Leroy 2021 P3 Later Cambrian
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Early Paleozoic Life & Extinctions (Part 1)
NJU Course Extinctions: Past, Present & Future Prof. Norman MacLeod School of Earth Sciences & Engineering, Nanjing University Extinctions: Past, Present & Future Extinctions: Past, Present & Future Course Syllabus (Revised) Section Week Title Introduction 1 Course Introduction, Intro. To Extinction Introduction 2 History of Extinction Studies Introduction 3 Evolution, Fossils, Time & Extinction Precambrian Extinctions 4 Origin of Life & Precambrian Extionctions Paleozoic Extinctions 5 Early Paleozoic World & Extinctions Paleozoic Extinctions 6 Middle Paleozoic World & Extinctions Paleozoic Extinctions 7 Late Paleozoic World & Extinctions Assessment 8 Mid-Term Examination Mesozoic Extinctions 9 Triassic-Jurassic World & Extinctions Mesozoic Extinctions 10 Labor Day Holiday Cenozoic Extinctions 11 Cretaceous World & Extinctions Cenozoic Extinctions 12 Paleogene World & Extinctions Cenozoic Extinctions 13 Neogene World & Extinctions Modern Extinctions 14 Quaternary World & Extinctions Modern Extinctions 15 Modern World: Floras, Faunas & Environment Modern Extinctions 16 Modern World: Habitats & Organisms Assessment 17 Final Examination Early Paleozoic World, Life & Extinctions Norman MacLeod School of Earth Sciences & Engineering, Nanjing University Early Paleozoic World, Life & Extinctions Objectives Understand the structure of the early Paleozoic world in terms of timescales, geography, environ- ments, and organisms. Understand the structure of early Paleozoic extinction events. Understand the major Paleozoic extinction drivers. Understand -
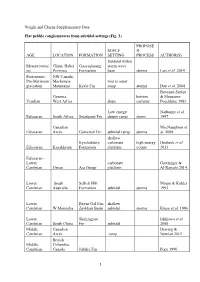
Wright and Cherns Supplementary Data Flat
Wright and Cherns Supplementary Data Flat pebble conglomerates from subtidal settings (Fig. 2) PROPOSE SHELF D AGE LOCATION FORMATION SETTING PROCESS AUTHOR(S) Subtidal within Mesoproteroz China, Hebei Gaoyuzhuang storm wave oic Province Formation base storms Luo et al. 2014 Proterozoic, NW Canada, Pre-Marinoan Mackenzie mid to outer glaciation Mountains Keele Fm ramp storms Day et al. 2004 Bertrand-Sarfati Gourma, bottom & Moussine- Vendian West Africa slope currents' Pouchkine 1983 Low energy Narbonne et al. Ediacaran South Africa Swartpunt Fm deeper ramp storm 1997 Canadian MacNaughton et Ediacaran Arctic Gametrail Fm subtidal ramp storms al. 2008 shallow Kyrshabakty carbonate high energy Heubeck et al. Ediacaran Kazakhstan Formation platform events 2013 Ediacaran - Lower carbonate Grotzinger & Cambrian Oman Ara Group platform Al-Rawahi 2014 Lower South Sellick Hill Mount & Kidder Cambrian Australia Formation subtidal storms 1993 Lower Bayan Gol Fm, shallow Cambrian W Mongolia Zavkhan Basin subtidal storms Kruse et al. 1996 Lower Shuijingtuo Ishikawa et al. Cambrian South China Fm subtidal 2008 Middle Canadian Dewing & Cambrian Arctic ramp Nowlan 2012 British Middle Columbia, Cambrian Canada Jubilee Fm Pope 1990 1 Middle Pratt & Cambrian Argentina La Laja Fm subtidal shelf tsunamis Bordonaro 2007 low energy Middle shallow Cambrian Australia Ranken Lst subtidal storms Kruse 1996 Middle Wyoming, Upper Gros Cambrian USA Ventre Shale Csonka 2009 middle upper Middle W Utah, upper Wheeler, carbonate belt - Cambrian USA Marjum fms subtidal shelf Robison 1964 Middle- Upper Supratidal to Cambrian NW China subtidal fpc storms Liang et al. 1993 Ust’- Brus,Labaz, Middle Orakta, Cambrian - Kulyumbe, Lower Ujgur and Iltyk Kouchinsky et Ordovician Siberia fms al. -

