In Drinking-Water
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Effects of Calcium Chelation on Digitalis-Induced Cardiac Arrhythmias by Paul Szekely and N
Br Heart J: first published as 10.1136/hrt.25.5.589 on 1 September 1963. Downloaded from Brit. Heart J., 1963, 25, 589. EFFECTS OF CALCIUM CHELATION ON DIGITALIS-INDUCED CARDIAC ARRHYTHMIAS BY PAUL SZEKELY AND N. A. WYNNE From the Cardiovascular Department, Newcastle General Hospital, and the Department ofPhysiology, King's College, University of Durham Received November 19, 1962 Several studies have already shown that cardiac arrhythmias caused by digitalis can be abolished by the induction of hypocalcaemia (Page and Real, 1955; Smith and Grinnell, 1955; Gubner and Kallman, 1957; Kabakow and Brothers, 1958; Surawicz et al., 1959; Cohen et al., 1959; Rosenbaum, Mason, and Seven, 1960; Surawicz, 1960; Soffer, Toribara, and Sayman, 1961). The rationale of inducing hypocalcemia as an anti-arrhythmic measure is based on experimental and clinical obser- vations relating to the direct myocardial action of calcium and to the interaction between calcium, potassium, and digitalis. Low calcium concentration decreases myocardial irritability (Brooks et al., 1955) and it also increases the intracellular potassium concentration (Rosenbaum et al., 1960). There is also a synergistic action between calcium and digitalis, which was observed in the presence of digitalis intoxication (Nalbandian et al., 1957). The present study was undertaken copyright. in order to assess the value of induced hypocalcemia in the management of cardiac arrhythmias caused by digitalis. MATERIAL AND METHODS Forty-eight experiments were carried out under general aneesthesia on 46 cats and 2 dogs. Cardiac arrhythmia was induced by the intravenous administration of tincture of digitalis as previously described http://heart.bmj.com/ (Szekely and Wynne, 1951). -

New Brunswick Drug Plans Formulary
New Brunswick Drug Plans Formulary August 2019 Administered by Medavie Blue Cross on Behalf of the Government of New Brunswick TABLE OF CONTENTS Page Introduction.............................................................................................................................................I New Brunswick Drug Plans....................................................................................................................II Exclusions............................................................................................................................................IV Legend..................................................................................................................................................V Anatomical Therapeutic Chemical (ATC) Classification of Drugs A Alimentary Tract and Metabolism 1 B Blood and Blood Forming Organs 23 C Cardiovascular System 31 D Dermatologicals 81 G Genito Urinary System and Sex Hormones 89 H Systemic Hormonal Preparations excluding Sex Hormones 100 J Antiinfectives for Systemic Use 107 L Antineoplastic and Immunomodulating Agents 129 M Musculo-Skeletal System 147 N Nervous System 156 P Antiparasitic Products, Insecticides and Repellants 223 R Respiratory System 225 S Sensory Organs 234 V Various 240 Appendices I-A Abbreviations of Dosage forms.....................................................................A - 1 I-B Abbreviations of Routes................................................................................A - 4 I-C Abbreviations of Units...................................................................................A -

Chelation Therapy
Medical Policy Chelation Therapy Table of Contents • Policy: Commercial • Coding Information • Information Pertaining to All Policies • Policy: Medicare • Description • References • Authorization Information • Policy History Policy Number: 122 BCBSA Reference Number: 8.01.02 NCD/LCD: N/A Related Policies None Policy Commercial Members: Managed Care (HMO and POS), PPO, and Indemnity Medicare HMO BlueSM and Medicare PPO BlueSM Members Chelation therapy in the treatment of the following conditions is MEDICALLY NECESSARY: • Extreme conditions of metal toxicity • Treatment of chronic iron overload due to blood transfusions (transfusional hemosiderosis) or due to nontransfusion-dependent thalassemia (NTDT) • Wilson's disease (hepatolenticular degeneration), or • Lead poisoning. Chelation therapy in the treatment of the following conditions is MEDICALLY NECESSARY if other modalities have failed: • Control of ventricular arrhythmias or heart block associated with digitalis toxicity • Emergency treatment of hypercalcemia. NaEDTA as chelation therapy is considered NOT MEDICALLY NECESSARY. Off-label applications of chelation therapy are considered INVESTIGATIONAL, including, but not limited to: • Alzheimer’s disease • Arthritis (includes rheumatoid arthritis) • Atherosclerosis, (e.g., coronary artery disease, secondary prevention in patients with myocardial infarction, or peripheral vascular disease) • Autism • Diabetes • Multiple sclerosis. 1 Prior Authorization Information Inpatient • For services described in this policy, precertification/preauthorization IS REQUIRED for all products if the procedure is performed inpatient. Outpatient • For services described in this policy, see below for products where prior authorization might be required if the procedure is performed outpatient. Outpatient Commercial Managed Care (HMO and POS) Prior authorization is not required. Commercial PPO and Indemnity Prior authorization is not required. Medicare HMO BlueSM Prior authorization is not required. -

