Type I IFN Receptor Signaling Controls IL7-Dependent Accumulation and Activity of Protumoral IL17A-Producing Gdt Cells in Breast Cancer Emmanuel C
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supporting Information
Supporting Information Celhar et al. 10.1073/pnas.1507052112 SI Materials and Methods using a Nanodrop spectrophotometer (Thermo Fisher Scien- Proteinuria. Proteinuria was assessed using Albustix (Bayer). Al- tific). A TaqMan RNA-to-CT 1-Step Kit (Applied Biosystems) bumin levels in urine were assayed using an Albumin Mouse was used to perform the reverse transcription and quantitative ELISA Kit (Abcam) according to the manufacturer’s instructions; PCR reactions according to the manufacturer’s instructions samples were assayed at a dilution of 1:400. Samples were nor- using TaqMan gene expression assays (Applied Biosystems) to malized for creatinine using a Creatinine (urinary) Colorimetric either Tlr7 (Mm00446590) or the B2m housekeeping gene Assay Kit (Cayman Chemical) according to the manufacturer’s (Mm00437762). Real-time PCR was performed on the 7900H instructions; initial sample dilution of 1:10. fast real-time PCR system and analyzed using SDS 2.4 (Applied Biosystems). Relative mRNA expression was calculated using the Cell Sorting, RNA Isolation, and RT-PCR. Splenic B cells were comparative C method. + − + + t sorted as live CD45 Gr1 B220 CD19 , splenic T cells as live + − + + CD45 Gr1 CD3 CD5 and peritoneal macrophages as live Imaging. Kidney sections from OCT embedded tissue were fixed + − CD45 Gr1 CD11bhiF4/80hi. Sorted cells were centrifuged, re- with 4% paraformaldehyde before permeabilization with acetone suspended in TRIzol (Life Technologies) and stored at −80°. RNA and stained with Phalloidin (AF647) and anti-CD3d (unlabeled was extracted by TRIzol/chloroform and purified with the Qiagen Ab followed by secondary staining with donkey anti-goat Dylight RNeasy Mini purification kit according to the manufacturer’s 550). -

Type I Interferons and the Development of Impaired Vascular Function and Repair in Human and Murine Lupus
Type I Interferons and the Development of Impaired Vascular Function and Repair in Human and Murine Lupus by Seth G Thacker A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Immunology) in The University of Michigan 2011 Doctoral Committee: Associate Professor Mariana J. Kaplan, Chair Professor David A. Fox Professor Alisa E. Koch Professor Matthias Kretzler Professor Nicholas W. Lukacs Associate Professor Daniel T. Eitzman © Seth G Thacker 2011 Sharon, this work is dedicated to you. This achievement is as much yours as it is mine. Your support through all six years of this Ph.D. process has been incredible. You put up with my countless miscalculations on when I would finish experiments, and still managed to make me and our kids feel loved and special. Without you this would have no meaning. Sharon, you are the safe harbor in my life. ii Acknowledgments I have been exceptionally fortunate in my time here at the University of Michigan. I have been able to interact with so many supportive people over the years. I would like to express my thanks and admiration for my mentor. Mariana has taught me so much about writing, experimental design and being a successful scientist in general. I could never have made it here without her help. I would also like to thank Mike Denny. He had a hand in the beginning of all of my projects in one way or another, and was always quick and eager to help in whatever way he could. He really made my first year in the lab successful. -

Molecular Mechanisms Involved Involved in the Interaction Effects of HCV and Ethanol on Liver Cirrhosis
