Thermochemistry of Amino Acids and Constrained Diamines
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

NINDS Custom Collection II
ACACETIN ACEBUTOLOL HYDROCHLORIDE ACECLIDINE HYDROCHLORIDE ACEMETACIN ACETAMINOPHEN ACETAMINOSALOL ACETANILIDE ACETARSOL ACETAZOLAMIDE ACETOHYDROXAMIC ACID ACETRIAZOIC ACID ACETYL TYROSINE ETHYL ESTER ACETYLCARNITINE ACETYLCHOLINE ACETYLCYSTEINE ACETYLGLUCOSAMINE ACETYLGLUTAMIC ACID ACETYL-L-LEUCINE ACETYLPHENYLALANINE ACETYLSEROTONIN ACETYLTRYPTOPHAN ACEXAMIC ACID ACIVICIN ACLACINOMYCIN A1 ACONITINE ACRIFLAVINIUM HYDROCHLORIDE ACRISORCIN ACTINONIN ACYCLOVIR ADENOSINE PHOSPHATE ADENOSINE ADRENALINE BITARTRATE AESCULIN AJMALINE AKLAVINE HYDROCHLORIDE ALANYL-dl-LEUCINE ALANYL-dl-PHENYLALANINE ALAPROCLATE ALBENDAZOLE ALBUTEROL ALEXIDINE HYDROCHLORIDE ALLANTOIN ALLOPURINOL ALMOTRIPTAN ALOIN ALPRENOLOL ALTRETAMINE ALVERINE CITRATE AMANTADINE HYDROCHLORIDE AMBROXOL HYDROCHLORIDE AMCINONIDE AMIKACIN SULFATE AMILORIDE HYDROCHLORIDE 3-AMINOBENZAMIDE gamma-AMINOBUTYRIC ACID AMINOCAPROIC ACID N- (2-AMINOETHYL)-4-CHLOROBENZAMIDE (RO-16-6491) AMINOGLUTETHIMIDE AMINOHIPPURIC ACID AMINOHYDROXYBUTYRIC ACID AMINOLEVULINIC ACID HYDROCHLORIDE AMINOPHENAZONE 3-AMINOPROPANESULPHONIC ACID AMINOPYRIDINE 9-AMINO-1,2,3,4-TETRAHYDROACRIDINE HYDROCHLORIDE AMINOTHIAZOLE AMIODARONE HYDROCHLORIDE AMIPRILOSE AMITRIPTYLINE HYDROCHLORIDE AMLODIPINE BESYLATE AMODIAQUINE DIHYDROCHLORIDE AMOXEPINE AMOXICILLIN AMPICILLIN SODIUM AMPROLIUM AMRINONE AMYGDALIN ANABASAMINE HYDROCHLORIDE ANABASINE HYDROCHLORIDE ANCITABINE HYDROCHLORIDE ANDROSTERONE SODIUM SULFATE ANIRACETAM ANISINDIONE ANISODAMINE ANISOMYCIN ANTAZOLINE PHOSPHATE ANTHRALIN ANTIMYCIN A (A1 shown) ANTIPYRINE APHYLLIC -