Post-Injury Calcium Chelation Rescues Skeletal Muscle Regeneration in Mice Matthew .D Magda University of Connecticut - Storrs, [email protected]
University of Connecticut OpenCommons@UConn Honors Scholar Theses Honors Scholar Program Summer 8-1-2013 Post-Injury Calcium Chelation Rescues Skeletal Muscle Regeneration in Mice Matthew .D Magda University of Connecticut - Storrs, [email protected] Follow this and additional works at: https://opencommons.uconn.edu/srhonors_theses Part of the Cell Biology Commons, and the Molecular Biology Commons Recommended Citation Magda, Matthew D., "Post-Injury Calcium Chelation Rescues Skeletal Muscle Regeneration in Mice" (2013). Honors Scholar Theses. 321. https://opencommons.uconn.edu/srhonors_theses/321 1 Post-Injury Calcium Chelation Rescues Skeletal Muscle Regeneration in Mice Honors Thesis Matthew Magda Research Advisor: Dr. Morgan Carlson Honors Advisor: Dr. Kenneth Noll August 2013 2 Abstract: Antibiotics, surgery and organ transplants have pushed average lifespans towards the upper limits of the human body. Drastically reduced morbidity from infection, toxins and traumatic injury have allowed ever greater portions of the populace can reach eighty or ninety years old before dying of old age. Despite the increased role of aging as a source of morbidity, many aspects of aging are poorly characterized. Sarcopenia, progressive muscle loss, and loss of adult myogenic potential, the ability to produce new muscle tissue from adult stem cell sources, are key causes of decreased mobility and strength in aged individuals. If more youthful muscle quality could be restored in old patients they would experience greatly improved quality of life and perhaps even longer lifespans. Satellite cell populations are known to decline sharply by 6-7th decade of life but traditional treatments for sarcopenia, namely exercise intervention, have been shown to exacerbate the degeneration in aged such patients. -

Chelation Therapy
Corporate Medical Policy Chelation Therapy File Name: chelation_therapy Origination: 12/1995 Last CAP Review: 2/2021 Next CAP Review: 2/2022 Last Review: 2/2021 Description of Procedure or Service Chelation therapy is an established treatment for the removal of metal toxins by converting them to a chemically inert form that can be excreted in the urine. Chelation therapy comprises intravenous or oral administration of chelating agents that remove metal ions such as lead, aluminum, mercury, arsenic, zinc, iron, copper, and calcium from the body. Specific chelating agents are used for particular heavy metal toxicities. For example, desferroxamine (not Food and Drug Administration [FDA] approved) is used for patients with iron toxicity, and calcium-ethylenediaminetetraacetic acid (EDTA) is used for patients with lead poisoning. Note that disodium-EDTA is not recommended for acute lead poisoning due to the increased risk of death from hypocalcemia. Another class of chelating agents, called metal protein attenuating compounds (MPACs), is under investigation for the treatment of Alzheimer’s disease, which is associated with the disequilibrium of cerebral metals. Unlike traditional systemic chelators that bind and remove metals from tissues systemically, MPACs have subtle effects on metal homeostasis and abnormal metal interactions. In animal models of Alzheimer’s disease, they promote the solubilization and clearance of β-amyloid protein by binding to its metal-ion complex and also inhibit redox reactions that generate neurotoxic free radicals. MPACs therefore interrupt two putative pathogenic processes of Alzheimer’s disease. However, no MPACs have received FDA approval for treating Alzheimer’s disease. Chelation therapy has also been investigated as a treatment for other indications including atherosclerosis and autism spectrum disorder. -

