Matter Reference 1. Atom – 2. Proton – Subatomic Particles with a Positive Charge 3. Neutron
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
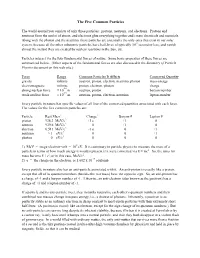
The Five Common Particles
The Five Common Particles The world around you consists of only three particles: protons, neutrons, and electrons. Protons and neutrons form the nuclei of atoms, and electrons glue everything together and create chemicals and materials. Along with the photon and the neutrino, these particles are essentially the only ones that exist in our solar system, because all the other subatomic particles have half-lives of typically 10-9 second or less, and vanish almost the instant they are created by nuclear reactions in the Sun, etc. Particles interact via the four fundamental forces of nature. Some basic properties of these forces are summarized below. (Other aspects of the fundamental forces are also discussed in the Summary of Particle Physics document on this web site.) Force Range Common Particles It Affects Conserved Quantity gravity infinite neutron, proton, electron, neutrino, photon mass-energy electromagnetic infinite proton, electron, photon charge -14 strong nuclear force ≈ 10 m neutron, proton baryon number -15 weak nuclear force ≈ 10 m neutron, proton, electron, neutrino lepton number Every particle in nature has specific values of all four of the conserved quantities associated with each force. The values for the five common particles are: Particle Rest Mass1 Charge2 Baryon # Lepton # proton 938.3 MeV/c2 +1 e +1 0 neutron 939.6 MeV/c2 0 +1 0 electron 0.511 MeV/c2 -1 e 0 +1 neutrino ≈ 1 eV/c2 0 0 +1 photon 0 eV/c2 0 0 0 1) MeV = mega-electron-volt = 106 eV. It is customary in particle physics to measure the mass of a particle in terms of how much energy it would represent if it were converted via E = mc2. -
Proton Therapy ACKNOWLEDGEMENTS
AMERICAN BRAIN TUMOR ASSOCIATION Proton Therapy ACKNOWLEDGEMENTS ABOUT THE AMERICAN BRAIN TUMOR ASSOCIATION Founded in 1973, the American Brain Tumor Association (ABTA) was the first national nonprofit organization dedicated solely to brain tumor research. For over 40 years, the Chicago-based ABTA has been providing comprehensive resources that support the complex needs of brain tumor patients and caregivers, as well as the critical funding of research in the pursuit of breakthroughs in brain tumor diagnosis, treatment and care. To learn more about the ABTA, visit www.abta.org. We gratefully acknowledge Anita Mahajan, Director of International Development, MD Anderson Proton Therapy Center, director, Pediatric Radiation Oncology, co-section head of Pediatric and CNS Radiation Oncology, The University of Texas MD Anderson Cancer Center; Kevin S. Oh, MD, Department of Radiation Oncology, Massachusetts General Hospital; and Sridhar Nimmagadda, PhD, assistant professor of Radiology, Medicine and Oncology, Johns Hopkins University, for their review of this edition of this publication. This publication is not intended as a substitute for professional medical advice and does not provide advice on treatments or conditions for individual patients. All health and treatment decisions must be made in consultation with your physician(s), utilizing your specific medical information. Inclusion in this publication is not a recommendation of any product, treatment, physician or hospital. COPYRIGHT © 2015 ABTA REPRODUCTION WITHOUT PRIOR WRITTEN PERMISSION IS PROHIBITED AMERICAN BRAIN TUMOR ASSOCIATION Proton Therapy INTRODUCTION Brain tumors are highly variable in their treatment and prognosis. Many are benign and treated conservatively, while others are malignant and require aggressive combinations of surgery, radiation and chemotherapy. -
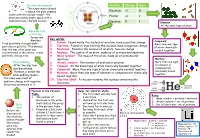
Key Words: 1. Proton: Found Inside the Nucleus of an Atom, Have a Positive Charge 2. Electron: Found in Rings Orbiting the Nucle