Virginia Commonwealth University VCU Scholars Compass Theses and Dissertations Graduate School 2010 Molecular Mechanisms Involved Involved in the Interaction Effects of HCV and Ethanol on Liver Cirrhosis Ryan Fassnacht Virginia Commonwealth University Follow this and additional works at: https://scholarscompass.vcu.edu/etd Part of the Physiology Commons © The Author Downloaded from https://scholarscompass.vcu.edu/etd/2246 This Thesis is brought to you for free and open access by the Graduate School at VCU Scholars Compass. It has been accepted for inclusion in Theses and Dissertations by an authorized administrator of VCU Scholars Compass. For more information, please contact [email protected]. Ryan C. Fassnacht 2010 All Rights Reserved Molecular Mechanisms Involved in the Interaction Effects of HCV and Ethanol on Liver Cirrhosis A thesis submitted in partial fulfillment of the requirements for the degree of Master of Science at Virginia Commonwealth University. by Ryan Christopher Fassnacht, B.S. Hampden Sydney University, 2005 M.S. Virginia Commonwealth University, 2010 Director: Valeria Mas, Ph.D., Associate Professor of Surgery and Pathology Division of Transplant Department of Surgery Virginia Commonwealth University Richmond, Virginia July 9, 2010 Acknowledgement The Author wishes to thank his family and close friends for their support. He would also like to thank the members of the molecular transplant team for their help and advice. This project would not have been possible with out the help of Dr. Valeria Mas and her endearing -

A Functional C-Terminal TRAF3-Binding Site in MAVS Participates in Positive and Negative Regulation of the IFN Antiviral Response
Cell Research (2011) 21:895-910. © 2011 IBCB, SIBS, CAS All rights reserved 1001-0602/11 $ 32.00 npg ORIGINAL ARTICLE www.nature.com/cr A functional C-terminal TRAF3-binding site in MAVS participates in positive and negative regulation of the IFN antiviral response Suzanne Paz1, 2, Myriam Vilasco3, Steven J Werden1, Meztli Arguello1, Deshanthe Joseph-Pillai1, 2, Tiejun Zhao1, Thi Lien-Anh Nguyen1, Qiang Sun1, Eliane F Meurs3, Rongtuan Lin1, 4, John Hiscott1, 2, 4 1Terry Fox Molecular Oncology Group, Lady Davis Institute, Jewish General Hospital, Montreal, Quebec H3T1E2, Canada; 2Department of Microbiology and Immunology, McGill University Montreal, Quebec H3A 2B4, Canada; 3Department of Virology, Unit of Hepacivirus and Innate Immunity, Pasteur Institute, Paris 75724 France; 4Department of Medicine, McGill University Montreal, Quebec H3A 2B4, Canada Recognition of viral RNA structures by the cytosolic sensor retinoic acid-inducible gene-I (RIG-I) results in the activation of signaling cascades that culminate with the generation of the type I interferon (IFN) antiviral response. Onset of antiviral and inflammatory responses to viral pathogens necessitates the regulated spatiotemporal recruitment of signaling adapters, kinases and transcriptional proteins to the mitochondrial antiviral signaling protein (MAVS). We previously demonstrated that the serine/threonine kinase IKKε is recruited to the C-terminal region of MAVS following Sendai or vesicular stomatitis virus (VSV) infection, mediated by Lys63-linked polyubiquitination of MAVS at Lys500, resulting in inhibition of downstream IFN signaling (Paz et al, Mol Cell Biol, 2009). In this study, we demonstrate that C-terminus of MAVS harbors a novel TRAF3-binding site in the aa450- 468 region of MAVS. -

Supplemental Materials ZNF281 Enhances Cardiac Reprogramming