NON-HAZARDOUS CHEMICALS May Be Disposed of Via Sanitary Sewer Or Solid Waste
NON-HAZARDOUS CHEMICALS May Be Disposed Of Via Sanitary Sewer or Solid Waste (+)-A-TOCOPHEROL ACID SUCCINATE (+,-)-VERAPAMIL, HYDROCHLORIDE 1-AMINOANTHRAQUINONE 1-AMINO-1-CYCLOHEXANECARBOXYLIC ACID 1-BROMOOCTADECANE 1-CARBOXYNAPHTHALENE 1-DECENE 1-HYDROXYANTHRAQUINONE 1-METHYL-4-PHENYL-1,2,5,6-TETRAHYDROPYRIDINE HYDROCHLORIDE 1-NONENE 1-TETRADECENE 1-THIO-B-D-GLUCOSE 1-TRIDECENE 1-UNDECENE 2-ACETAMIDO-1-AZIDO-1,2-DIDEOXY-B-D-GLYCOPYRANOSE 2-ACETAMIDOACRYLIC ACID 2-AMINO-4-CHLOROBENZOTHIAZOLE 2-AMINO-2-(HYDROXY METHYL)-1,3-PROPONEDIOL 2-AMINOBENZOTHIAZOLE 2-AMINOIMIDAZOLE 2-AMINO-5-METHYLBENZENESULFONIC ACID 2-AMINOPURINE 2-ANILINOETHANOL 2-BUTENE-1,4-DIOL 2-CHLOROBENZYLALCOHOL 2-DEOXYCYTIDINE 5-MONOPHOSPHATE 2-DEOXY-D-GLUCOSE 2-DEOXY-D-RIBOSE 2'-DEOXYURIDINE 2'-DEOXYURIDINE 5'-MONOPHOSPHATE 2-HYDROETHYL ACETATE 2-HYDROXY-4-(METHYLTHIO)BUTYRIC ACID 2-METHYLFLUORENE 2-METHYL-2-THIOPSEUDOUREA SULFATE 2-MORPHOLINOETHANESULFONIC ACID 2-NAPHTHOIC ACID 2-OXYGLUTARIC ACID 2-PHENYLPROPIONIC ACID 2-PYRIDINEALDOXIME METHIODIDE 2-STEP CHEMISTRY STEP 1 PART D 2-STEP CHEMISTRY STEP 2 PART A 2-THIOLHISTIDINE 2-THIOPHENECARBOXYLIC ACID 2-THIOPHENECARBOXYLIC HYDRAZIDE 3-ACETYLINDOLE 3-AMINO-1,2,4-TRIAZINE 3-AMINO-L-TYROSINE DIHYDROCHLORIDE MONOHYDRATE 3-CARBETHOXY-2-PIPERIDONE 3-CHLOROCYCLOBUTANONE SOLUTION 3-CHLORO-2-NITROBENZOIC ACID 3-(DIETHYLAMINO)-7-[[P-(DIMETHYLAMINO)PHENYL]AZO]-5-PHENAZINIUM CHLORIDE 3-HYDROXYTROSINE 1 9/26/2005 NON-HAZARDOUS CHEMICALS May Be Disposed Of Via Sanitary Sewer or Solid Waste 3-HYDROXYTYRAMINE HYDROCHLORIDE 3-METHYL-1-PHENYL-2-PYRAZOLIN-5-ONE -

Effect of L-Canavanine, an Inhibitor of Inducible Nitric Oxide Synthase, on Myocardial Dysfunction During Septic Shock
J Nippon Med Sch 2002; 69(1) 13 ―Original― Effect of L-canavanine, an Inhibitor of Inducible Nitric Oxide Synthase, on Myocardial Dysfunction During Septic Shock Norihito Suzuki, Atsuhiro Sakamoto and Ryo Ogawa Department of Anesthesiology, Nippon Medical School Abstract Overproduction of nitric oxide(NO)by inducible NO synthase(iNOS)plays aroleinthe pathophysiology of septic shock. The depression of cardiac contractilityinsuchsituationsis mediated by proinflammatory cytokines, including interleukin-1β(IL-1β), and tumor necrosis factor-α(TNF-α). The effects of two NOS inhibitors with different isoform selectivity were compared in isolated working rat hearts. The depression of contractility by IL-1β and TNF-α was prevented by administration of a nonselective nitric oxide synthase inhibitor, NG-nitro-L- arginine methyl ester(L-NAME)or an inhibitor of inducible nitric oxide synthase, L-canavanine. In contrast, when L-NAME was administered in the absence of IL-1β and TNF-α, it depressed contractility over the 2h perfusion period by significantly reducing coronary flow. These results support current thinking that the depression of myocardial functionbyIL-1β and TNF-α is mediated, at least in part, by an intracardiac increase in inducible nitric oxide synthase, and that in contrast to L-NAME, the decline in coronary conductance seen in cytokine-treated is not prevented by L-canavanine hearts. L-canavanine shows selective inhibition of inducible nitric oxide synthase unlike the vasopressor action of L-NAME in cytokine-treated hearts. (J Nippon Med Sch 2002; 69: 13―18) Key words: nitric oxide(NO), nitric oxide(NO)synthase, working heart, L-canavanine, NG-nitro-L-arginine methyl ester(L-NAME) We have previously demonstrated that admini- Introduction stration of L-canavanine or L-NAME attenuated the endotoxin-induced hypotension and vascular hypore- Nitric oxide(NO)may be produced within the activity to adrenaline, and the beneficial hemody- heart by either constitutive or inducible NO1,2. -