Nucleus development Particle Charge Mass This experiment allowed Rutherford to replace the plum pudding Electron -1 0 model with the nuclear model – the Proton +1 1 atom was mainly empty space with a small positively charged nucleus Neutron 0 1 Element All the same type of atom Alpha particle scattering Geiger and Key words: Marsden 1. Proton: Found inside the nucleus of an atom, have a positive charge Compound fired positively-charged alpha More than one type 2. Electron: Found in rings orbiting the nucleus, have a negative charge particles at gold foil. This showed of atom chemically that the mas of an atom was 3. Neutrons: Found in the nucleus of an atom, have no charge bonded together concentrated in the centre, it was 4. Nucleus: The centre of an atom, made up of protons and neutrons positively charged too 5. Mass number: The mass of the atom, made up of protons and neutrons Mixture Plum pudding 6. Atomic number: The number of protons in an atom More than one type After the electron 7. Element: All the same type of atom chemically bonded together of element or was discovered, 8. Compound: More than one type of atom chemically bonded together compound not chemically bound Thomson created the 9. Mixture: More than one type of element or compound not chemically together plum pudding model – bound together the atom was a ball of 10. Electron Shell: A ring surrounding the nucleus containing the positive charge with negative electrons electrons scattered in it Position in the Periodic Rules for electron shells: Table: 1. -

Detection of a Strange Particle
10 extraordinary papers Within days, Watson and Crick had built a identify the full set of codons was completed in forensics, and research into more-futuristic new model of DNA from metal parts. Wilkins by 1966, with Har Gobind Khorana contributing applications, such as DNA-based computing, immediately accepted that it was correct. It the sequences of bases in several codons from is well advanced. was agreed between the two groups that they his experiments with synthetic polynucleotides Paradoxically, Watson and Crick’s iconic would publish three papers simultaneously in (see go.nature.com/2hebk3k). structure has also made it possible to recog- Nature, with the King’s researchers comment- With Fred Sanger and colleagues’ publica- nize the shortcomings of the central dogma, ing on the fit of Watson and Crick’s structure tion16 of an efficient method for sequencing with the discovery of small RNAs that can reg- to the experimental data, and Franklin and DNA in 1977, the way was open for the com- ulate gene expression, and of environmental Gosling publishing Photograph 51 for the plete reading of the genetic information in factors that induce heritable epigenetic first time7,8. any species. The task was completed for the change. No doubt, the concept of the double The Cambridge pair acknowledged in their human genome by 2003, another milestone helix will continue to underpin discoveries in paper that they knew of “the general nature in the history of DNA. biology for decades to come. of the unpublished experimental results and Watson devoted most of the rest of his ideas” of the King’s workers, but it wasn’t until career to education and scientific administra- Georgina Ferry is a science writer based in The Double Helix, Watson’s explosive account tion as head of the Cold Spring Harbor Labo- Oxford, UK. -

8.1 the Neutron-To-Proton Ratio
M. Pettini: Introduction to Cosmology | Lecture 8 PRIMORDIAL NUCLEOSYNTHESIS Our discussion at the end of the previous lecture concentrated on the rela- tivistic components of the Universe, photons and leptons. Baryons (which are non-relativistic) did not figure because they make a trifling contribu- tion to the energy density. However, from t ∼ 1 s a number of nuclear reactions involving baryons took place. The end result of these reactions was to lock up most of the free neutrons into 4He nuclei and to create trace amounts of D, 3He, 7Li and 7Be. This is Primordial Nucleosynthesis, also referred to as Big Bang Nucleosynthesis (BBN). 