Alternative Splicing in Prostate and Breast Cancer
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Atlas Antibodies in Breast Cancer Research Table of Contents
ATLAS ANTIBODIES IN BREAST CANCER RESEARCH TABLE OF CONTENTS The Human Protein Atlas, Triple A Polyclonals and PrecisA Monoclonals (4-5) Clinical markers (6) Antibodies used in breast cancer research (7-13) Antibodies against MammaPrint and other gene expression test proteins (14-16) Antibodies identified in the Human Protein Atlas (17-14) Finding cancer biomarkers, as exemplified by RBM3, granulin and anillin (19-22) Co-Development program (23) Contact (24) Page 2 (24) Page 3 (24) The Human Protein Atlas: a map of the Human Proteome The Human Protein Atlas (HPA) is a The Human Protein Atlas consortium cell types. All the IHC images for Swedish-based program initiated in is mainly funded by the Knut and Alice the normal tissue have undergone 2003 with the aim to map all the human Wallenberg Foundation. pathology-based annotation of proteins in cells, tissues and organs expression levels. using integration of various omics The Human Protein Atlas consists of technologies, including antibody- six separate parts, each focusing on References based imaging, mass spectrometry- a particular aspect of the genome- 1. Sjöstedt E, et al. (2020) An atlas of the based proteomics, transcriptomics wide analysis of the human proteins: protein-coding genes in the human, pig, and and systems biology. mouse brain. Science 367(6482) 2. Thul PJ, et al. (2017) A subcellular map of • The Tissue Atlas shows the the human proteome. Science. 356(6340): All the data in the knowledge resource distribution of proteins across all eaal3321 is open access to allow scientists both major tissues and organs in the 3. -
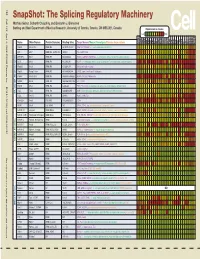
Snapshot: the Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J
192 Cell SnapShot: The Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J. Blencowe 133 Banting and Best Department of Medical Research, University of Toronto, Toronto, ON M5S 3E1, Canada Expression in mouse , April4, 2008©2008Elsevier Inc. Low High Name Other Names Protein Domains Binding Sites Target Genes/Mouse Phenotypes/Disease Associations Amy Ceb Hip Hyp OB Eye SC BM Bo Ht SM Epd Kd Liv Lu Pan Pla Pro Sto Spl Thy Thd Te Ut Ov E6.5 E8.5 E10.5 SRp20 Sfrs3, X16 RRM, RS GCUCCUCUUC SRp20, CT/CGRP; −/− early embryonic lethal E3.5 9G8 Sfrs7 RRM, RS, C2HC Znf (GAC)n Tau, GnRH, 9G8 ASF/SF2 Sfrs1 RRM, RS RGAAGAAC HipK3, CaMKIIδ, HIV RNAs; −/− embryonic lethal, cond. KO cardiomyopathy SC35 Sfrs2 RRM, RS UGCUGUU AChE; −/− embryonic lethal, cond. KO deficient T-cell maturation, cardiomyopathy; LS SRp30c Sfrs9 RRM, RS CUGGAUU Glucocorticoid receptor SRp38 Fusip1, Nssr RRM, RS ACAAAGACAA CREB, type II and type XI collagens SRp40 Sfrs5, HRS RRM, RS AGGAGAAGGGA HipK3, PKCβ-II, Fibronectin SRp55 Sfrs6 RRM, RS GGCAGCACCUG cTnT, CD44 DOI 10.1016/j.cell.2008.03.010 SRp75 Sfrs4 RRM, RS GAAGGA FN1, E1A, CD45; overexpression enhances chondrogenic differentiation Tra2α Tra2a RRM, RS GAAARGARR GnRH; overexpression promotes RA-induced neural differentiation SR and SR-Related Proteins Tra2β Sfrs10 RRM, RS (GAA)n HipK3, SMN, Tau SRm160 Srrm1 RS, PWI AUGAAGAGGA CD44 SWAP Sfrs8 RS, SWAP ND SWAP, CD45, Tau; possible asthma susceptibility gene hnRNP A1 Hnrnpa1 RRM, RGG UAGGGA/U HipK3, SMN2, c-H-ras; rheumatoid arthritis, systemic lupus -

Logistic Regression Analysis for the Identification of the Metastasis-Associated Signaling Pathways of Osteosarcoma