And Ordovician (Sardic) Felsic Magmatic Events in South-Western Europe: Underplating of Hot Mafic Magmas Linked to the Opening of the Rheic Ocean
Solid Earth, 11, 2377–2409, 2020 https://doi.org/10.5194/se-11-2377-2020 © Author(s) 2020. This work is distributed under the Creative Commons Attribution 4.0 License. Comparative geochemical study on Furongian–earliest Ordovician (Toledanian) and Ordovician (Sardic) felsic magmatic events in south-western Europe: underplating of hot mafic magmas linked to the opening of the Rheic Ocean J. Javier Álvaro1, Teresa Sánchez-García2, Claudia Puddu3, Josep Maria Casas4, Alejandro Díez-Montes5, Montserrat Liesa6, and Giacomo Oggiano7 1Instituto de Geociencias (CSIC-UCM), Dr. Severo Ochoa 7, 28040 Madrid, Spain 2Instituto Geológico y Minero de España, Ríos Rosas 23, 28003 Madrid, Spain 3Dpt. Ciencias de la Tierra, Universidad de Zaragoza, 50009 Zaragoza, Spain 4Dpt. de Dinàmica de la Terra i de l’Oceà, Universitat de Barcelona, Martí Franquès s/n, 08028 Barcelona, Spain 5Instituto Geológico y Minero de España, Plaza de la Constitución 1, 37001 Salamanca, Spain 6Dpt. de Mineralogia, Petrologia i Geologia aplicada, Universitat de Barcelona, Martí Franquès s/n, 08028 Barcelona, Spain 7Dipartimento di Scienze della Natura e del Territorio, 07100 Sassari, Italy Correspondence: J. Javier Álvaro ([email protected]) Received: 1 April 2020 – Discussion started: 20 April 2020 Revised: 14 October 2020 – Accepted: 19 October 2020 – Published: 11 December 2020 Abstract. A geochemical comparison of early Palaeo- neither metamorphism nor penetrative deformation; on the zoic felsic magmatic episodes throughout the south- contrary, their unconformities are associated with foliation- western European margin of Gondwana is made and in- free open folds subsequently affected by the Variscan defor- cludes (i) Furongian–Early Ordovician (Toledanian) activ- mation. -

Substantial Vegetation Response to Early Jurassic Global Warming with Impacts on Oceanic Anoxia
ARTICLES https://doi.org/10.1038/s41561-019-0349-z Substantial vegetation response to Early Jurassic global warming with impacts on oceanic anoxia Sam M. Slater 1*, Richard J. Twitchett2, Silvia Danise3 and Vivi Vajda 1 Rapid global warming and oceanic oxygen deficiency during the Early Jurassic Toarcian Oceanic Anoxic Event at around 183 Ma is associated with a major turnover of marine biota linked to volcanic activity. The impact of the event on land-based eco- systems and the processes that led to oceanic anoxia remain poorly understood. Here we present analyses of spore–pollen assemblages from Pliensbachian–Toarcian rock samples that record marked changes on land during the Toarcian Oceanic Anoxic Event. Vegetation shifted from a high-diversity mixture of conifers, seed ferns, wet-adapted ferns and lycophytes to a low-diversity assemblage dominated by cheirolepid conifers, cycads and Cerebropollenites-producers, which were able to sur- vive in warm, drought-like conditions. Despite the rapid recovery of floras after Toarcian global warming, the overall community composition remained notably different after the event. In shelf seas, eutrophication continued throughout the Toarcian event. This is reflected in the overwhelming dominance of algae, which contributed to reduced oxygen conditions and to a marked decline in dinoflagellates. The substantial initial vegetation response across the Pliensbachian/Toarcian boundary compared with the relatively minor marine response highlights that the impacts of the early stages of volcanogenic -

