hydroxy-substituted lower alkyl, lower alkyl U.S. C. 260-333 9 Claims 10 lower alkoxy, or R and R3 together with the carbons to which they are joined form a phenyl ring which may be unsubstituted or substituted by halogen or nitro, and m ABSTRACT OF THE DESCLOSURE is an integer from 0 to 3. New lactone intermediates useful in the synthesis of 5 DETAILED DESCRIPTION OF THE INVENTION peptides are prepared by reacting an a-amino acid with Any a-amino acid may be treated according to this an active carbonyl compound which forms a Schiff's base. invention. This includes the naturally occurring ox-amino The latter is treated with a condensing agent so that acids see for example, Hackh's Chemical Dictionary, cyclization occurs and a lactone is formed from which a 3rd ed. -

(12) Patent Application Publication (10) Pub. No.: US 2007/0161543 A1 Yu Et Al
US 2007 O161543A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2007/0161543 A1 Yu et al. (43) Pub. Date: Jul. 12, 2007 (54) N-(PHOSPHONOALKYL)-AMINO ACIDS, (52) U.S. Cl. .............................. 514/7: 514/114: 530/331; DERVATIVES THEREOF AND 558/170 COMPOSITIONS AND METHODS OF USE (76) Inventors: Ruey J. Yu, Chalfont, PA (US); Eugene J. Van Scott, Abington, PA (US) (57) ABSTRACT Correspondence Address: AKN GUMPSTRAUSS HAUER & FELD The present invention relates to an N-(phosphonoalkyl)- 6. COMMERCE SQUARE amino acid, a related compound or a derivative thereof, the 2005 MARKET STREET, SUITE 2200 N-(phosphonoalkyl)-amino acid, related compound or PHILADELPHIA, PA 19103 (US) derivative thereof being in a form as a free acid, salt, partial salt, lactone, amide or ester, or in Stereoisomeric or non (21) Appl. No.: 11/621,287 Stereoisomeric form, other than N-(phosphonomethyl)-gly (22) Filed: Jan. 9, 2007 cine or N.N-bis(phosphonomethyl)-glycine. Also included is a composition including an N-(phosphonoalkyl)-amino acid, Related U.S. Application Data a related compound or a derivative thereof in a form as a free (60) Provisional application No. 60/757,614, filed on Jan. acid, salt, partial salt, lactone, amide or ester, or in Stereoi 10, 2006. sy u. Is Someric or non-stereoisomeric form, and a cosmetically or pharmaceutically acceptable vehicle for topical or systemic Publication Classification administration to a mammalian Subject, as well as a method of administering an effective amount of Such a composition (51) Int. Cl. A6 IK 38/05 (2006.01) for alleviating or improving a condition, disorder, symptom A6 IK 3/66 (2006.01) or syndrome associated with at least one of a nervous, C07K 5/10 (2006.01) vascular, musculoskeletal or cutaneous system. -
Generated by SRI International Pathway Tools Version 25.0, Authors S
Authors: Pallavi Subhraveti Ingrid Keseler Quang Ong Ron Caspi An online version of this diagram is available at BioCyc.org. Biosynthetic pathways are positioned in the left of the cytoplasm, degradative pathways on the right, and reactions not assigned to any pathway are in the far right of the cytoplasm. Transporters and membrane proteins are shown on the membrane. Tim Holland Peter D Karp Periplasmic (where appropriate) and extracellular reactions and proteins may also be shown. Pathways are colored according to their cellular function. Saur904776Cyc: Staphylococcus aureus aureus IS-24 Cellular Overview Connections between pathways are omitted for legibility. Anamika Kothari guaninehypoxanthine indole- pro 3-acetate (S)-lactate (S)-lactate uracil H + H + H + H + H + H + H + H + H + ammonium IS24_0964 IS24_2784 AtpC AtpF AtpI AtpA AtpD AtpE AtpB AtpH AtpG Amt PbuG PutP PyrP IS24_1600 + + + + + + + + + indole- H H H H H H H H H uracil (S)-lactate (S)-lactate ammonium 3-acetate guanine pro hypoxanthine Cofactor, Prosthetic Group, Electron Carrier, and Vitamin Biosynthesis Chemoautotrophic Amino Acid Degradation Energy Metabolism Detoxification UDP-N-acetyl- an [apo BCAA an apo-[methylmalonyl- campest- a (3R)-3- 3-(4- valine degradation I a [formate C testosterone a phosphate cob(II)yrinate a,c- glutamine SAM a reduced flavodoxin α-D-muramoyl- di-trans,octa-cis 4-methyl-2- dehydrogenase CoA:pyruvate biotin 5-hydroxytryptophol β hydroxyhexanoyl- hydroxyphenyl) flavin biosynthesis I (bacteria and plants) L-ascorbate biosynthesis V L-ascorbate -