EDTA, Tetrasodium Tetrahydrate Salt
EDTA, Tetrasodium Tetrahydrate Salt sc-204735 Material Safety Data Sheet Hazard Alert Code Key: EXTREME HIGH MODERATE LOW Section 1 - CHEMICAL PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME EDTA, Tetrasodium Tetrahydrate Salt STATEMENT OF HAZARDOUS NATURE CONSIDERED A HAZARDOUS SUBSTANCE ACCORDING TO OSHA 29 CFR 1910.1200. NFPA FLAMMABILITY1 HEALTH3 HAZARD INSTABILITY0 SUPPLIER Company: Santa Cruz Biotechnology, Inc. Address: 2145 Delaware Ave Santa Cruz, CA 95060 Telephone: 800.457.3801 or 831.457.3800 Emergency Tel: CHEMWATCH: From within the US and Canada: 877-715-9305 Emergency Tel: From outside the US and Canada: +800 2436 2255 (1-800-CHEMCALL) or call +613 9573 3112 PRODUCT USE Chelating/sequestering agent for the clarification of liquids, pharmaceuticals, detergents, shampoos, agent for metal poisoning, decontamination of radioactive liquids and in analytical chemistry. Used in agricultural chemical sprays; metal cleaning and plating; to remove insoluble deposits of magnesium and calcium salts; in textiles to improve dying; scouring and detergent operations; decreasing blood cholesterol. SYNONYMS C10-H12-N2-Na4-O8, (OOCCH2)2NCH2CH2CH2N(CH2COO).Na4, "Tetracemate tetrasodium", "Versene 220", Aquamollin, "Edathanil tetrasodium", Tetracemin, Sequestrene, Complexon, Tyclarosol, "Endrate tetrasodium", Tetrine, Irrgalon, "Nervanaid B", Questex, Kalex, Calsol, Warkeelate, "edate terasodium", "Trilon B", Nervanoid, Aquamoline, "tetrasodium EDTA", Komplexon, "Syntes 12a", "tetrasodium edetate", Nullapon, Chemcolox, Conigon, ethylenebis(imminodiacetate), -

7 EDTA, Tetraso
REPORT Safety Assessment of EDTA, Calcium Disodium EDTA. Diammonium EDTA, Dipotassium EDTA. 7 Disodium EDTA, TEA- EDTA, Tetrasod=um EDTA, Tripotassium EDTA, Trisodium EDTA, HEDTA and Trisodium HEDTA ABSTRACT EDTA (ethylenediamine tetraacetic acid) and its salts are substituted diamines. HEDTA (hydroxyethyl ethylenediamine triacetic acid) and its trisodium salt are substituted amines. These ingredients function as chelating agents in cosmetic formulations. The typical concentration of use of EDTA is less than 2%, with the other salts in current use at even lower concentrations. The lowest dose reported to cause a toxic effect in animals was 750 mg/kg/day. These chelating agents are cytotoxic and weakly genotoxic, but not carcinogenic. Oral exposures to EDTA produced adverse reproductive and developmental effects in animals. Clinical tests reported no absorption of an EDTA salt through the skin. These ingredients, are likely, however, to affect the passage of other chemicals into the skin because they will chelate calcium. Exposure to ED TA in most cosmetic formulations, therefore, would produce systemic exposure levels well below those seen to be toxic in oral dosing studies. Exposure to EDTA in cosmetic formulations that may be inhaled, however, was a concern. An exposure assessment done using conservative assumptions predicted that the maximum EDTA dose via inhalation of an aerosolized cosmetic formulation is below that which has been shown to produce reproductive or developmental toxicity. Because of the potential to increase the penetration of other chemicals, formulators should continue to be aware of this when combining these ingredients with ingredients that previously have been determined to be safe primarily because they were not significanUy absorbed. -

Chelation of Actinides
UC Berkeley UC Berkeley Previously Published Works Title Chelation of Actinides Permalink https://escholarship.org/uc/item/4b57t174 Author Abergel, RJ Publication Date 2017 DOI 10.1039/9781782623892-00183 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Chapter 6 Chelation of Actinides rebecca J. abergela aChemical Sciences Division, lawrence berkeley National laboratory, One Cyclotron road, berkeley, Ca 94720, USa *e-mail: [email protected] 6.1 The Medical and Public Health Relevance of Actinide Chelation the use of actinides in the civilian industry and defense sectors over the past 60 years has resulted in persistent environmental and health issues, since a large inventory of radionuclides, including actinides such as thorium (th), uranium (U), neptunium (Np), plutonium (pu), americium (am) and curium 1 Downloaded by Lawrence Berkeley National Laboratory on 22/06/2018 20:28:11. (Cm), are generated and released during these activities. Controlled process- Published on 18 October 2016 http://pubs.rsc.org | doi:10.1039/9781782623892-00183 ing and disposal of wastes from the nuclear fuel cycle are the main source of actinide dissemination. however, significant quantities of these radionu- clides have also been dispersed as a consequence of nuclear weapons testing, nuclear power plant accidents, and compromised storage of nuclear materi- als.1 In addition, events of the last fifteen years have heightened public con- cern that actinides may be released as the result of the potential terrorist use of radiological dispersal devices or after a natural disaster affecting nuclear power plants or nuclear material storage sites.2,3 all isotopes of the 15 ele- ments of the actinide series (atomic numbers 89 through 103, Figure 6.1) are radioactive and have the potential to be harmful; the heaviest members, however, are too unstable to be isolated in quantities larger than a few atoms at a time,4 and those elements cited above (U, Np, pu, am, Cm) are the most RSC Metallobiology Series No. -