A New Middle Cambrian Trilobite with a Specialized Cephalon from Shandong Province, North China
Editors' choice A new middle Cambrian trilobite with a specialized cephalon from Shandong Province, North China ZHIXIN SUN, HAN ZENG, and FANGCHEN ZHAO Sun, Z., Zeng, H., and Zhao, F. 2020. A new middle Cambrian trilobite with a specialized cephalon from Shandong Province, North China. Acta Palaeontologica Polonica 65 (4): 709–718. Trilobites achieved their maximum generic diversity in the Cambrian, but the peak of morphological disparity of their cranidia occurred in the Middle to Late Ordovician. Early to middle Cambrian trilobites with a specialized cephalon are rare, especially among the ptychoparioids, a group of libristomates featuring the so-called “generalized” bauplan. Here we describe an unusual ptychopariid trilobite Phantaspis auritus gen. et sp. nov. from the middle Cambrian (Miaolingian, Wuliuan) Mantou Formation in the Shandong Province, North China. This new taxon is characterized by a cephalon with an extended anterior area of double-lobate shape resembling a pair of rabbit ears in later ontogenetic stages; a unique type of cephalic specialization that has not been reported from other trilobites. Such a peculiar cephalon as in Phantaspis provides new insights into the variations of cephalic morphology in middle Cambrian trilobites, and may represent a heuristic example of ecological specialization to predation or an improved discoidal enrollment. Key words: Trilobita, Ptychopariida, ontogeny, specialization, Miaolingian, Paleozoic, Longgang, Asia. Zhixin Sun [[email protected]], Han Zeng [[email protected]], and Fangchen Zhao [[email protected]] (cor responding author), State Key Laboratory of Palaeobiology and Stratigraphy, Nanjing Institute of Geology and Palae ontology and Center for Excellence in Life and Palaeoenvironment, Chinese Academy of Sciences, Nanjing 210008, China; University of Chinese Academy of Sciences, Beijing 100049, China. -

Appendix 3.Pdf
A Geoconservation perspective on the trace fossil record associated with the end – Ordovician mass extinction and glaciation in the Welsh Basin Item Type Thesis or dissertation Authors Nicholls, Keith H. Citation Nicholls, K. (2019). A Geoconservation perspective on the trace fossil record associated with the end – Ordovician mass extinction and glaciation in the Welsh Basin. (Doctoral dissertation). University of Chester, United Kingdom. Publisher University of Chester Rights Attribution-NonCommercial-NoDerivatives 4.0 International Download date 26/09/2021 02:37:15 Item License http://creativecommons.org/licenses/by-nc-nd/4.0/ Link to Item http://hdl.handle.net/10034/622234 International Chronostratigraphic Chart v2013/01 Erathem / Era System / Period Quaternary Neogene C e n o z o i c Paleogene Cretaceous M e s o z o i c Jurassic M e s o z o i c Jurassic Triassic Permian Carboniferous P a l Devonian e o z o i c P a l Devonian e o z o i c Silurian Ordovician s a n u a F y r Cambrian a n o i t u l o v E s ' i k s w o Ichnogeneric Diversity k p e 0 10 20 30 40 50 60 70 S 1 3 5 7 9 11 13 15 17 19 21 n 23 r e 25 d 27 o 29 M 31 33 35 37 39 T 41 43 i 45 47 m 49 e 51 53 55 57 59 61 63 65 67 69 71 73 75 77 79 81 83 85 87 89 91 93 Number of Ichnogenera (Treatise Part W) Ichnogeneric Diversity 0 10 20 30 40 50 60 70 1 3 5 7 9 11 13 15 17 19 21 n 23 r e 25 d 27 o 29 M 31 33 35 37 39 T 41 43 i 45 47 m 49 e 51 53 55 57 59 61 c i o 63 z 65 o e 67 a l 69 a 71 P 73 75 77 79 81 83 n 85 a i r 87 b 89 m 91 a 93 C Number of Ichnogenera (Treatise Part W) -

