3142.Full.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Type I Interferons and the Development of Impaired Vascular Function and Repair in Human and Murine Lupus
Type I Interferons and the Development of Impaired Vascular Function and Repair in Human and Murine Lupus by Seth G Thacker A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Immunology) in The University of Michigan 2011 Doctoral Committee: Associate Professor Mariana J. Kaplan, Chair Professor David A. Fox Professor Alisa E. Koch Professor Matthias Kretzler Professor Nicholas W. Lukacs Associate Professor Daniel T. Eitzman © Seth G Thacker 2011 Sharon, this work is dedicated to you. This achievement is as much yours as it is mine. Your support through all six years of this Ph.D. process has been incredible. You put up with my countless miscalculations on when I would finish experiments, and still managed to make me and our kids feel loved and special. Without you this would have no meaning. Sharon, you are the safe harbor in my life. ii Acknowledgments I have been exceptionally fortunate in my time here at the University of Michigan. I have been able to interact with so many supportive people over the years. I would like to express my thanks and admiration for my mentor. Mariana has taught me so much about writing, experimental design and being a successful scientist in general. I could never have made it here without her help. I would also like to thank Mike Denny. He had a hand in the beginning of all of my projects in one way or another, and was always quick and eager to help in whatever way he could. He really made my first year in the lab successful. -

IL-7) and IL-7 Splice Variants Affect Differentiation of Human Neural Progenitor Cells
Genes and Immunity (2010) 11, 11–20 & 2010 Macmillan Publishers Limited All rights reserved 1466-4879/10 $32.00 www.nature.com/gene ORIGINAL ARTICLE Interleukin-7 (IL-7) and IL-7 splice variants affect differentiation of human neural progenitor cells M Moors1,4, NK Vudattu2,, J Abel1, U Kra¨mer1, L Rane2, N Ulfig3, S Ceccatelli4, V Seyfert-Margolies5, E Fritsche1 and MJ Maeurer2 1Group of Toxicology, Group of Epidemiology, Institut fu¨r Umweltmedizinische Forschung, Du¨sseldorf, Germany; 2Microbiology, Tumor and Cell Biology and Smittskyddsinstitutet, Stockholm, Sweden; 3Department of Anatomy, University of Rostock, Rostock, Germany; 4Division of Neurotoxicology, Department of Neuroscience, Karolinska Institutet, Sweden and 5Department of Medicine, University of California, San Francisco, CA, USA Alternative splicing of pre-mRNA increases proteomic diversity, a crucial mechanism in defining tissue identity. We demonstrate differentially spliced interleukin (IL)-7 in distinct anatomic areas in the adult, in developing human brains and in normal human neuronal progenitor (NHNP) cells. IL-7c (c, the canonical form spanning all six exons) or its variants IL-7d5, d4 or d4/5 were cloned and expressed as recombinant proteins. IL-7 and splice variants were able to shift the differentiation of NHNP cells as compared with the diluent control (Po0.01) defined by anti-b (III)-tubulin and glial fibrillary acidic protein expression, with different degrees (IL-7c4d4/54IL-7d5); IL-7d4 exhibited a significantly weaker potency. Differentiation was confirmed by transcriptome analysis of IL-7c-stimulated neural NHNP cells, resulting in 58 differentially expressed genes; some of these are involved in neural differentiation, for example, the developmentally regulated transcription factor kru¨ppel-like factor 12, musashi 2, a translational regulator of cell fate or the sonic hedgehog receptor patch 1. -
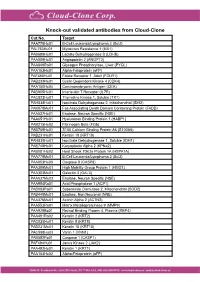
Knock-Out Validated Antibodies from Cloud-Clone Cat.No
Knock-out validated antibodies from Cloud-Clone Cat.No. Target PAA778Hu01 B-Cell Leukemia/Lymphoma 2 (Bcl2) PAL763Hu01 Myxovirus Resistance 1 (MX1) PAB698Hu01 Lactate Dehydrogenase B (LDHB) PAA009Hu01 Angiopoietin 2 (ANGPT2) PAA849Ra01 Glycogen Phosphorylase, Liver (PYGL) PAA153Hu01 Alpha-Fetoprotein (aFP) PAF460Hu01 Folate Receptor 1, Adult (FOLR1) PAB233Hu01 Cyclin Dependent Kinase 4 (CDK4) PAA150Hu04 Carcinoembryonic Antigen (CEA) PAB905Hu01 Interleukin 7 Receptor (IL7R) PAC823Hu01 Thymidine Kinase 1, Soluble (TK1) PAH838Hu01 Isocitrate Dehydrogenase 2, mitochondrial (IDH2) PAK078Mu01 Fas Associating Death Domain Containing Protein (FADD) PAA537Hu01 Enolase, Neuron Specific (NSE) PAA651Hu01 Hyaluronan Binding Protein 1 (HABP1) PAB215Hu02 Fibrinogen Beta (FGb) PAB769Hu01 S100 Calcium Binding Protein A6 (S100A6) PAB231Hu01 Keratin 18 (KRT18) PAH839Hu01 Isocitrate Dehydrogenase 1, Soluble (IDH1) PAE748Hu01 Karyopherin Alpha 2 (KPNa2) PAB081Hu02 Heat Shock 70kDa Protein 1A (HSPA1A) PAA778Mu01 B-Cell Leukemia/Lymphoma 2 (Bcl2) PAA853Hu03 Caspase 8 (CASP8) PAA399Mu01 High Mobility Group Protein 1 (HMG1) PAA303Mu01 Galectin 3 (GAL3) PAA537Mu02 Enolase, Neuron Specific (NSE) PAA994Ra01 Acid Phosphatase 1 (ACP1) PAB083Ra01 Superoxide Dismutase 2, Mitochondrial (SOD2) PAB449Mu01 Enolase, Non Neuronal (NNE) PAA376Mu01 Actinin Alpha 2 (ACTN2) PAA553Ra01 Matrix Metalloproteinase 9 (MMP9) PAA929Bo01 Retinol Binding Protein 4, Plasma (RBP4) PAA491Ra02 Keratin 2 (KRT2) PAC025Hu01 Keratin 8 (KRT8) PAB231Mu01 Keratin 18 (KRT18) PAC598Hu03 Vanin 1 (VNN1) -