Nitric Oxide Synthase Inhibitors As Antidepressants
Pharmaceuticals 2010, 3, 273-299; doi:10.3390/ph3010273 OPEN ACCESS pharmaceuticals ISSN 1424-8247 www.mdpi.com/journal/pharmaceuticals Review Nitric Oxide Synthase Inhibitors as Antidepressants Gregers Wegener 1,* and Vallo Volke 2 1 Centre for Psychiatric Research, University of Aarhus, Skovagervej 2, DK-8240 Risskov, Denmark 2 Department of Physiology, University of Tartu, Ravila 19, EE-70111 Tartu, Estonia; E-Mail: [email protected] (V.V.) * Author to whom correspondence should be addressed; E-Mail: [email protected]; Tel.: +4577893524; Fax: +4577893549. Received: 10 November 2009; in revised form: 7 January 2010 / Accepted: 19 January 2010 / Published: 20 January 2010 Abstract: Affective and anxiety disorders are widely distributed disorders with severe social and economic effects. Evidence is emphatic that effective treatment helps to restore function and quality of life. Due to the action of most modern antidepressant drugs, serotonergic mechanisms have traditionally been suggested to play major roles in the pathophysiology of mood and stress-related disorders. However, a few clinical and several pre-clinical studies, strongly suggest involvement of the nitric oxide (NO) signaling pathway in these disorders. Moreover, several of the conventional neurotransmitters, including serotonin, glutamate and GABA, are intimately regulated by NO, and distinct classes of antidepressants have been found to modulate the hippocampal NO level in vivo. The NO system is therefore a potential target for antidepressant and anxiolytic drug action in acute therapy as well as in prophylaxis. This paper reviews the effect of drugs modulating NO synthesis in anxiety and depression. Keywords: nitric oxide; antidepressants; psychiatry; depression; anxiety 1. Introduction Recent data from Denmark and Europe [1,2], indicate that brain disorders account for 12% of all direct costs in the Danish health system and 9% of the total drug consumption was used for treatment of brain diseases. -
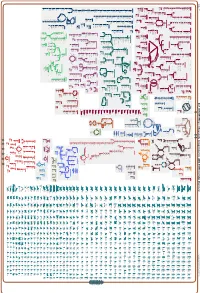
Generate Metabolic Map Poster
Authors: Pallavi Subhraveti Ron Caspi Peter Midford Peter D Karp An online version of this diagram is available at BioCyc.org. Biosynthetic pathways are positioned in the left of the cytoplasm, degradative pathways on the right, and reactions not assigned to any pathway are in the far right of the cytoplasm. Transporters and membrane proteins are shown on the membrane. Ingrid Keseler Periplasmic (where appropriate) and extracellular reactions and proteins may also be shown. Pathways are colored according to their cellular function. Gcf_003855395Cyc: Shewanella livingstonensis LMG 19866 Cellular Overview Connections between pathways are omitted for legibility. -

Antiviral Compounds Antivirale Verbindungen Composes Antiviraux
(19) TZZ_Z _T (11) EP 1 778 702 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: C07F 9/141 (2006.01) C07D 207/08 (2006.01) 13.07.2011 Bulletin 2011/28 A61K 31/662 (2006.01) A61P 31/12 (2006.01) (21) Application number: 05791144.8 (86) International application number: PCT/US2005/025503 (22) Date of filing: 18.07.2005 (87) International publication number: WO 2006/020276 (23.02.2006 Gazette 2006/08) (54) ANTIVIRAL COMPOUNDS ANTIVIRALE VERBINDUNGEN COMPOSES ANTIVIRAUX (84) Designated Contracting States: (74) Representative: Reitstötter - Kinzebach AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Patentanwälte HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI Sternwartstrasse 4 SK TR 81679 München (DE) Designated Extension States: AL BA HR MK YU (56) References cited: EP-A- 1 337 550 WO-A-00/09543 (30) Priority: 16.07.2004 US 588633 P WO-A-00/59929 WO-A-98/17679 27.07.2004 US 591635 P WO-A-99/07733 WO-A-02/060926 WO-A-03/053349 WO-A-03/064416 (43) Date of publication of application: WO-A-03/064455 WO-A-03/064456 02.05.2007 Bulletin 2007/18 WO-A-03/066103 WO-A-03/099274 WO-A-03/099316 US-A1- 2003 186 895 (60) Divisional application: US-A1- 2003 224 977 US-A1- 2004 048 802 10178084.9 / 2 316 839 US-B1- 6 608 027 (73) Proprietor: GILEAD SCIENCES, INC. -