Lee-Riding-2018.Pdf
Earth-Science Reviews 181 (2018) 98–121 Contents lists available at ScienceDirect Earth-Science Reviews journal homepage: www.elsevier.com/locate/earscirev Marine oxygenation, lithistid sponges, and the early history of Paleozoic T skeletal reefs ⁎ Jeong-Hyun Leea, , Robert Ridingb a Department of Geology and Earth Environmental Sciences, Chungnam National University, Daejeon 34134, Republic of Korea b Department of Earth and Planetary Sciences, University of Tennessee, Knoxville, TN 37996, USA ARTICLE INFO ABSTRACT Keywords: Microbial carbonates were major components of early Paleozoic reefs until coral-stromatoporoid-bryozoan reefs Cambrian appeared in the mid-Ordovician. Microbial reefs were augmented by archaeocyath sponges for ~15 Myr in the Reef gap early Cambrian, by lithistid sponges for the remaining ~25 Myr of the Cambrian, and then by lithistid, calathiid Dysoxia and pulchrilaminid sponges for the first ~25 Myr of the Ordovician. The factors responsible for mid–late Hypoxia Cambrian microbial-lithistid sponge reef dominance remain unclear. Although oxygen increase appears to have Lithistid sponge-microbial reef significantly contributed to the early Cambrian ‘Explosion’ of marine animal life, it was followed by a prolonged period dominated by ‘greenhouse’ conditions, as sea-level rose and CO2 increased. The mid–late Cambrian was unusually warm, and these elevated temperatures can be expected to have lowered oxygen solubility, and to have promoted widespread thermal stratification resulting in marine dysoxia and hypoxia. Greenhouse condi- tions would also have stimulated carbonate platform development, locally further limiting shallow-water cir- culation. Low marine oxygenation has been linked to episodic extinctions of phytoplankton, trilobites and other metazoans during the mid–late Cambrian. -

International Stratigraphic Chart
INTERNATIONAL STRATIGRAPHIC CHART ICS International Commission on Stratigraphy Ma Ma Ma Ma Era Era Era Era Age Age Age Age Age Eon Eon Age Eon Age Eon Stage Stage Stage GSSP GSSP GSSA GSSP GSSP Series Epoch Series Epoch Series Epoch Period Period Period Period System System System System Erathem Erathem Erathem Erathem Eonothem Eonothem Eonothem 145.5 ±4.0 Eonothem 359.2 ±2.5 542 Holocene Tithonian Famennian Ediacaran 0.0117 150.8 ±4.0 Upper 374.5 ±2.6 Neo- ~635 Upper Upper Kimmeridgian Frasnian Cryogenian 0.126 ~ 155.6 385.3 ±2.6 proterozoic 850 “Ionian” Oxfordian Givetian Tonian Pleistocene 0.781 161.2 ±4.0 Middle 391.8 ±2.7 1000 Calabrian Callovian Eifelian Stenian Quaternary 1.806 164.7 ±4.0 397.5 ±2.7 Meso- 1200 Gelasian Bathonian Emsian Ectasian proterozoic 2.588 Middle 167.7 ±3.5 Devonian 407.0 ±2.8 1400 Piacenzian Bajocian Lower Pragian Calymmian Pliocene 3.600 171.6 ±3.0 411.2 ±2.8 1600 Zanclean Aalenian Lochkovian Statherian Jurassic 5.332 175.6 ±2.0 416.0 ±2.8 Proterozoic 1800 Messinian Toarcian Pridoli Paleo- Orosirian 7.246 183.0 ±1.5 418.7 ±2.7 2050 Tortonian Pliensbachian Ludfordian proterozoic Rhyacian 11.608 Lower 189.6 ±1.5 Ludlow 421.3 ±2.6 2300 Serravallian Sinemurian Gorstian Siderian Miocene 13.82 196.5 ±1.0 422.9 ±2.5 2500 Neogene Langhian Hettangian Homerian 15.97 199.6 ±0.6 Wenlock 426.2 ±2.4 Neoarchean Burdigalian M e s o z i c Rhaetian Sheinwoodian 20.43 203.6 ±1.5 428.2 ±2.3 2800 Aquitanian Upper Norian Silurian Telychian 23.03 216.5 ±2.0 436.0 ±1.9 P r e c a m b i n P Mesoarchean C e n o z i c Chattian Carnian -

The Weeks Formation Konservat-Lagerstätte and the Evolutionary Transition of Cambrian Marine Life