Phase I Study of Recombinant Human Interleukin-7 Administration in Subjects with Refractory Malignancy
Published OnlineFirst January 12, 2010; DOI: 10.1158/1078-0432.CCR-09-1303 Cancer Therapy: Clinical Clinical Cancer Research Phase I Study of Recombinant Human Interleukin-7 Administration in Subjects with Refractory Malignancy Claude Sportès1, Rebecca R. Babb1, Michael C. Krumlauf1, Frances T. Hakim1, Seth M. Steinberg2, Catherine K. Chow5, Margaret R. Brown6, Thomas A. Fleisher6, Pierre Noel6, Irina Maric6, Maryalice Stetler-Stevenson3, Julie Engel7, Renaud Buffet7, Michel Morre7, Robert J. Amato8, Andrew Pecora9, Crystal L. Mackall4, and Ronald E. Gress1 Abstract Purpose: Interleukin-7 (IL-7) has critical and nonredundant roles in T-cell development, hematopoi- esis, and postdevelopmental immune functions as a prototypic homeostatic cytokine. Based on a large body of preclinical evidence, it may have multiple therapeutic applications in immunodeficiency states, either physiologic (immunosenescence), pathologic (HIV), or iatrogenic (postchemotherapy and posthe- matopoietic stem cell transplant), and may have roles in immune reconstitution or enhancement of im- munotherapy. We report here on the toxicity and biological activity of recombinant human IL-7 (rhIL-7) in humans. Design: Subjects with incurable malignancy received rhIL-7 subcutaneously every other day for 2 weeks in a phase I interpatient dose escalation study (3, 10, 30, and 60 μg/kg/dose). The objectives were safety and dose-limiting toxicity determination, identification of a range of biologically active doses, and char- acterization of biological and, possibly, antitumor effects. Results: Mild to moderate constitutional symptoms, reversible spleen and lymph node enlargement, and marked increase in peripheral CD3+, CD4+, and CD8+ lymphocytes were seen in a dose-dependent and age-independent manner in all subjects receiving ≥10 μg/kg/dose, resulting in a rejuvenated circulat- ing T-cell profile, resembling that seen earlier in life. -