8.1 The Neutron-to-Proton Ratio Before neutrino decoupling at around 1 MeV, neutrons and protons are kept in mutual thermal equilibrium through charged-current weak interactions: + n + e ! p +ν ¯e − p + e ! n + νe (8.1) − n ! p + e +ν ¯e While equilibrium persists, the relative number densities of neutrons and protons are given by a Boltzmann factor based on their mass difference: n ∆m c2 1:5 = exp − = exp − (8.2) p eq kT T10 where 10 ∆m = (mn − mp) = 1:29 MeV = 1:5 × 10 K (8.3) 10 and T10 is the temperature in units of 10 K. As discussed in Lecture 7.1.2, this equilibrium will be maintained so long as the timescale for the weak interactions is short compared with the timescale of the cosmic expansion (which increases the mean distance between parti- cles). In 7.2.1 we saw that the ratio of the two rates varies approximately as: Γ T 3 ' : (8.4) H 1:6 × 1010 K 1 This steep dependence on temperature can be appreciated as follows. -

Charm and Strange Quark Contributions to the Proton Structure
Report series ISSN 0284 - 2769 of SE9900247 THE SVEDBERG LABORATORY and \ DEPARTMENT OF RADIATION SCIENCES UPPSALA UNIVERSITY Box 533, S-75121 Uppsala, Sweden http://www.tsl.uu.se/ TSL/ISV-99-0204 February 1999 Charm and Strange Quark Contributions to the Proton Structure Kristel Torokoff1 Dept. of Radiation Sciences, Uppsala University, Box 535, S-751 21 Uppsala, Sweden Abstract: The possibility to have charm and strange quarks as quantum mechanical fluc- tuations in the proton wave function is investigated based on a model for non-perturbative QCD dynamics. Both hadron and parton basis are examined. A scheme for energy fluctu- ations is constructed and compared with explicit energy-momentum conservation. Resulting momentum distributions for charniand_strange quarks in the proton are derived at the start- ing scale Qo f°r the perturbative QCD evolution. Kinematical constraints are found to be important when comparing to the "intrinsic charm" model. Master of Science Thesis Linkoping University Supervisor: Gunnar Ingelman, Uppsala University 1 kuldsepp@tsl .uu.se 30-37 Contents 1 Introduction 1 2 Standard Model 3 2.1 Introductory QCD 4 2.2 Light-cone variables 5 3 Experiments 7 3.1 The HERA machine 7 3.2 Deep Inelastic Scattering 8 4 Theory 11 4.1 The Parton model 11 4.2 The structure functions 12 4.3 Perturbative QCD corrections 13 4.4 The DGLAP equations 14 5 The Edin-Ingelman Model 15 6 Heavy Quarks in the Proton Wave Function 19 6.1 Extrinsic charm 19 6.2 Intrinsic charm 20 6.3 Hadronisation 22 6.4 The El-model applied to heavy quarks -
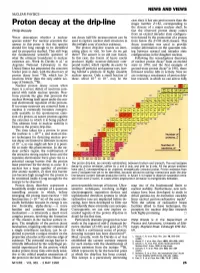
Proton Decay at the Drip-Line Magic Number Z = 82, Corresponding to the Closure of a Major Nuclear Shell
NEWS AND VIEWS NUCLEAR PHYSICS-------------------------------- case since it has one proton more than the Proton decay at the drip-line magic number Z = 82, corresponding to the closure of a major nuclear shell. In Philip Woods fact the observed proton decay comes from an excited intruder state configura WHAT determines whether a nuclear ton decay half-life measurements can be tion formed by the promotion of a proton species exists? For nuclear scientists the used to explore nuclear shell structures in from below the Z=82 shell closure. This answer to this poser is that the species this twilight zone of nuclear existence. decay transition was used to provide should live long enough to be identified The proton drip-line sounds an inter unique information on the quantum mix and its properties studied. This still begs esting place to visit. So how do we get ing between normal and intruder state the fundamental scientific question of there? The answer is an old one: fusion. configurations in the daughter nucleus. what the ultimate boundaries to nuclear In this case, the fusion of heavy nuclei Following the serendipitous discovery existence are. Work by Davids et al. 1 at produces highly neutron-deficient com of nuclear proton decay4 from an excited Argonne National Laboratory in the pound nuclei, which rapidly de-excite by state in 1970, and the first example of United States has pinpointed the remotest boiling off particles and gamma-rays, leav ground-state proton decay5 in 1981, there border post to date, with the discovery of ing behind a plethora of highly unstable followed relative lulls in activity. -

Chapter 8. the Atomic Nucleus