INTERNATIONAL JOURNAL OF MOLECULAR MEDICINE 41: 1233-1244, 2018 Logistic regression analysis for the identification of the metastasis-associated signaling pathways of osteosarcoma YANG LIU1, WEI SUN2, XIAOJUN MA2, YUEDONG HAO3, GANG LIU3, XIAOHUI HU3, HOULAI SHANG3, PENGFEI WU3, ZEXUE ZHAO3 and WEIDONG LIU3 1Department of Orthopedics, Affiliated Hospital of Inner Mongolia University for The Nationalities, Tongliao, Inner Mongolia 028007; 2Department of Orthopaedics, Shanghai General Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai 200080; 3Department of Orthopedics, Huai'an First People's Hospital, Nanjing Medical University, Huai'an, Jiangsu 223300, P.R. China Received February 20, 2017; Accepted September 26, 2017 DOI: 10.3892/ijmm.2018.3360 Abstract. Osteosarcoma (OS) is the most common histological migration were identified as the terms that were significantly type of primary bone cancer. The present study was designed different. ICAM1, PDGFB, PDGFRB and TGFB1 were iden- to identify the key genes and signaling pathways involved in the tified to be enriched in cell migration and regulation of cell metastasis of OS. Microarray data of GSE39055 were down- migration. Nucleotide excision repair and the Wnt signaling loaded from the Gene Expression Omnibus database, which pathway were the metastasis-associated pathways of OS. In included 19 OS biopsy specimens before metastasis (control addition, ICAM1, PDGFB, PDGFRB and TGFB1, which were group) and 18 OS biopsy specimens after metastasis (case involved in cell migration and regulation of cell migration may group). After the differentially expressed genes (DEGs) were affect the metastasis of OS. identified using the Linear Models for Microarray Analysis package, hierarchical clustering analysis and unsupervised Introduction clustering analysis were performed separately, using orange software and the self-organization map method. -

Analysis of Alternative Splicing Associated with Aging and Neurodegeneration in the Human Brain
Downloaded from genome.cshlp.org on October 2, 2021 - Published by Cold Spring Harbor Laboratory Press Research Analysis of alternative splicing associated with aging and neurodegeneration in the human brain James R. Tollervey,1 Zhen Wang,1 Tibor Hortoba´gyi,2 Joshua T. Witten,1 Kathi Zarnack,3 Melis Kayikci,1 Tyson A. Clark,4 Anthony C. Schweitzer,4 Gregor Rot,5 Tomazˇ Curk,5 Blazˇ Zupan,5 Boris Rogelj,2 Christopher E. Shaw,2 and Jernej Ule1,6 1MRC Laboratory of Molecular Biology, Hills Road, Cambridge CB2 0QH, United Kingdom; 2MRC Centre for Neurodegeneration Research, King’s College London, Institute of Psychiatry, De Crespigny Park, London SE5 8AF, United Kingdom; 3EMBL–European Bioinformatics Institute, Wellcome Trust Genome Campus, Hinxton, Cambridge CB10 1SD, United Kingdom; 4Expression Research, Affymetrix, Inc., Santa Clara, California 95051, USA; 5Faculty of Computer and Information Science, University of Ljubljana, Trzˇasˇka 25, SI-1000 Ljubljana, Slovenia Age is the most important risk factor for neurodegeneration; however, the effects of aging and neurodegeneration on gene expression in the human brain have most often been studied separately. Here, we analyzed changes in transcript levels and alternative splicing in the temporal cortex of individuals of different ages who were cognitively normal, affected by frontotemporal lobar degeneration (FTLD), or affected by Alzheimer’s disease (AD). We identified age-related splicing changes in cognitively normal individuals and found that these were present also in 95% of individuals with FTLD or AD, independent of their age. These changes were consistent with increased polypyrimidine tract binding protein (PTB)– dependent splicing activity. We also identified disease-specific splicing changes that were present in individuals with FTLD or AD, but not in cognitively normal individuals. -

Dynamic Changes in RNA–Protein Interactions and RNA Secondary Structure in Mammalian Erythropoiesis