Downloaded from http://jgs.lyellcollection.org/ by guest on October 1, 2021 Review focus Journal of the Geological Society Published Online First https://doi.org/10.1144/jgs2018-042 The Weeks Formation Konservat-Lagerstätte and the evolutionary transition of Cambrian marine life Rudy Lerosey-Aubril1*, Robert R. Gaines2, Thomas A. Hegna3, Javier Ortega-Hernández4,5, Peter Van Roy6, Carlo Kier7 & Enrico Bonino7 1 Palaeoscience Research Centre, School of Environmental and Rural Science, University of New England, Armidale, NSW 2351, Australia 2 Geology Department, Pomona College, Claremont, CA 91711, USA 3 Department of Geology, Western Illinois University, 113 Tillman Hall, 1 University Circle, Macomb, IL 61455, USA 4 Department of Zoology, University of Cambridge, Downing Street, Cambridge CB2 3EJ, UK 5 Museum of Comparative Zoology and Department of Organismic and Evolutionary Biology, Harvard University, 26 Oxford Street, Cambridge, MA 02138, USA 6 Department of Geology, Ghent University, Krijgslaan 281/S8, B-9000 Ghent, Belgium 7 Back to the Past Museum, Carretera Cancún, Puerto Morelos, Quintana Roo 77580, Mexico R.L.-A., 0000-0003-2256-1872; R.R.G., 0000-0002-3713-5764; T.A.H., 0000-0001-9067-8787; J.O.-H., 0000-0002- 6801-7373 * Correspondence: [email protected] Abstract: The Weeks Formation in Utah is the youngest (c. 499 Ma) and least studied Cambrian Lagerstätte of the western USA. It preserves a diverse, exceptionally preserved fauna that inhabited a relatively deep water environment at the offshore margin of a carbonate platform, resembling the setting of the underlying Wheeler and Marjum formations. However, the Weeks fauna differs significantly in composition from the other remarkable biotas of the Cambrian Series 3 of Utah, suggesting a significant Guzhangian faunal restructuring. -

Ediacaran and Cambrian Stratigraphy in Estonia: an Updated Review
Estonian Journal of Earth Sciences, 2017, 66, 3, 152–160 https://doi.org/10.3176/earth.2017.12 Ediacaran and Cambrian stratigraphy in Estonia: an updated review Tõnu Meidla Department of Geology, Institute of Ecology and Earth Sciences, Faculty of Science and Technology, University of Tartu, Ravila 14a, 50411 Tartu, Estonia; [email protected] Received 18 December 2015, accepted 18 May 2017, available online 6 July 2017 Abstract. Previous late Precambrian and Cambrian correlation charts of Estonia, summarizing the regional stratigraphic nomenclature of the 20th century, date back to 1997. The main aim of this review is updating these charts based on recent advances in the global Precambrian and Cambrian stratigraphy and new data from regions adjacent to Estonia. The term ‘Ediacaran’ is introduced for the latest Precambrian succession in Estonia to replace the formerly used ‘Vendian’. Correlation with the dated sections in adjacent areas suggests that only the latest 7–10 Ma of the Ediacaran is represented in the Estonian succession. The gap between the Ediacaran and Cambrian may be rather substantial. The global fourfold subdivision of the Cambrian System is introduced for Estonia. The lower boundary of Series 2 is drawn at the base of the Sõru Formation and the base of Series 3 slightly above the former lower boundary of the ‘Middle Cambrian’ in the Baltic region, marked by a gap in the Estonian succession. The base of the Furongian is located near the base of the Petseri Formation. Key words: Ediacaran, Cambrian, correlation chart, biozonation, regional stratigraphy, Estonia, East European Craton. INTRODUCTION The latest stratigraphic chart of the Cambrian System in Estonia (Mens & Pirrus 1997b, p. -