Chapter 8. The Atomic Nucleus Notes: • Most of the material in this chapter is taken from Thornton and Rex, Chapters 12 and 13. 8.1 Nuclear Properties Atomic nuclei are composed of protons and neutrons, which are referred to as nucleons. Although both types of particles are not fundamental or elementary, they can still be considered as basic constituents for the purpose of understanding the atomic nucleus. Protons and neutrons have many characteristics in common. For example, their masses are very similar with 1.0072765 u (938.272 MeV) for the proton and 1.0086649 u (939.566 MeV) for the neutron. The symbol ‘u’ stands for the atomic mass unit defined has one twelfth of the mass of the main isotope of carbon (i.e., 12 C ), which is known to contain six protons and six neutrons in its nucleus. We thus have that 1 u = 1.66054 ×10−27 kg (8.1) = 931.49 MeV/c2 . Protons and neutrons also both have the same intrinsic spin, but different magnetic moments (see below). Their main difference, however, pertains to their electrical charges: the proton, as we know, has a charge of +e , while the neutron has none, as its name implies. Atomic nuclei are designated using the symbol A Z XN , (8.2) with Z, N and A the number of protons (atomic element number), the number of neutrons, and the atomic mass number ( A = Z + N ), respectively, while X is the chemical element symbol. It is often the case that Z and N are omitted, when there is no chance of confusion. -

Quarks and Their Discovery
Quarks and Their Discovery Parashu Ram Poudel Department of Physics, PN Campus, Pokhara Email: [email protected] Introduction charge (e) of one proton. The different fl avors of Quarks are the smallest building blocks of matter. quarks have different charges. The up (u), charm They are the fundamental constituents of all the (c) and top (t) quarks have electric charge +2e/3 hadrons. They have fractional electronic charge. and the down (d), strange (s) and bottom (b) quarks Quarks never exist alone in nature. They are always have charge -e/3; -e is the charge of an electron. The found in combination with other quarks or antiquark masses of these quarks vary greatly, and of the six, in larger particle of matter. By studying these larger only the up and down quarks, which are by far the particles, scientists have determined the properties lightest, appear to play a direct role in normal matter. of quarks. Protons and neutrons, the particles that make up the nuclei of the atoms consist of quarks. There are four forces that act between the quarks. Without quarks there would be no atoms, and without They are strong force, electromagnetic force, atoms, matter would not exist as we know it. Quarks weak force and gravitational force. The quantum only form triplets called baryons such as proton and of strong force is gluon. Gluons bind quarks or neutron or doublets called mesons such as Kaons and quark and antiquark together to form hadrons. The pi mesons. Quarks exist in six varieties: up (u), down electromagnetic force has photon as quantum that (d), charm (c), strange (s), bottom (b), and top (t) couples the quarks charge. -

Physics Results from the First Electron-Proton Collider HERA
DESY <>r> -02r, March 1995 KS001829947 R: FI 0EC07443217 ■*DE007443217* Physics Results from the First Electron-Proton Collider HERA A. De Roeck Drutachcs Elektrotten-Synchrotron DESY. Hamburg DISTRIBUTION OF THIS DOCUMENT IS UNLIMITED FOREIGN SALES PROHIBITED ISSN 04IS-9S13 BEST 95-025 ISS35 0418-9833 March 1995 PHYSICS RESULTS FROM THE FIRST ELECTRON-PROTON COLLIDER HERA * Albert De Roeck Deutsches Eiektronen-Synchroiron DESY. Hamburg 1 Introduction On the 31st of May 1992 the first electron-proton (ep) collisions were observed in the HI and ZEUS experiments at the newly commissioned high energy collider HERA, in Hamburg. Ger many. HERA is the first electron-proton coEider in the world: 26.7 GeV electrons collide on 820 GeV protons, yielding an ep centre of mass system (CMS) energy of 296 GeV. Already the results from the first data collected by the experiments have given important new information on the structure