Published Online: 27 July, 2021 | Supp Info: http://doi.org/10.26508/lsa.202000659 Downloaded from life-science-alliance.org on 30 September, 2021 Resource Dynamic changes in RNA–protein interactions and RNA secondary structure in mammalian erythropoiesis Mengge Shan1,2 , Xinjun Ji3, Kevin Janssen5 , Ian M Silverman3 , Jesse Humenik3, Ben A Garcia5, Stephen A Liebhaber3,4, Brian D Gregory1,2 Two features of eukaryotic RNA molecules that regulate their and RNA secondary structure. These techniques generally either post-transcriptional fates are RNA secondary structure and RNA- use chemical probing agents or structure-specific RNases (single- binding protein (RBP) interaction sites. However, a comprehen- stranded RNases (ssRNases) and double-stranded RNases [dsRNa- sive global overview of the dynamic nature of these sequence ses]) to provide site-specific evidence for a region being in single- or features during erythropoiesis has never been obtained. Here, we double-stranded configurations (4, 5). use our ribonuclease-mediated structure and RBP-binding site To date, the known repertoire of RBP–RNA interaction sites has mapping approach to reveal the global landscape of RNA sec- been built on a protein-by-protein basis, with studies identifying ondary structure and RBP–RNA interaction sites and the dynamics the targets of a single protein of interest, often through the use of of these features during this important developmental process. techniques such as crosslinking and immunoprecipitation se- We identify dynamic patterns of RNA secondary structure and RBP quencing (CLIP-seq) (6). In CLIP-seq, samples are irradiated with UV binding throughout the process and determine a set of corre- to induce the cross-linking of proteins to their RNA targets. -

Large-Scale Analysis of Genome and Transcriptome Alterations in Multiple Tumors Unveils Novel Cancer-Relevant Splicing Networks
Downloaded from genome.cshlp.org on October 2, 2021 - Published by Cold Spring Harbor Laboratory Press Large-scale analysis of genome and transcriptome alterations in multiple tumors unveils novel cancer-relevant splicing networks Endre Sebestyén1,*, Babita Singh1,*, Belén Miñana1,2, Amadís Pagès1, Francesca Mateo3, Miguel Angel Pujana3, Juan Valcárcel1,2,4, Eduardo Eyras1,4,5 1Universitat Pompeu Fabra, Dr. Aiguader 88, E08003 Barcelona, Spain 2Centre for Genomic Regulation, Dr. Aiguader 88, E08003 Barcelona, Spain 3Program Against Cancer Therapeutic Resistance (ProCURE), Catalan Institute of Oncology (ICO), Bellvitge Institute for Biomedical Research (IDIBELL), E08908 L’Hospitalet del Llobregat, Spain. 4Catalan Institution for Research and Advanced Studies, Passeig Lluís Companys 23, E08010 Barcelona, Spain *Equal contribution 5Correspondence to: [email protected] Keywords: alternative splicing, RNA binding proteins, splicing networks, cancer 1 Downloaded from genome.cshlp.org on October 2, 2021 - Published by Cold Spring Harbor Laboratory Press Abstract Alternative splicing is regulated by multiple RNA-binding proteins and influences the expression of most eukaryotic genes. However, the role of this process in human disease, and particularly in cancer, is only starting to be unveiled. We systematically analyzed mutation, copy number and gene expression patterns of 1348 RNA-binding protein (RBP) genes in 11 solid tumor types, together with alternative splicing changes in these tumors and the enrichment of binding motifs in the alternatively spliced sequences. Our comprehensive study reveals widespread alterations in the expression of RBP genes, as well as novel mutations and copy number variations in association with multiple alternative splicing changes in cancer drivers and oncogenic pathways. Remarkably, the altered splicing patterns in several tumor types recapitulate those of undifferentiated cells. -