EDTA (Ethylenediaminetetraacetic Acid) C Barton, Oak Ridge Institute for Science and Education, Oakridge, TN, USA
EDTA (Ethylenediaminetetraacetic Acid) C Barton, Oak Ridge Institute for Science and Education, Oakridge, TN, USA Ó 2014 Elsevier Inc. All rights reserved. This article is a revision of the previous edition article by C. Charles Barton and Harihara M. Mehendale, volume 2, pp 147–148, Ó 2005, Elsevier Inc. l Name: EDTA (Ethylenediaminetetraacetic Acid) depending on the salt. Algae and invertebrates are among the l Chemical Abstracts Service Registry Number: 60-00-4 most sensitive species based on predictive modeling for acute l Synonyms: Ethylenedinitrilotetraacetic acid; Celon A; and chronic endpoints for EDTA, depending on the compound. Cheelox; Edetic acid; Nullapon B Acid; Trilon BW; Versene EDTA and its salts also do not appear to be very toxic for l Molecular Formula: C10H16N2O8 terrestrial wild mammals, and adverse effects from reasonably l Chemical Structure: expected agricultural uses are not expected. According to ChemIDPlus, EDTA has the following physi- OH cochemical properties: melting point ¼ 245 C, pKa dissocia- tion constant ¼ 0.26, log P (octanol–water) ¼3.860, water À solubility ¼ 1000 mg l 1, vapor pressure ¼ 4.98E-13 mm Hg, O O À Henry’s law constant ¼ 1.17E-23 atm-m3 mol 1, and atmo- ¼ 3 À1 N OH spheric OH rate constant 1.82E-10 cm molecule-s . HO N Based on its physicochemical properties, EDTA is not expected to volatilize from soil or water. When released to the O O atmosphere, EDTA should sorb to particulate matter, and appears to have the potential to photolyze. OH Exposure and Exposure Monitoring Background Exposure to EDTA may be through FDA-approved uses as food Ethylenediaminetetraacetic acid (EDTA) was developed by Franz additives, in sanitizing solutions, in pharmaceutical products, Munz in Germany during the 1930s as an alternative to citric acid. -