Pka Data Compiled by R
pKa Data Compiled by R. Williams page-1 pKa Values INDEX Inorganic 2 Phenazine 24 Phosphates 3 Pyridine 25 Carboxylic acids 4, 8 Pyrazine 26 Aliphatic 4, 8 Aromatic 7, 8 Quinoline 27 Phenols 9 Quinazoline 27 Alcohols and oxygen acids 10, 11 Quinoxaline 27 Amino Acids 12 Special Nitrogen Compounds 28 Peptides 13 Hydroxylamines 28 Nitrogen Compounds 14 Hydrazines 28 Aliphatic amines 15, 17, 19 Semicarbazones 28 Cyanoamines 16 Amidoximes 28 Anilines 17, 18, 20 Thiols 29 Nucleosides 21 Carbon Acids 30,31 Special Table Heterocycles 22 Indicators 31 Acridine 23 References 32-34 Benzoquinoline 24 Cinnoline 23 Hydantoin 24 Imidazole 24 For complex chelating agents, see also reference 77. Note. This document was compiled by W.P. Jencks and has been added to by F.H. Westheimer pKa Data Compiled by R. Williams page-2 ACIDS Compound pK Ref. H3PO2 2.0, 2.23* 28 H2PO4– 7.21* 77 AgOH 3.96 4 HPO4_ 12.32* 77 Al(OH)3 11.2 28 H3PO3 2.0 28 As(OH)3 9.22 28 H PO – 6.58* 77 H3AsO4 2.22, 7.0, 13.0 28 2 3 H4P2O7 1.52* 77 H2AsO4– 6.98* 77 H P O – 2.36* 77 HAsO4* 11.53* 77 3 2 7 = As2O3 0 4 H2P2O7 6.60* 77 H3AsO3 9.22* HP2O7= 9.25* 77 H3BO3 9.23* 28 HReO4 -1.25 30 H2B4O7 4.00 34 HSCN 4.00 34 HB4O7 9.00 34 H2SeO3 2.6, 8.3, 2.62* 28 Be(OH)2 3.7 4 HSeO3 8.32 77 HBr -9.00 31 H2SeO4 Strong, 2.0 28 HOBr 8.7 28 HSeO4 2.00 34 HOCl 7.53, 7.46 28, 33 H3SiO3 10.0 34 HClO2 2.0 28 H2SO3 1.9, 7.0, 1.76* 28, 77 HClO3 -1.00 28 H2SO4 -3.0, 1.9 28 HClO4 (70%) -10.00 31 HSO3 7.21* 77 CH SO H -0.6 31 3 3 HSO4– 1.99* 77 HCN 9.40 34 H2S2O4 1.9 29 H CO 6.37, 6.35*, 3.58 34, 32 2 3 H2Se -

Thermochemistry of Amino Acids and Constrained Diamines
W&M ScholarWorks Dissertations, Theses, and Masters Projects Theses, Dissertations, & Master Projects 2012 Thermochemistry of Amino Acids and Constrained Diamines Erica Jane Tullo College of William & Mary - Arts & Sciences Follow this and additional works at: https://scholarworks.wm.edu/etd Part of the Analytical Chemistry Commons, Biochemistry Commons, and the Physical Chemistry Commons Recommended Citation Tullo, Erica Jane, "Thermochemistry of Amino Acids and Constrained Diamines" (2012). Dissertations, Theses, and Masters Projects. Paper 1539623596. https://dx.doi.org/doi:10.21220/s2-vp4j-1g27 This Dissertation is brought to you for free and open access by the Theses, Dissertations, & Master Projects at W&M ScholarWorks. It has been accepted for inclusion in Dissertations, Theses, and Masters Projects by an authorized administrator of W&M ScholarWorks. For more information, please contact [email protected]. Thermochemistry of Amino Acids and Constrained Diamines Erica Jane Tullo Oxford, Pennsylvania Master of Science, The College of William and Mary, 2006 Bachelor of Science, Washington College, 2000 A Dissertation presented to the Graduate Faculty of the College of William and Mary in Candidacy for the Degree of Doctor of Philosophy Department of Applied Science The College of William and Mary January, 2012 APPROVAL PAGE This Dissertation is submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy Committee Chair Marga t Hamilton Professor John C. Poutsma, Chemistry The College of William and Mary Professor Mark K. Hinders, C~r. Applied Science The College of William and Mary 2JtlUJJ Z1C- Assistant Professor Hannes C. Schniepp, Applied Science The College of William and Mary a~ ~L/b/ Professor David E."k:ranbuehl, cfiemistry The College of William and Mary ABSTRACT PAGE The gas-phase proton affinities of several highly basic amino acids and diamines were determined using the extended kinetic method in an ESI-quadrupole ion trap instrument.