Overview of Research on Fusion Genes in Prostate Cancer
2011 Review Article Overview of research on fusion genes in prostate cancer Chunjiao Song1,2, Huan Chen3 1Medical Research Center, Shaoxing People’s Hospital, Shaoxing University School of Medicine, Shaoxing 312000, China; 2Shaoxing Hospital, Zhejiang University School of Medicine, Shaoxing 312000, China; 3Key Laboratory of Microorganism Technology and Bioinformatics Research of Zhejiang Province, Zhejiang Institute of Microbiology, Hangzhou 310000, China Contributions: (I) Conception and design: C Song; (II) Administrative support: Shaoxing Municipal Health and Family Planning Science and Technology Innovation Project (2017CX004) and Shaoxing Public Welfare Applied Research Project (2018C30058); (III) Provision of study materials or patients: None; (IV) Collection and assembly of data: C Song; (V) Data analysis and interpretation: H Chen; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors. Correspondence to: Chunjiao Song. No. 568 Zhongxing Bei Road, Shaoxing 312000, China. Email: [email protected]. Abstract: Fusion genes are known to drive and promote carcinogenesis and cancer progression. In recent years, the rapid development of biotechnologies has led to the discovery of a large number of fusion genes in prostate cancer specimens. To further investigate them, we summarized the fusion genes. We searched related articles in PubMed, CNKI (Chinese National Knowledge Infrastructure) and other databases, and the data of 92 literatures were summarized after preliminary screening. In this review, we summarized approximated 400 fusion genes since the first specific fusion TMPRSS2-ERG was discovered in prostate cancer in 2005. Some of these are prostate cancer specific, some are high-frequency in the prostate cancer of a certain ethnic group. This is a summary of scientific research in related fields and suggests that some fusion genes may become biomarkers or the targets for individualized therapies. -

Genome-Wide Analysis of the Alternative Splicing Profiles
Journal of Cancer 2020, Vol. 11 1542 Ivyspring International Publisher Journal of Cancer 2020; 11(6): 1542-1554. doi: 10.7150/jca.36998 Research Paper Genome-wide Analysis of the Alternative Splicing Profiles Revealed Novel Prognostic Index for Kidney Renal Cell Clear Cell Carcinoma Li Gao1#, Rong-quan He2#, Zhi-guang Huang1, Yi-wu Dang1, Yong-yao Gu1, Hai-biao Yan3, Sheng-hua Li3, Gang Chen1 1. Department of Pathology, First Affiliated Hospital of Guangxi Medical University, Nanning, Guangxi Zhuang Autonomous Region 530021, P. R. China 2. Department of Medical Oncology, First Affiliated Hospital of Guangxi Medical University, Nanning, Guangxi Zhuang Autonomous Region 530021, P. R. China 3. Department of Urology, First Affiliated Hospital of Guangxi Medical University, Nanning, Guangxi Zhuang Autonomous Region 530021, P. R. China # Li Gao and Rong-quan He contributed equally to this study. Corresponding authors: Sheng-hua Li: Tel: +86 0771-5356533; Fax: 0086-771-5350031; Email: [email protected]; Gang Chen: Tel: +86 0771-5356534; Fax: 0086-771-5356534; Email: [email protected] © The author(s). This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See http://ivyspring.com/terms for full terms and conditions. Received: 2019.05.25; Accepted: 2019.12.13; Published: 2020.01.14 Abstract Alternative splicing (AS) is a major mechanism that greatly enhanced the diversity of proteome. Mounting evidence demonstrated that aberration of AS are important steps for the initiation and progression of human cancers. Here, we comprehensively investigated the association between whole landscape of AS profiles and the survival outcome of renal cell carcinoma (RCC) patients using RNA-seq data from TCGA SpliceSeq. -