Evolutionary Divergence and Functions of the Human Interleukin (IL) Gene Family Chad Brocker,1 David Thompson,2 Akiko Matsumoto,1 Daniel W
UPDATE ON GENE COMPLETIONS AND ANNOTATIONS Evolutionary divergence and functions of the human interleukin (IL) gene family Chad Brocker,1 David Thompson,2 Akiko Matsumoto,1 Daniel W. Nebert3* and Vasilis Vasiliou1 1Molecular Toxicology and Environmental Health Sciences Program, Department of Pharmaceutical Sciences, University of Colorado Denver, Aurora, CO 80045, USA 2Department of Clinical Pharmacy, University of Colorado Denver, Aurora, CO 80045, USA 3Department of Environmental Health and Center for Environmental Genetics (CEG), University of Cincinnati Medical Center, Cincinnati, OH 45267–0056, USA *Correspondence to: Tel: þ1 513 821 4664; Fax: þ1 513 558 0925; E-mail: [email protected]; [email protected] Date received (in revised form): 22nd September 2010 Abstract Cytokines play a very important role in nearly all aspects of inflammation and immunity. The term ‘interleukin’ (IL) has been used to describe a group of cytokines with complex immunomodulatory functions — including cell proliferation, maturation, migration and adhesion. These cytokines also play an important role in immune cell differentiation and activation. Determining the exact function of a particular cytokine is complicated by the influence of the producing cell type, the responding cell type and the phase of the immune response. ILs can also have pro- and anti-inflammatory effects, further complicating their characterisation. These molecules are under constant pressure to evolve due to continual competition between the host’s immune system and infecting organisms; as such, ILs have undergone significant evolution. This has resulted in little amino acid conservation between orthologous proteins, which further complicates the gene family organisation. Within the literature there are a number of overlapping nomenclature and classification systems derived from biological function, receptor-binding properties and originating cell type. -

Illuminating the Source of in Vivo Interleukin-7
DOI 10.4110/in.2011.11.1.1 REVIEW ARTICLE pISSN 1598-2629 eISSN 2092-6685 Seeing Is Believing: Illuminating the Source of In Vivo Interleukin-7 Grace Yoonhee Kim1,2,3, Changwan Hong1 and Jung-Hyun Park1* 1Experimental Immunology Branch, National Cancer Institute, National Institutes of Health, Bethesda, MD, 2Howard Hughes Medical Institute-NIH Research Scholars Program, Bethesda, MD, 3The Johns Hopkins University School of Medicine, Baltimore, MD, USA Interleukin-7 (IL-7) is an essential cytokine for T cells. INTRODUCTION However, IL-7 is not produced by T cells themselves such that T cells are dependent on extrinsic IL-7. In fact, in the ab- Originally described as a pre-B cell growth factor expressed sence of IL-7, T cell development in the thymus as well as in bone marrow stromal cells (1), interleukin-7 (IL-7) was survival of naive T cells in the periphery is severely impaired. subsequently identified as an essential and non-redundant cy- Furthermore, modulating IL-7 availability in vivo either by ge- tokine in T cells (2,3). In fact, IL-7 plays a critical role in netic means or other experimental approaches determines every aspect of T cell biology, including early T cell develop- the size, composition and function of the T cell pool. ment in the thymus, CD4/CD8 lineage choice during positive Consequently, understanding IL-7 expression is critical for understanding T cell immunity. Until most recently, however, selection as well as maintaining the survival and homeostasis the spatiotemporal expression of in vivo IL-7 has remained of naïve and memory T cells in the periphery (2-6). -

IL-7- and IL-15-Rich Environment Cells That Are Activated in An
Initial Antigen Encounter Programs CD8+ T Cells Competent to Develop into Memory Cells That Are Activated in an Antigen-Free, IL-7- and IL-15-Rich Environment This information is current as of September 26, 2021. Roberto Carrio, Oliver F. Bathe and Thomas R. Malek J Immunol 2004; 172:7315-7323; ; doi: 10.4049/jimmunol.172.12.7315 http://www.jimmunol.org/content/172/12/7315 Downloaded from References This article cites 52 articles, 26 of which you can access for free at: http://www.jimmunol.org/content/172/12/7315.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication by guest on September 26, 2021 *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2004 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Initial Antigen Encounter Programs CD8؉ T Cells Competent to Develop into Memory Cells That Are Activated in an Antigen-Free, IL-7- and IL-15-Rich Environment1 Roberto Carrio,* Oliver F. -