Purification and Characterization of the Higher Plant Enzyme L-Canaline Reductase (L-Canavanine Catabolism/Plant Nitrogen Metabolism/Leguminosae) GERALD A
Proc. Natd. Acad. Sci. USA Vol. 89, pp. 1780-1784, March 1992 Biochemistry Purification and characterization of the higher plant enzyme L-canaline reductase (L-canavanine catabolism/plant nitrogen metabolism/Leguminosae) GERALD A. ROSENTHAL T. H. Morgan School of Biological Sciences, University of Kentucky, Lexington, KY 40506 Communicated by John S. Boyer, December 3, 1991 (receivedfor review April 20, 1991) ABSTRACT A newly discovered enzyme, L-canaline re- oglutaric acid to generate stoichiometrically a canaline-2- ductase (NADPH:L-canaline oxidoreductase, EC 1.6.6.-), has oxoglutaric acid oxime (5). Canaline also reacts readily with been isolated and purified from 10-day-old leaves of the jack the pyridoxal phosphate moiety of vitamin B6-containing bean Canavalia ensiformis (Leguminosae). This higher plant is enzymes to form a stable, covalently linked complex (6, 7). representative of a large number of legumes that synthesize In vitro analysis of canaline interaction with homogeneous L-canavanine, an important nitrogen-storing nonprotein amino ornithine aminotransferase (ornithine-oxo-acid aminotrans- acid. Canavanine-storing legumes contain arginase, which ferase; L-ornithine:2-oxo-acid aminotransferase, EC hydrolyzes L-canavanine to form the toxic metabolite L-cana- 2.6.1.13) reveals its marked ability to form an oxime complex line. Canaline reductase, having a mass of =167 kDa and with and thereby inhibit this pyridoxal phosphate-dependent composed of 82-kDa dimers, catalyzes a NADPH-dependent enzyme (8). As little as 10 nM canaline causes a significant reductive cleavage of L-canaline to L-homoserine and ammo- reduction in ornithine aminotransferase activity (9). nia. This is the only enzyme known to use reduced NADP to Canavanine-storing legumes can accumulate high levels of cleave an O-N bond. -

(12) United States Patent (10) Patent No.: US 7,776,844 B2 Yu Et Al
USOO777684.4B2 (12) United States Patent (10) Patent No.: US 7,776,844 B2 Yu et al. (45) Date of Patent: Aug. 17, 2010 (54) N-(PHOSPHONOALKYL)-AMINO ACIDS, 7,429,575 B2 9/2008 Yu et al. DERVATIVES THEREOF AND 2005, 016491.6 A1 7/2005 Leadbetter et al. COMPOSITIONS AND METHODS OF USE OTHER PUBLICATIONS (76) Inventors: Ruey J. Yu, 655 Stump Rd., Chalfont, Struninet al., 1989, CAS. 111:233525.* PA (US)18914; Eugene J. Van Scott, 3 Shi et al., 2004, CAS;142. 231739.* Hidden La., Abington, PA (US) 19001 Li et al., 2005, CAS; 143: 158351.* Sandeman et al., 2002, CAS: 137:274431.* (*) Notice: Subject to any disclaimer, the term of this Single co's i. 164: patent is extended or adjusted under 35 Stahl e et et al., al., 1995, CAS:si. 124:563.10.* U.S.C. 154(b) by 0 days. Jezowska-Bojczuk et al., 1994, CAS: 120:28.1638.* Strumin et al., 1989, CAS: 111:194894.* (21) Appl. No.: 12/428,906 Balthazor et al., 1987, CAS: 106:50457.* The Merck Index. "An Encyclopedia of Chemicals, Drugs, and (22) Filed: Apr. 23, 2009 Biologicals.” (2001) p. 1768, (2 pgs.), 13th Edition, O'Neil et al. (Ed.), Merck & Co., Inc., Whitehouse Station, N.J. (65) Prior Publication Data Wester et al., 1991, CAS: 114:242443. US 2009/0208499 A1 Aug. 20, 2009 * cited by examiner Related U.S. Application Data Primary Examiner Rei-tsang Shiao - - - (74) Attorney, Agent, or Firm Panitch Schwarze Belisario & (62) Division of application No. 12/194203, filed on Aug. -