Supplemental Materials ZNF281 enhances cardiac reprogramming by modulating cardiac and inflammatory gene expression Huanyu Zhou, Maria Gabriela Morales, Hisayuki Hashimoto, Matthew E. Dickson, Kunhua Song, Wenduo Ye, Min S. Kim, Hanspeter Niederstrasser, Zhaoning Wang, Beibei Chen, Bruce A. Posner, Rhonda Bassel-Duby and Eric N. Olson Supplemental Table 1; related to Figure 1. Supplemental Table 2; related to Figure 1. Supplemental Table 3; related to the “quantitative mRNA measurement” in Materials and Methods section. Supplemental Table 4; related to the “ChIP-seq, gene ontology and pathway analysis” and “RNA-seq” and gene ontology analysis” in Materials and Methods section. Supplemental Figure S1; related to Figure 1. Supplemental Figure S2; related to Figure 2. Supplemental Figure S3; related to Figure 3. Supplemental Figure S4; related to Figure 4. Supplemental Figure S5; related to Figure 6. Supplemental Table S1. Genes included in human retroviral ORF cDNA library. Gene Gene Gene Gene Gene Gene Gene Gene Symbol Symbol Symbol Symbol Symbol Symbol Symbol Symbol AATF BMP8A CEBPE CTNNB1 ESR2 GDF3 HOXA5 IL17D ADIPOQ BRPF1 CEBPG CUX1 ESRRA GDF6 HOXA6 IL17F ADNP BRPF3 CERS1 CX3CL1 ETS1 GIN1 HOXA7 IL18 AEBP1 BUD31 CERS2 CXCL10 ETS2 GLIS3 HOXB1 IL19 AFF4 C17ORF77 CERS4 CXCL11 ETV3 GMEB1 HOXB13 IL1A AHR C1QTNF4 CFL2 CXCL12 ETV7 GPBP1 HOXB5 IL1B AIMP1 C21ORF66 CHIA CXCL13 FAM3B GPER HOXB6 IL1F3 ALS2CR8 CBFA2T2 CIR1 CXCL14 FAM3D GPI HOXB7 IL1F5 ALX1 CBFA2T3 CITED1 CXCL16 FASLG GREM1 HOXB9 IL1F6 ARGFX CBFB CITED2 CXCL3 FBLN1 GREM2 HOXC4 IL1F7 -

Human Ifnα4 / Ifna4 / Interferon Alpha-4 Protein (Fc Tag)
Human IFNα4 / IFNa4 / Interferon alpha-4 Protein (Fc Tag) Catalog Number: 10336-H01H General Information SDS-PAGE: Gene Name Synonym: IFN-alpha4a; INFA4; MGC142200 Protein Construction: A DNA sequence encoding the mature form of human IFN-α4a (NP_066546.1) (Cys 23-Asp 189) was expressed with the N-terminal fused Fc region of human IgG1. Source: Human Expression Host: HEK293 Cells QC Testing Purity: > 97 % as determined by SDS-PAGE Bio Activity: Protein Description Measured in antiviral assays using WISH human amnion cells infected Interferon, alpha 4 (IFNA4) belongs to the alpha/beta interferon family. Two with vesicular stomatitis virus (VSV). The EC50 for this effect is 0.2-1 variants of IFNA4 (IFNA4a and IFNA4b) are known, which differ from each ng/mL. other by changes in their coding regions at nucleotide positions 220 and 410 and can be distinguished by selective restriction enzyme analysis. Endotoxin: Interferons are produced by macrophages, IFN-alpha have antiviral activities. Interferon stimulates the production of two enzymes: a protein < 1.0 EU per μg of the protein as determined by the LAL method kinase and an oligoadenylate synthetase. IFN-alpha, the first cytokine to be produced by recombinant DNA technology, has emerged as an important Stability: regulator of growth and differentiation, affecting cellular communication and Samples are stable for up to twelve months from date of receipt at -70 ℃ signal transduction pathways as well as immunological control. Originally discovered as an antiviral substance, the efficacy of IFN-alpha in malignant, Predicted N terminal: Glu viral, immunological, angiogenic, inflammatory, and fibrotic diseases suggests a spectrum of interrelated pathophysiologies. -

IL-7) and IL-7 Splice Variants Affect Differentiation of Human Neural Progenitor Cells