Human Cytokine Response Profiles
Comprehensive Understanding of the Human Cytokine Response Profiles A. Background The current project aims to collect datasets profiling gene expression patterns of human cytokine treatment response from the NCBI GEO and EBI ArrayExpress databases. The Framework for Data Curation already hosted a list of candidate datasets. You will read the study design and sample annotations to select the relevant datasets and label the sample conditions to enable automatic analysis. If you want to build a new data collection project for your topic of interest instead of working on our existing cytokine project, please read section D. We will explain the cytokine project’s configurations to give you an example on creating your curation task. A.1. Cytokine Cytokines are a broad category of small proteins mediating cell signaling. Many cell types can release cytokines and receive cytokines from other producers through receptors on the cell surface. Despite some overlap in the literature terminology, we exclude chemokines, hormones, or growth factors, which are also essential cell signaling molecules. Meanwhile, we count two cytokines in the same family as the same if they share the same receptors. In this project, we will focus on the following families and use the member symbols as standard names (Table 1). Family Members (use these symbols as standard cytokine names) Colony-stimulating factor GCSF, GMCSF, MCSF Interferon IFNA, IFNB, IFNG Interleukin IL1, IL1RA, IL2, IL3, IL4, IL5, IL6, IL7, IL9, IL10, IL11, IL12, IL13, IL15, IL16, IL17, IL18, IL19, IL20, IL21, IL22, IL23, IL24, IL25, IL26, IL27, IL28, IL29, IL30, IL31, IL32, IL33, IL34, IL35, IL36, IL36RA, IL37, TSLP, LIF, OSM Tumor necrosis factor TNFA, LTA, LTB, CD40L, FASL, CD27L, CD30L, 41BBL, TRAIL, OPGL, APRIL, LIGHT, TWEAK, BAFF Unassigned TGFB, MIF Table 1. -

Usefulness of IL-21, IL-7, and IL-15 Conditioned Media for Expansion of Antigen-Specific CD8+ T Cells from Healthy Donor-Pbmcs Suitable for Immunotherapy
Cellular Immunology 360 (2021) 104257 Contents lists available at ScienceDirect Cellular Immunology journal homepage: www.elsevier.com/locate/ycimm Research paper Usefulness of IL-21, IL-7, and IL-15 conditioned media for expansion of antigen-specific CD8+ T cells from healthy donor-PBMCs suitable for immunotherapy Julian´ A. Chamucero-Millares a,b, David A. Bernal-Est´evez b, Carlos A. Parra-Lopez´ a,* a Immunology and Translational Medicine Research Group, Department of Microbiology, School of Medicine, Universidad Nacional de Colombia, Carrera 30 No. 45-03, Bogota,´ South-America, Colombia b Immunology and Clinical Oncology Research Group, Fundacion´ Salud de los Andes, Calle 44 #58-05, Bogota,´ South-America, Colombia ARTICLE INFO ABSTRACT Keywords: Clonal anergy and depletion of antigen-specific CD8+ T cells are characteristics of immunosuppressed patients Antigen-specific T cells such as cancer and post-transplant patients. This has promoted translational research on the adoptive transfer of Monocyte-derived dendritic cells T cells to restore the antigen-specific cellular immunity in these patients. In the present work, we compared the Dendritic cell-based immunotherapy capability of PBMCs and two types of mature monocyte-derived DCs (moDCs) to prime and to expand ex-vivo Adoptive cell transfer antigen-specific CD8+ T cells using culture conditioned media supplemented with IL-7, IL-15, and IL-21. The Cancer immunotherapy Viral immunity data obtained suggest that protocols involving moDCs are as efficient as PBMCs-based cultures in expanding CD8 T cell antigen-specific CD8+ T cell to ELA and CMV model epitopes. These three gamma common chain cytokines Immunological memory promote the expansion of naïve-like and central memory CD8+ T cells in PBMCs-based cultures and the expansion of effector memory T cells when moDCs were used. -

The Interleukin 7 Receptor Is Differentially Expressed in the Blood of Patients with Crohn’S Disease
1 The interleukin 7 receptor is differentially expressed in the blood of patients with Crohn’s Disease. 2 Shahan Mamoor1 3 1Thomas Jefferson School of Law 4 San Diego, CA 92101 [email protected] 5 The inflammatory bowel diseases include Crohn’s Disease and Ulcerative Colitis (1). Crohn’s 6 Disease has been understood as a complex genetic disorder of multifactorial nature (2) but these factors remain incompletely understood. We mined multiple published and public 7 datasets (3, 4, 5) to determine in an unbiased and systematic fashion which genes were expressed most differently in the blood cells of patients with Crohn’s Disease as compared to 8 healthy controls. We found that the gene encoding the interleukin 7 receptor, IL7R, was among 9 the genes whose expression changed most significantly between patients with Crohn’s Disease as compared to healthy controls, in the whole blood, in monocyte-derived macrophages, and in 10 regulatory T-cells (Treg). While patients with Crohn’s Disease expressed significantly lower levels of IL-7R in the whole blood and in monocyte-derived macrophage than healthy controls, 11 patients with Crohn’s Disease expressed significantly higher levels of IL-7R in Treg. IL-7R expression has been studied in the intestinal tissues of patients with Crohn’s Disease but this is 12 the first study to document differential expression of the IL-7 receptor in the blood cells of patients with Crohn’s Disease. 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Keywords: Crohn’s Disease, IBD, CD, IL7R, IL-7, interleukin 7, genetics of CD 28 1 1 Almost 20 years after the identification of the IBD1 susceptibility locus as NOD2 (6), the 2 genes that conspire to produce the Crohn’s Disease phenotype have still not been defined. -