| Mo Naman Attituunika Mitatti
|MO NAMAN ATTITUUNIKAUS009962450B2 MITATTI (12 ) United States Patent (10 ) Patent No. : US 9 , 962 , 450 B2 Kraynov et al. ( 45) Date of Patent : May 8 , 2018 ( 54 ) METHOD OF TREATING HEART FAILURE 4 ,659 ,839 A 4 / 1987 Nicolotti et al . WITH MODIFIED RELAXIN POLYPEPTIDES 4 ,670 ,417 A 6 / 1987 Iwasaki et al. 4 ,671 , 958 A 6 / 1987 Rodwell et al. 4 ,680 , 338 A 7 / 1987 Sundoro ( 71) Applicant : AMBRX , INC . , La Jolla , CA (US ) 4 ,689 ,406 A 8 / 1987 Banks et al . 4 ,699 ,784 A 10 / 1987 Shih et al . ( 72 ) Inventors : Vadim Kraynov , San Diego , CA (US ) ; 4 ,738 , 921 A 4 / 1988 Belagaje et al. Nick Knudsen , San Diego , CA (US ) ; 4 ,755 ,465 A 7 / 1988 Gray et al. 4 ,837 , 148 A 6 / 1989 Cregg Amha Hewet, Chula Vista , CA (US ) ; 4 ,859 ,600 A 8 / 1989 Gray et al . Kristine De Dios, San Diego , CA (US ) ; 4 , 876 , 197 A 10 / 1989 Burke et al. Jason Pinkstaff , Encinitas, CA (US ) ; 4 , 880 ,734 A 11/ 1989 Burke et al . Lorraine Sullivan , San Diego , CA 4 , 902 ,502 A 2 / 1990 Nitecki et al . 4 , 904 , 584 A 2 / 1990 Shaw ( US ) 4 ,929 , 555 A 5 / 1990 Cregg et al. 5 ,021 , 234 A 6 / 1991 Ehrenfeld (73 ) Assignee : AMBRX , INC. , La Jolla , CA (US ) 5 ,089 ,398 A 2 / 1992 Rosenberg et al . 5 , 122 ,614 A 6 / 1992 Zalipsky ( * ) Notice : Subject to any disclaimer , the term of this 5 , 145 , 962 A 9 / 1992 Hudson et al. -

Zone Finhibton Inmillimeters
US006191168B1 (12) United States Patent (10) Patent N0.: US 6,191,168 B1 Rubenstein (45) Date of Patent: Feb. 20, 2001 (54) METHODS FOR THE USE OF NONPROTEIN CA 126: 268319, Breton et al, 1997.* AMINO ACIDS AS THERAPEUTIC AGENTS Bell. (1958). “Canavanine and Related Compounds in Legu minosae,” Biochem J. 70:617—619. (75) Inventor: Edward Rubenstein, 5 Waverly Pl., Bisby et al. (1994). Phytochemical Dictionary of the Legu Hillsborough, CA (US) 94010 minose, vol. 1—2. (Title page and table of contents only). Butler et al. (1967). “Uptake and Metabolism of Inorganic (73) Assignee: Edward Rubenstein, Hillsborough, CA Forms of Selenium—75 by Spiroa'ela Oligorrhiza, ” Aust. J. (Us) Biol. Sci. 20:77—86. Conn. (1981). “Secondary Plant Products” The Biochemistry (*) Notice: Under 35 U.S.C. 154(b), the term of this of Plants vol. 7(Title page and table of contents only). patent shall be extended for 0 days. CoWie et al. (1957). “Biosynthesis by Escherichia Coli of Active Altered Proteins Containing Selenium Instead of (21) Appl. No.: 09/324,181 Sulfur,” Biochem et Biophysica Acta. 26:252—261. Gennaro ed. (1995). Remington." The Science and Practice Filed: Jun. 1, 1999 (22) The Science and of Pharmacy (Title page and table of Related US. Application Data contents only). (60) Provisional application No. 60/087,746, ?led on Jun. 2, (List continued on neXt page.) 1998. Primary Examiner—Rebecca Cook (51) Int. Cl.7 ................................................. .. A61K 31/195 (74) Attorney, Agent, or Firm—Morrison & Foerster LLP (52) US. Cl. ........................ .. 514/561; 514/210; 514/274; 514/419; 514/423; 514/315; 514/354; 514/538; (57) ABSTRACT 514/562; 514/563; 514/564; 514/565 Provided are compositions comprising nonprotein amino (58) Field of Search .................................. -