Genes and Immunity (2010) 11, 11–20 & 2010 Macmillan Publishers Limited All rights reserved 1466-4879/10 $32.00 www.nature.com/gene ORIGINAL ARTICLE Interleukin-7 (IL-7) and IL-7 splice variants affect differentiation of human neural progenitor cells M Moors1,4, NK Vudattu2,, J Abel1, U Kra¨mer1, L Rane2, N Ulfig3, S Ceccatelli4, V Seyfert-Margolies5, E Fritsche1 and MJ Maeurer2 1Group of Toxicology, Group of Epidemiology, Institut fu¨r Umweltmedizinische Forschung, Du¨sseldorf, Germany; 2Microbiology, Tumor and Cell Biology and Smittskyddsinstitutet, Stockholm, Sweden; 3Department of Anatomy, University of Rostock, Rostock, Germany; 4Division of Neurotoxicology, Department of Neuroscience, Karolinska Institutet, Sweden and 5Department of Medicine, University of California, San Francisco, CA, USA Alternative splicing of pre-mRNA increases proteomic diversity, a crucial mechanism in defining tissue identity. We demonstrate differentially spliced interleukin (IL)-7 in distinct anatomic areas in the adult, in developing human brains and in normal human neuronal progenitor (NHNP) cells. IL-7c (c, the canonical form spanning all six exons) or its variants IL-7d5, d4 or d4/5 were cloned and expressed as recombinant proteins. IL-7 and splice variants were able to shift the differentiation of NHNP cells as compared with the diluent control (Po0.01) defined by anti-b (III)-tubulin and glial fibrillary acidic protein expression, with different degrees (IL-7c4d4/54IL-7d5); IL-7d4 exhibited a significantly weaker potency. Differentiation was confirmed by transcriptome analysis of IL-7c-stimulated neural NHNP cells, resulting in 58 differentially expressed genes; some of these are involved in neural differentiation, for example, the developmentally regulated transcription factor kru¨ppel-like factor 12, musashi 2, a translational regulator of cell fate or the sonic hedgehog receptor patch 1. -

Supplementary Material DNA Methylation in Inflammatory Pathways Modifies the Association Between BMI and Adult-Onset Non- Atopic
Supplementary Material DNA Methylation in Inflammatory Pathways Modifies the Association between BMI and Adult-Onset Non- Atopic Asthma Ayoung Jeong 1,2, Medea Imboden 1,2, Akram Ghantous 3, Alexei Novoloaca 3, Anne-Elie Carsin 4,5,6, Manolis Kogevinas 4,5,6, Christian Schindler 1,2, Gianfranco Lovison 7, Zdenko Herceg 3, Cyrille Cuenin 3, Roel Vermeulen 8, Deborah Jarvis 9, André F. S. Amaral 9, Florian Kronenberg 10, Paolo Vineis 11,12 and Nicole Probst-Hensch 1,2,* 1 Swiss Tropical and Public Health Institute, 4051 Basel, Switzerland; [email protected] (A.J.); [email protected] (M.I.); [email protected] (C.S.) 2 Department of Public Health, University of Basel, 4001 Basel, Switzerland 3 International Agency for Research on Cancer, 69372 Lyon, France; [email protected] (A.G.); [email protected] (A.N.); [email protected] (Z.H.); [email protected] (C.C.) 4 ISGlobal, Barcelona Institute for Global Health, 08003 Barcelona, Spain; [email protected] (A.-E.C.); [email protected] (M.K.) 5 Universitat Pompeu Fabra (UPF), 08002 Barcelona, Spain 6 CIBER Epidemiología y Salud Pública (CIBERESP), 08005 Barcelona, Spain 7 Department of Economics, Business and Statistics, University of Palermo, 90128 Palermo, Italy; [email protected] 8 Environmental Epidemiology Division, Utrecht University, Institute for Risk Assessment Sciences, 3584CM Utrecht, Netherlands; [email protected] 9 Population Health and Occupational Disease, National Heart and Lung Institute, Imperial College, SW3 6LR London, UK; [email protected] (D.J.); [email protected] (A.F.S.A.) 10 Division of Genetic Epidemiology, Medical University of Innsbruck, 6020 Innsbruck, Austria; [email protected] 11 MRC-PHE Centre for Environment and Health, School of Public Health, Imperial College London, W2 1PG London, UK; [email protected] 12 Italian Institute for Genomic Medicine (IIGM), 10126 Turin, Italy * Correspondence: [email protected]; Tel.: +41-61-284-8378 Int. -
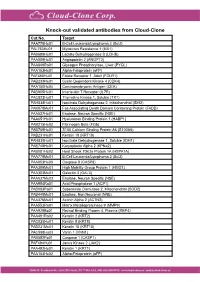
Knock-Out Validated Antibodies from Cloud-Clone Cat.No