1 Supplementary Materials and Methods 1 S1 Expanded
1 Supplementary Materials and Methods 2 S1 Expanded Geologic and Paleogeographic Information 3 The carbonate nodules from Montañez et al., (2007) utilized in this study were collected from well-developed and 4 drained paleosols from: 1) the Eastern Shelf of the Midland Basin (N.C. Texas), 2) Paradox Basin (S.E. Utah), 3) Pedregosa 5 Basin (S.C. New Mexico), 4) Anadarko Basin (S.C. Oklahoma), and 5) the Grand Canyon Embayment (N.C. Arizona) (Fig. 6 1a; Richey et al., (2020)). The plant cuticle fossils come from localities in: 1) N.C. Texas (Lower Pease River [LPR], Lake 7 Kemp Dam [LKD], Parkey’s Oil Patch [POP], and Mitchell Creek [MC]; all representing localities that also provided 8 carbonate nodules or plant organic matter [POM] for Montañez et al., (2007), 2) N.C. New Mexico (Kinney Brick Quarry 9 [KB]), 3) S.E. Kansas (Hamilton Quarry [HQ]), 4) S.E. Illinois (Lake Sara Limestone [LSL]), and 5) S.W. Indiana (sub- 10 Minshall [SM]) (Fig. 1a, S2–4; Richey et al., (2020)). These localities span a wide portion of the western equatorial portion 11 of Euramerica during the latest Pennsylvanian through middle Permian (Fig. 1b). 12 13 S2 Biostratigraphic Correlations and Age Model 14 N.C. Texas stratigraphy and the position of pedogenic carbonate samples from Montañez et al., (2007) and cuticle were 15 inferred from N.C. Texas conodont biostratigraphy and its relation to Permian global conodont biostratigraphy (Tabor and 16 Montañez, 2004; Wardlaw, 2005; Henderson, 2018). The specific correlations used are (C. Henderson, personal 17 communication, August 2019): (1) The Stockwether Limestone Member of the Pueblo Formation contains Idiognathodus 18 isolatus, indicating that the Carboniferous-Permian boundary (298.9 Ma) and base of the Asselian resides in the Stockwether 19 Limestone (Wardlaw, 2005). -

GEOLOGIC TIME SCALE V
GSA GEOLOGIC TIME SCALE v. 4.0 CENOZOIC MESOZOIC PALEOZOIC PRECAMBRIAN MAGNETIC MAGNETIC BDY. AGE POLARITY PICKS AGE POLARITY PICKS AGE PICKS AGE . N PERIOD EPOCH AGE PERIOD EPOCH AGE PERIOD EPOCH AGE EON ERA PERIOD AGES (Ma) (Ma) (Ma) (Ma) (Ma) (Ma) (Ma) HIST HIST. ANOM. (Ma) ANOM. CHRON. CHRO HOLOCENE 1 C1 QUATER- 0.01 30 C30 66.0 541 CALABRIAN NARY PLEISTOCENE* 1.8 31 C31 MAASTRICHTIAN 252 2 C2 GELASIAN 70 CHANGHSINGIAN EDIACARAN 2.6 Lopin- 254 32 C32 72.1 635 2A C2A PIACENZIAN WUCHIAPINGIAN PLIOCENE 3.6 gian 33 260 260 3 ZANCLEAN CAPITANIAN NEOPRO- 5 C3 CAMPANIAN Guada- 265 750 CRYOGENIAN 5.3 80 C33 WORDIAN TEROZOIC 3A MESSINIAN LATE lupian 269 C3A 83.6 ROADIAN 272 850 7.2 SANTONIAN 4 KUNGURIAN C4 86.3 279 TONIAN CONIACIAN 280 4A Cisura- C4A TORTONIAN 90 89.8 1000 1000 PERMIAN ARTINSKIAN 10 5 TURONIAN lian C5 93.9 290 SAKMARIAN STENIAN 11.6 CENOMANIAN 296 SERRAVALLIAN 34 C34 ASSELIAN 299 5A 100 100 300 GZHELIAN 1200 C5A 13.8 LATE 304 KASIMOVIAN 307 1250 MESOPRO- 15 LANGHIAN ECTASIAN 5B C5B ALBIAN MIDDLE MOSCOVIAN 16.0 TEROZOIC 5C C5C 110 VANIAN 315 PENNSYL- 1400 EARLY 5D C5D MIOCENE 113 320 BASHKIRIAN 323 5E C5E NEOGENE BURDIGALIAN SERPUKHOVIAN 1500 CALYMMIAN 6 C6 APTIAN LATE 20 120 331 6A C6A 20.4 EARLY 1600 M0r 126 6B C6B AQUITANIAN M1 340 MIDDLE VISEAN MISSIS- M3 BARREMIAN SIPPIAN STATHERIAN C6C 23.0 6C 130 M5 CRETACEOUS 131 347 1750 HAUTERIVIAN 7 C7 CARBONIFEROUS EARLY TOURNAISIAN 1800 M10 134 25 7A C7A 359 8 C8 CHATTIAN VALANGINIAN M12 360 140 M14 139 FAMENNIAN OROSIRIAN 9 C9 M16 28.1 M18 BERRIASIAN 2000 PROTEROZOIC 10 C10 LATE