Download Product Insert (PDF)
PRODUCT INFORMATION L-Canaline Item No. 9002357 CAS Registry No.: 496-93-5 Formal Name: O-amino-L-homoserine O MF: C H N O 4 10 2 3 O FW: 134.1 HO NH2 Purity: ≥95% NH Stability: ≥2 years at -20°C 2 Supplied as: A crystalline solid Laboratory Procedures For long term storage, we suggest that L-canaline be stored as supplied at -20°C. It should be stable for at least two years. L-Canaline is supplied as a crystalline solid. A stock solution may be made by dissolving the L-canaline in the solvent of choice. L-Canaline is soluble in organic solvents such as DMSO which should be purged with an inert gas. The solubility of L-canaline in this solvent is approximately 1 mg/ml. Further dilutions of the stock solution into aqueous buffers or isotonic saline should be made prior to performing biological experiments. Ensure that the residual amount of organic solvent is insignificant, since organic solvents may have physiological effects at low concentrations. Organic solvent-free aqueous solutions of L-canaline can be prepared by directly dissolving the crystalline solid in aqueous buffers. The solubility of L-canaline in PBS, pH 7.2, is approximately 10 mg/ml. We do not recommend storing the aqueous solution for more than one day. Description L-Canaline is an aminooxy analog of ornithine that irreversibly inhibits aminotransferases (transaminases), 1-3 including ornithine aminotransferase (Ki = 2 µM). It forms oximes with α-keto acids and aldehydes, most notably with pyridoxal phosphate, an essential cofactor of aminotransferases.3 L-Canaline is naturally found in plants, including legumes, and is involved in the metabolism of L-canavanine, an aminooxy analog of arginine.4 It is cytotoxic to a range of organisms, including bacteria, insects, and parasites.2,4,5 References 1. -

Maintenance of Renal Vascular Reactivity Contributes to Acute Renal Failure During Endotoxemic Shock
Maintenance of Renal Vascular Reactivity Contributes to Acute Renal Failure during Endotoxemic Shock Jean-Jacques Boffa and William J. Arendshorst Department of Cell and Molecular Physiology, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina Septic shock is characterized by hypotension and decreased systemic vascular resistance and impaired vascular reactivity. Renal vasoconstriction markedly contrasts with sepsis-induced generalized systemic vasodilation, which is strongly depen- dent on nitric oxide. Whether maintained renal vascular reactivity to vasoconstrictors contributes to the decrease in renal blood flow (RBF) and GFR observed during LPS-induced sepsis was tested by assessment of the acute effects of pressor agents on mean arterial pressure (MAP) and renal hemodynamics in endotoxemic and control mice. LPS-injected mice displayed lower MAP, RBF, and GFR than controls (P < 0.001). Despite a lower MAP, basal renal vascular resistance (RVR) was higher during endotoxemia (P < 0.02). Angiotensin II infusion produced a weaker MAP response in septic mice (24 versus 37%; P < 0.005), suggesting impaired vasoconstriction and hyporeactivity. A similar MAP increase was observed between groups during G norepinephrine (NE) infusion. The MAP increase to nitric oxide synthase inhibition by N -nitro-L-arginine methyl ester indicating a strong influence of nitric oxide in ,(0.01 ؍ L-NAME) was much greater in LPS-treated mice (41 versus 15%, P) sepsis. In contrast, the RBF and RVR responses to angiotensin II, NE, or L-NAME were similar in both groups. Moreover, vasopressin produced greater changes in MAP, RBF, and RVR in septic mice than in controls. Among the vasoconstrictor challenges, only NE ameliorated the decrease in GFR 14 h after LPS injection.