Effect of Alpha Lipoic Acid on the Blood Cell Count and Iron Kinetics In
Nutr Hosp. 2015;31(2):883-889 ISSN 0212-1611 • CODEN NUHOEQ S.V.R. 318 Original / Farmacia Effect of alpha lipoic acid on the blood cell count and iron kinetics in hypertensive patients Paula Renata Florêncio Mendes, Danielle dos Santos Félix, Paulo César Dantas da Silva, Guêdijany Henrique Pereira and Mônica Oliveira da Silva Simões Dean of Graduate Studies and Research, State University of Paraiba, Campina Grande, Brazil. Abstract EFECTO DEL ÁCIDO LIPOICO EN EL RECUENTO DE SANGRE Y EL METABOLISMO Introduction: The α-lipoic acid (ALA) has been used DE HIERRO EN PACIENTES HIPERTENSOS as a treatment to reduce oxidative damage in Systemic Arterial Hypertension (SAH), but there are no in vivo studies reporting the effect of its mechanism of action on Resumen iron metabolism. Introducción: El Ácido α-Lipóico (ALA) ha sido uti- α Objective: To evaluate the antioxidant effect of -lipoic lizado como recurso terapéutico para reducir daño oxi- acid on Blood cell count (CBC) and iron metabolism in dativo en la Hipertensión Arterial Sistémica (HAS), pero hypertensive subjects with or without anemia. aún no existen estudios in vivo que reporten sobre su me- Method: Double-blind, randomized, placebo-contro- canismo de acción en el metabolismo del hierro. lled clinical trial. The sample consisted of 60 hyperten- Objetivo: Evaluar el efecto antioxidante del ácido Al- sive patients that were randomly divided into treatment fa-Lipóico sobre el hemograma y metabolismo del hierro group (n = 32), receiving 600 mg / day of ALA for twelve en individuos hipertensos con o sin anemia. weeks and control group (n = 28), receiving placebo for Métodos: Estudio clínico doble-ciego, randomizado y the same period. -
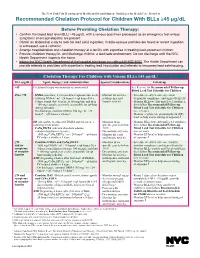
Recommended Chelation Protocol for Children with Blls ≥45 Μg/Dl
The New York City Department of Health and Mental Hygiene Guidelines for Health Care Providers Recommended Chelation Protocol for Children With BLLs ≥45 μg/dL Before Providing Chelation Therapy: • Confirm the blood lead level (BLL) ≥45 μg/dL with a venous specimen processed as an emergency test unless symptoms of encephalopathy are present. • Obtain an abdominal x-ray to look for lead solid ingestion; if radio-opaque particles are found or recent ingestion is witnessed, use a cathartic. • Arrange hospitalization and chelation therapy at a facility with expertise in treating lead-poisoned children. • Provide chelation therapy in, and discharge child to, a lead-safe environment. Do not discharge until the NYC Health Department inspects the home. • Inform the NYC Health Department of the hospital admission by calling 646-632-6002. The Health Department can provide referrals to providers with expertise in treating lead intoxication and referrals to temporary lead-safe housing. Chelation Therapy For Children with Venous BLLs ≥45 μg/dL1 BLL (μg/dL) Agent, Dosage,* and Administration Special Considerations Follow-up <45 Chelation therapy not routinely recommended See Reverse for Recommended Follow-up Blood Lead Test Schedule for Children 45 to <70 • DMSA (succimer, 2,3-meso-dimercaptosuccinic acid) • Monitor for anemia, • Schedule weekly health care visits • 1050 mg DMSA / m2 / 24 hours* ÷ q8 hours PO x neutropenia, and to monitor compliance and signs of toxicity. 5 days; round dose to nearest 100 mg/day, and then hepatic toxicity. • Monitor BLLs weekly until level stabilizes, ÷ 100-mg capsules as evenly as possible for q8-hour then follow Recommended Follow-up dosing schedule. -

Report Worst Ingredients in Liquid Soap Italian Products
Date Dec 9, 2011 Report Worst Ingredients in Liquid Soap Italian products EuroConsumers EnviroPlanning AB Lilla Bommen 5 C, SE-411 04 Göteborg, Sweden Visitor address Lilla Bommen 5 C Telephone +46 31 771 87 40 Telefax +46 31 771 87 41 Web site www.enviroplanning.se e-mail [email protected] Worst Ingredients in Liquid Soap Date Dec 9, 2011 Italian products Version Final Doc.no 4084-01\10\01\Report.doc About the report Title Worst Ingredients in Liquid Soap Italian products Version 001 Date December 9, 2011 Client Euroconsumers Servizi Editoriali S.R.L. Via Valassina, 22 20159 MILANO Italy Project number 4084-01 Document number 4084-01\10\01\Report.doc Cover photo Helena Norin Report written by Helena Norin and Niklas Hanson Report reviewed by Jenny Robinson Report verified by Helena Norin U:\4084-01\10-udo\01-utr\Report 4084-01.doc I (III) Worst Ingredients in Liquid Soap Date Dec 9, 2011 Italian products Version Final Doc.no 4084-01\10\01\Report.doc Summary Euroconsumers is preparing a review of liquid soaps on the Italian market. One part of this review looks at the environmental properties of the ingredients in the chosen products. EnviroPlanning has been hired to assess the environmental properties of the ingredients in order to identify and list ten or more of the most un-wanted ingredients. Severe health aspects of the ingredients are also taken into account when establishing the list. The following ingredients are considered as the most unwanted in the examined 27 soaps: BHT (butylated hydroxytoluene) Methylchloroisothiazolinone Zinc Oxide Methylisothiazolinone Limonene Lauric Acid 2-Bromo-2-Nitropropane-1,3-Diol Benzyl Benzoate Butylphenyl Methylpropional Disodium EDTA Formaldehyde The review shows that the liquid soaps on the Italian market have a wide range of ingredients.