RNA-Seq Analysis of Prostate Cancer in the Chinese Population Identifies Recurrent Gene Fusions, Cancer-Associated Long Noncodin
npg RNA-seq analysis of prostate cancer in Chinese population Cell Research (2012) 22:806-821. 806 © 2012 IBCB, SIBS, CAS All rights reserved 1001-0602/12 $ 32.00 npg ORIGINAL ARTICLE www.nature.com/cr RNA-seq analysis of prostate cancer in the Chinese population identifies recurrent gene fusions, cancer-associated long noncoding RNAs and aberrant alternative splicings Shancheng Ren1, *, Zhiyu Peng2, *, Jian-Hua Mao3, *, Yongwei Yu4, Changjun Yin5, Xin Gao6, Zilian Cui1, Jibin Zhang2, Kang Yi2, Weidong Xu1, Chao Chen2, Fubo Wang1, Xinwu Guo2, Ji Lu1, Jun Yang2, Min Wei1, Zhijian Tian2, Yinghui Guan3, Liang Tang1, Chuanliang Xu1, Linhui Wang1, Xu Gao1, Wei Tian2, Jian Wang2, Huanming Yang2, Jun Wang2, Yinghao Sun1 1Department of Urology, Shanghai Changhai Hospital, Second Military Medical University, Shanghai 200433, China; 2Beijing Ge- nomics Institute at Shenzhen, Shenzhen, Guangdong 518083, China; 3Life Sciences Division, Lawrence Berkeley National Labora- tory, One Cyclotron Road, Berkeley, CA 94720, USA; 4Department of Pathology, Shanghai Changhai Hospital, Second Military Medical University, Shanghai 200433, China; 5Department of Urology, Jiangsu Provincial People’s Hospital, Nanjing Medical University, Nanjing, Jiangsu 210029, China; 6Department of Urology, the Third Affiliated Hospital of Sun Yat Sen University, Guangzhou, Guangdong 510630, China There are remarkable disparities among patients of different races with prostate cancer; however, the mechanism underlying this difference remains unclear. Here, we present a comprehensive landscape of the transcriptome profiles of 14 primary prostate cancers and their paired normal counterparts from the Chinese population using RNA-seq, revealing tremendous diversity across prostate cancer transcriptomes with respect to gene fusions, long noncoding RNAs (long ncRNA), alternative splicing and somatic mutations. -

A Fast, Non-Radioactive Method to Identify in Vivo Targets of RNA-Binding Proteins
bioRxiv preprint doi: https://doi.org/10.1101/158410; this version posted July 1, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. uvCLAP: a fast, non-radioactive method to identify in vivo targets of RNA-binding proteins Daniel Maticzka1,4, Ibrahim Avsar Ilik2,4, Tugce Aktas2,4, Rolf Backofen1,3,# and Asifa Akhtar2,#* 1 Bioinformatics Group, Department of Computer Science, University of Freiburg, Freiburg, Germany 2 Max Planck Institute of Immunobiology and Epigenetics, 79108 Freiburg im Breisgau, Germany 3 Centre for Biological Signalling Studies (BIOSS), University of Freiburg, Freiburg, Germany 4 These authors contributed equally to this work # Corresponding author [email protected] Phone: +49 761 203 7461 Fax: +49 761 203 7462 # Corresponding author, * Lead contact [email protected] Phone: +49 (0)7615108565 Fax: +49 (0)7615108566 1 bioRxiv preprint doi: https://doi.org/10.1101/158410; this version posted July 1, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract RNA-binding proteins (RBPs) play important and essential roles in eukaryotic gene expression regulating splicing, localization, translation and stability of mRNAs. Understanding the exact contribution of RBPs to gene regulation is crucial as many RBPs are frequently mis-regulated in several neurological diseases and certain cancers. While recently developed techniques provide binding sites of RBPs, they are labor-intensive and generally rely on radioactive labeling of RNA. With more than 1,000 RBPs in a human cell, it is imperative to develop easy, robust, reproducible and high-throughput methods to determine in vivo targets of RBPs. -