Knock-out validated antibodies from Cloud-Clone Cat.No. Target PAA778Hu01 B-Cell Leukemia/Lymphoma 2 (Bcl2) PAL763Hu01 Myxovirus Resistance 1 (MX1) PAB698Hu01 Lactate Dehydrogenase B (LDHB) PAA009Hu01 Angiopoietin 2 (ANGPT2) PAA849Ra01 Glycogen Phosphorylase, Liver (PYGL) PAA153Hu01 Alpha-Fetoprotein (aFP) PAF460Hu01 Folate Receptor 1, Adult (FOLR1) PAB233Hu01 Cyclin Dependent Kinase 4 (CDK4) PAA150Hu04 Carcinoembryonic Antigen (CEA) PAB905Hu01 Interleukin 7 Receptor (IL7R) PAC823Hu01 Thymidine Kinase 1, Soluble (TK1) PAH838Hu01 Isocitrate Dehydrogenase 2, mitochondrial (IDH2) PAK078Mu01 Fas Associating Death Domain Containing Protein (FADD) PAA537Hu01 Enolase, Neuron Specific (NSE) PAA651Hu01 Hyaluronan Binding Protein 1 (HABP1) PAB215Hu02 Fibrinogen Beta (FGb) PAB769Hu01 S100 Calcium Binding Protein A6 (S100A6) PAB231Hu01 Keratin 18 (KRT18) PAH839Hu01 Isocitrate Dehydrogenase 1, Soluble (IDH1) PAE748Hu01 Karyopherin Alpha 2 (KPNa2) PAB081Hu02 Heat Shock 70kDa Protein 1A (HSPA1A) PAA778Mu01 B-Cell Leukemia/Lymphoma 2 (Bcl2) PAA853Hu03 Caspase 8 (CASP8) PAA399Mu01 High Mobility Group Protein 1 (HMG1) PAA303Mu01 Galectin 3 (GAL3) PAA537Mu02 Enolase, Neuron Specific (NSE) PAA994Ra01 Acid Phosphatase 1 (ACP1) PAB083Ra01 Superoxide Dismutase 2, Mitochondrial (SOD2) PAB449Mu01 Enolase, Non Neuronal (NNE) PAA376Mu01 Actinin Alpha 2 (ACTN2) PAA553Ra01 Matrix Metalloproteinase 9 (MMP9) PAA929Bo01 Retinol Binding Protein 4, Plasma (RBP4) PAA491Ra02 Keratin 2 (KRT2) PAC025Hu01 Keratin 8 (KRT8) PAB231Mu01 Keratin 18 (KRT18) PAC598Hu03 Vanin 1 (VNN1) -

Phase I Study of Recombinant Human Interleukin-7 Administration in Subjects with Refractory Malignancy
Published OnlineFirst January 12, 2010; DOI: 10.1158/1078-0432.CCR-09-1303 Cancer Therapy: Clinical Clinical Cancer Research Phase I Study of Recombinant Human Interleukin-7 Administration in Subjects with Refractory Malignancy Claude Sportès1, Rebecca R. Babb1, Michael C. Krumlauf1, Frances T. Hakim1, Seth M. Steinberg2, Catherine K. Chow5, Margaret R. Brown6, Thomas A. Fleisher6, Pierre Noel6, Irina Maric6, Maryalice Stetler-Stevenson3, Julie Engel7, Renaud Buffet7, Michel Morre7, Robert J. Amato8, Andrew Pecora9, Crystal L. Mackall4, and Ronald E. Gress1 Abstract Purpose: Interleukin-7 (IL-7) has critical and nonredundant roles in T-cell development, hematopoi- esis, and postdevelopmental immune functions as a prototypic homeostatic cytokine. Based on a large body of preclinical evidence, it may have multiple therapeutic applications in immunodeficiency states, either physiologic (immunosenescence), pathologic (HIV), or iatrogenic (postchemotherapy and posthe- matopoietic stem cell transplant), and may have roles in immune reconstitution or enhancement of im- munotherapy. We report here on the toxicity and biological activity of recombinant human IL-7 (rhIL-7) in humans. Design: Subjects with incurable malignancy received rhIL-7 subcutaneously every other day for 2 weeks in a phase I interpatient dose escalation study (3, 10, 30, and 60 μg/kg/dose). The objectives were safety and dose-limiting toxicity determination, identification of a range of biologically active doses, and char- acterization of biological and, possibly, antitumor effects. Results: Mild to moderate constitutional symptoms, reversible spleen and lymph node enlargement, and marked increase in peripheral CD3+, CD4+, and CD8+ lymphocytes were seen in a dose-dependent and age-independent manner in all subjects receiving ≥10 μg/kg/dose, resulting in a rejuvenated circulat- ing T-cell profile, resembling that seen earlier in life. -