of the proton, on interactions of high energetic photons with matter and on searches for exotic particles. These lectures give a summary of the physics results obtained by the Hi and ZEUS experiments using the data collected in 1992 and 1993. Electron-proton, or more general lepton-hadron experiments, have been playing a major role in our understanding of the structure of matter for the last 30 years. At the end of the sixties experiments with electron beams on proton targets performed at the Stanford Lin ear Accelerator revealed that the proton had an internal structure.1 It was suggested that the proton consists of pointlike objects, called partons. 2 These partoas were subsequently identified with quarks which until then were only mathematical objects for the fundamen tal representation of the SU(3) symmetry group, used to explain the observed multiplets in hadron spectroscopy. -

Make an Atom Vocabulary Grade Levels
MAKE AN ATOM Fundamental to physical science is a basic understanding of the atom. Atoms are comprised of protons, neutrons, and electrons. Protons and neutrons are at the center of the atom while electrons live in lobe-shaped clouds outside the nucleus. The number of electrons usually matches the number of protons, yielding a net neutral charge for the atom. Sometimes an atom has less neutrons or more neutrons than protons. This is called an isotope. If an atom has different numbers of electrons than protons, then it is an ion. If an atom has different numbers of protons, it is a different element all together. Scientists at Idaho National Laboratory study, create, and use radioactive isotopes like Uranium 234. The 234 means this isotope has an atomic mass of 234 Atomic Mass Units (AMU). GRADE LEVELS: 3-8 VOCABULARY Atom – The basic unit of a chemical element. Proton – A stable subatomic particle occurring in all atomic nuclei, with a positive electric charge equal in magnitude to that of an electron, but of opposite sign. Neutron – A subatomic particle of about the same mass as a proton but without an electric charge, present in all atomic nuclei except those of ordinary hydrogen. Electron – A stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids. Orbital – Each of the actual or potential patterns of electron density that may be formed on an atom or molecule by one or more electrons. Ion – An atom or molecule with a net electric charge due to the loss or gain of one or more electrons. -

Photon, Neutron, Proton, Electron, Gamma Ray, Carbon Ion)
Fundamental types of radiation particle beams (Photon, Neutron, Proton, Electron, Gamma Ray, Carbon Ion) Photon This is the most common, widely used type of radiation, and is available at most treatment centers and hospitals. This is widely used to treat most cancers including known or suspect residual ACC which was not able to be removed with surgery by itself. In simple terms, it is fundamentally a very high dose X-ray beam. This is also the type of radiation beam utilized for most of the pinpoint targeted types of radiation treatments, such as CyberKnife, TomoTherapy and Novalis. Most hospitals have photon radiation available and the beam is generated by a beam accelerator device that takes up the space of a typical bedroom. Some of the newer manufacturers have developed a more compact beam accelerator, such as the CyberKnife. Neutron Neutron radiation therapy is a unique, high-LET radiation (the particles are accelerated at a very fast rate) with very specific differences in how it affects DNA in ACC cancer cells when compared to standard Photon radiation. Neutrons are produced from a large and expensive particle accelerator called a cyclotron. This high-LET (high linear-energy-transfer) radiation is also referred to as “fast neutron therapy”. Neutrons, pions and heavy ions (such as carbon, neon and argon) deposit more energy along their path than x-rays or gamma rays, thus causing more damage to the cells they hit. The cyclotron accelerator produces protons and then a series of powerful magnets bend and aim the beam to strike a beryllium target, where the interaction produces neutrons.