Original Article High Frequency of the SDK1:AMACR Fusion Transcript in Chinese Prostate Cancer
Int J Clin Exp Med 2015;8(9):15127-15136 www.ijcem.com /ISSN:1940-5901/IJCEM0012785 Original Article High frequency of the SDK1:AMACR fusion transcript in Chinese prostate cancer Yanling Zhang1,2, Xue-Ying Mao3, Xiaoyan Liu1, Rong-Rong Song1, Daniel Berney3, Yong-Jie Lu3, Guoping Ren1 1Department of Pathology, The First Affiliated Hospital, Zhejiang University Medical College, Hangzhou, China; 2Department of Gynecology and Obstetrics, Sir Run Run Shaw Hospital, Zhejiang University Medical College, Hangzhou, China; 3Centre for Molecular Oncology, Barts Cancer Institute, Queen Mary University of London, UK Received July 12, 2015; Accepted September 10, 2015; Epub September 15, 2015; Published September 30, 2015 Abstract: Chromosomal rearrangements and fusion genes play important roles in tumor development and pro- gression. Four high-frequency prostate cancer (CaP) specific fusion genes, SDK1:AMACR, RAD50:PDLIM4, CTAGE5:KHDRBS3 and USP9Y:TTTY15 have been reported in Chinese CaP samples through a transcriptome se- quencing study. We previously reported that USP9Y:TTTY15 is a transcription-mediated chimeric RNA, which is expressed in both tumor and non-malignant samples, and here we attempted to confirm the existence of the other three fusion genes SDK1:AMACR, RAD50:PDLIM and CTAGE5:KHDRBS3. We detected SDK1:AMACR fusion tran- script in 23 of 100 Chinese CaP samples, but did not detect RAD50:PDLIM4 and CTAGE5:KHDRBS3 transcripts in any of those samples. SDK1:AMACR fusion transcript is Chinese CaP specific, which was neither detected in non- malignant prostate tissues adjacent to cancer from Chinese patient nor in CaP samples from UK patients. However, we did not detect genomic rearrangement of SDK1 gene by fluorescence in situ hybridization analysis, indicating that SDK1:AMACR is also a transcription-mediated chimeric RNA. -

KHDRBS3 Antibody Order 021-34695924 [email protected] Support 400-6123-828 50Ul [email protected] 100 Ul √ √ Web
TD9598 KHDRBS3 Antibody Order 021-34695924 [email protected] Support 400-6123-828 50ul [email protected] 100 uL √ √ Web www.ab-mart.com.cn Description: RNA-binding protein that plays a role in the regulation of alternative splicing and influences mRNA splice site selection and exon inclusion. Binds preferentially to the 5'-[AU]UAAA-3' motif in vitro. Binds optimally to RNA containing 5'-[AU]UAA-3' as a bipartite motif spaced by more than 15 nucleotides. Binds poly(A). RNA-binding abilities are down-regulated by tyrosine kinase PTK6. Involved in splice site selection of vascular endothelial growth factor. In vitro regulates CD44 alternative splicing by direct binding to purine-rich exonic enhancer (By similarity). Can regulate alternative splicing of neurexins NRXN1-3 in the laminin G-like domain 6 containing the evolutionary conserved neurexin alternative spliced segment 4 (AS4) involved in neurexin selective targeting to postsynaptic partners such as neuroligins and LRRTM family members. Targeted, cell-type specific splicing regulation of NRXN1 at AS4 is involved in neuronal glutamatergic synapse function and plasticity (By similarity). May regulate expression of KHDRBS2/SLIM-1 in defined brain neuron populations by modifying its alternative splicing (By similarity). Can bind FABP9 mRNA (By similarity). May play a role as a negative regulator of cell growth. Inhibits cell proliferation.(Microbial infection) Involved in post-transcriptional regulation of HIV-1 gene expression. Uniprot:O75525 Alternative Names: Etle; etoile; KH domain