(IL-7) Selectively Promotes Mouse and Human IL-17–Producing Γδ Cells
Interleukin 7 (IL-7) selectively promotes mouse and human IL-17–producing γδ cells Marie-Laure Michela,b, Dick J. Pangc, Syeda F. Y. Haqueb, Alexandre J. Potocnikd, Daniel J. Penningtonc, and Adrian C. Haydaya,b,1 aLondon Research Institute, Cancer Research UK, London WC2A 3LY, United Kingdom; bThe Peter Gorer Department of Immunobiology, King’s College London, Guy’s Hospital, London SE19RT, United Kingdom; cThe Blizard Institute, Barts and The London School of Medicine, Queen Mary University of London, London E12AT, United Kingdom; and dDivision of Molecular Immunology, Medical Research Council National Institute for Medical Research, London NW71AA, United Kingdom Edited by Robert L. Coffman, Dynavax Technologies, Berkeley, CA, and approved September 18, 2012 (received for review March 14, 2012) − − IL-17–producing CD27 γδ cells (γδ27 cells) are widely viewed as Although IL-17–producing γδ cells are likewise commonly innate immune cells that make critical contributions to host pro- evoked in human immune responses and immunopathologies, tection and autoimmunity. However, factors that promote them very little is known about these cells, because they have proved over IFN-γ–producing γδ27+ cells are poorly elucidated. Moreover, particularly hard to isolate and characterize (16). Thus, it seemed 27− although human IL-17–producing γδ cells are commonly implicated logical that by elucidating stimuli for murine γδ cells, one might in inflammation, such cells themselves have proved difficult to iso- identify the means to expand their human counterparts. This γδ27− study identifies IL-7 as a profound and selective activator of IL- late and characterize. Here, murine T cells and thymocytes are – γδ shown to be rapidly and substantially expanded by IL-7 in vitro and 17 producing cells in mouse and in human neonates. -

Interleukin 7 and Patient Selection in Immunotherapy for Prostate Cancer
Interleukin 7 and Patient Selection in Immunotherapy for Prostate Cancer Caroline Schroten-Loef Interleukin 7 and Patient Selection in Immunotherapy for Prostate Cancer Caroline Schroten-Loef Publication of this thesis was financially supported by: ISBN/EAN: 978-90-9026960-3 The studies described in this thesis were performed at the laboratory of the Department of Experimental Urology, Josephine Nefkens Institute, Erasmus MC, Rotterdam, The Netherlands, and at the laboratory of Clinical & Tumor Immunology, Department of Medical Oncology, Erasmus MC – Daniel den Hoed, Rotterdam, The Netherlands. The research for this thesis was performed within the framework of the Erasmus Postgraduate School Molecular Medicine. Layout by M ART en DESIGN, Waddinxveen Printed by Print Service Ede BV Cover: Borobudur, Yogyakarta, Indonesia (by ocipalla; Burhan Bunardi) © 2012 C. Schroten-Loef Interleukin 7 and Patient Selection in Immunotherapy for Prostate Cancer Interleukine 7 en patiëntselectie ten bate van immunotherapie voor de behandeling van prostaatkanker. Proefschrift ter verkrijging van de graad van doctor aan de Erasmus Universiteit Rotterdam op gezag van de rector magnificus Prof.dr. H.G. Schmidt en volgens besluit van het College voor Promoties. De openbare verdediging zal plaats vinden op woensdag 16 januari 2013 om 11.30 uur door Caroline Schroten-Loef geboren te Waddinxveen Promotiecommissie Promotor : Prof.dr. C.H. Bangma Overige leden : Prof.dr.ir. G.W. Jenster Prof.dr.ir. J. Trapman Prof.dr. J.D. Laman Copromotoren : Dr. R. Kraaij Dr. R.A. Willemsen When one door of happiness closes, another opens; but often we look so long at the closed door that we do not see the one which has been opened for us.