Evolutionary Divergence and Functions of the Human Interleukin (IL) Gene Family Chad Brocker,1 David Thompson,2 Akiko Matsumoto,1 Daniel W
UPDATE ON GENE COMPLETIONS AND ANNOTATIONS Evolutionary divergence and functions of the human interleukin (IL) gene family Chad Brocker,1 David Thompson,2 Akiko Matsumoto,1 Daniel W. Nebert3* and Vasilis Vasiliou1 1Molecular Toxicology and Environmental Health Sciences Program, Department of Pharmaceutical Sciences, University of Colorado Denver, Aurora, CO 80045, USA 2Department of Clinical Pharmacy, University of Colorado Denver, Aurora, CO 80045, USA 3Department of Environmental Health and Center for Environmental Genetics (CEG), University of Cincinnati Medical Center, Cincinnati, OH 45267–0056, USA *Correspondence to: Tel: þ1 513 821 4664; Fax: þ1 513 558 0925; E-mail: [email protected]; [email protected] Date received (in revised form): 22nd September 2010 Abstract Cytokines play a very important role in nearly all aspects of inflammation and immunity. The term ‘interleukin’ (IL) has been used to describe a group of cytokines with complex immunomodulatory functions — including cell proliferation, maturation, migration and adhesion. These cytokines also play an important role in immune cell differentiation and activation. Determining the exact function of a particular cytokine is complicated by the influence of the producing cell type, the responding cell type and the phase of the immune response. ILs can also have pro- and anti-inflammatory effects, further complicating their characterisation. These molecules are under constant pressure to evolve due to continual competition between the host’s immune system and infecting organisms; as such, ILs have undergone significant evolution. This has resulted in little amino acid conservation between orthologous proteins, which further complicates the gene family organisation. Within the literature there are a number of overlapping nomenclature and classification systems derived from biological function, receptor-binding properties and originating cell type. -

Illuminating the Source of in Vivo Interleukin-7
DOI 10.4110/in.2011.11.1.1 REVIEW ARTICLE pISSN 1598-2629 eISSN 2092-6685 Seeing Is Believing: Illuminating the Source of In Vivo Interleukin-7 Grace Yoonhee Kim1,2,3, Changwan Hong1 and Jung-Hyun Park1* 1Experimental Immunology Branch, National Cancer Institute, National Institutes of Health, Bethesda, MD, 2Howard Hughes Medical Institute-NIH Research Scholars Program, Bethesda, MD, 3The Johns Hopkins University School of Medicine, Baltimore, MD, USA Interleukin-7 (IL-7) is an essential cytokine for T cells. INTRODUCTION However, IL-7 is not produced by T cells themselves such that T cells are dependent on extrinsic IL-7. In fact, in the ab- Originally described as a pre-B cell growth factor expressed sence of IL-7, T cell development in the thymus as well as in bone marrow stromal cells (1), interleukin-7 (IL-7) was survival of naive T cells in the periphery is severely impaired. subsequently identified as an essential and non-redundant cy- Furthermore, modulating IL-7 availability in vivo either by ge- tokine in T cells (2,3). In fact, IL-7 plays a critical role in netic means or other experimental approaches determines every aspect of T cell biology, including early T cell develop- the size, composition and function of the T cell pool. ment in the thymus, CD4/CD8 lineage choice during positive Consequently, understanding IL-7 expression is critical for understanding T cell immunity. Until most recently, however, selection as well as maintaining the survival and homeostasis the spatiotemporal expression of in vivo IL-7 has remained of naïve and memory T cells in the periphery (2-6).