Evidence for Local and Global Redox Conditions at an Early Ordovician (Tremadocian) Mass Extinction ∗ ∗∗ Cole T
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Diversity Partitioning During the Cambrian Radiation
Diversity partitioning during the Cambrian radiation Lin Naa,1 and Wolfgang Kiesslinga,b aGeoZentrum Nordbayern, Paleobiology and Paleoenvironments, Friedrich-Alexander-Universität Erlangen-Nürnberg, 91054 Erlangen, Germany; and bMuseum für Naturkunde, Leibniz Institute for Research on Evolution and Biodiversity at the Humboldt University Berlin, 10115 Berlin, Germany Edited by Douglas H. Erwin, Smithsonian National Museum of Natural History, Washington, DC, and accepted by the Editorial Board March 10, 2015 (received for review January 2, 2015) The fossil record offers unique insights into the environmental and Results geographic partitioning of biodiversity during global diversifica- Raw gamma diversity exhibits a strong increase in the first three tions. We explored biodiversity patterns during the Cambrian Cambrian stages (informally referred to as early Cambrian in this radiation, the most dramatic radiation in Earth history. We as- work) (Fig. 1A). Gamma diversity dropped in Stage 4 and de- sessed how the overall increase in global diversity was partitioned clined further through the rest of the Cambrian. The pattern is between within-community (alpha) and between-community (beta) robust to sampling standardization (Fig. 1B) and insensitive to components and how beta diversity was partitioned among environ- including or excluding the archaeocyath sponges, which are po- ments and geographic regions. Changes in gamma diversity in the tentially oversplit (16). Alpha and beta diversity increased from Cambrian were chiefly driven by changes in beta diversity. The the Fortunian to Stage 3, and fluctuated erratically through the combined trajectories of alpha and beta diversity during the initial following stages (Fig. 2). Our estimate of alpha (and indirectly diversification suggest low competition and high predation within beta) diversity is based on the number of genera in published communities. -

Extinction Patterns, Δ18 O Trends, and Magnetostratigraphy from a Southern High-Latitude Cretaceous–Paleogene Section: Links with Deccan Volcanism
Palaeogeography, Palaeoclimatology, Palaeoecology 350–352 (2012) 180–188 Contents lists available at SciVerse ScienceDirect Palaeogeography, Palaeoclimatology, Palaeoecology journal homepage: www.elsevier.com/locate/palaeo Extinction patterns, δ18 O trends, and magnetostratigraphy from a southern high-latitude Cretaceous–Paleogene section: Links with Deccan volcanism Thomas S. Tobin a,⁎, Peter D. Ward a, Eric J. Steig a, Eduardo B. Olivero b, Isaac A. Hilburn c, Ross N. Mitchell d, Matthew R. Diamond c, Timothy D. Raub e, Joseph L. Kirschvink c a University of Washington, Earth and Space Sciences, Box 351310, Seattle WA 98195, United States b CADIC-CONICET, Bernardo Houssay, V9410CAB, Ushuaia, Tierra del Fuego, Argentina c California Institute of Technology, Geological and Planetary Sciences, 1200 E. California Blvd. Pasadena CA 91125, United States d Yale University, Geology & Geophysics, 230 Whitney Ave. New Haven, CT 06511, United States e University of St. Andrews, Department of Earth Sciences, St. Andrews KY16 9AL, UK article info abstract Article history: Although abundant evidence now exists for a massive bolide impact coincident with the Cretaceous–Paleogene Received 19 April 2012 (K–Pg) mass extinction event (~65.5 Ma), the relative importance of this impact as an extinction mechanism is Received in revised form 27 May 2012 still the subject of debate. On Seymour Island, Antarctic Peninsula, the López de Bertodano Formation yields one Accepted 8 June 2012 of the most expanded K–Pg boundary sections known. Using a new chronology from magnetostratigraphy, and Available online 10 July 2012 isotopic data from carbonate-secreting macrofauna, we present a high-resolution, high-latitude paleotemperature record spanning this time interval. -

Estimating Rates of Local Species Extinction, Colonization, and Turnover in Animal Communities
Ecological Applications, 8(4), 1998, pp. 1213±1225 q 1998 by the Ecological Society of America ESTIMATING RATES OF LOCAL SPECIES EXTINCTION, COLONIZATION, AND TURNOVER IN ANIMAL COMMUNITIES JAMES D. NICHOLS,1 THIERRY BOULINIER,2 JAMES E. HINES,1 KENNETH H. POLLOCK,3 AND JOHN R. SAUER1 1U.S. Geological Survey, Biological Resources Division, Patuxent Wildlife Research Center, Laurel, Maryland 20708 USA 2North Carolina Cooperative Fish and Wildlife Research Unit, North Carolina State University, Raleigh, North Carolina 27695 USA 3Institute of Statistics, North Carolina State University, Box 8203, Raleigh, North Carolina 27695-8203 USA Abstract. Species richness has been identi®ed as a useful state variable for conservation and management purposes. Changes in richness over time provide a basis for predicting and evaluating community responses to management, to natural disturbance, and to changes in factors such as community composition (e.g., the removal of a keystone species). Prob- abilistic capture±recapture models have been used recently to estimate species richness from species count and presence±absence data. These models do not require the common assumption that all species are detected in sampling efforts. We extend this approach to the development of estimators useful for studying the vital rates responsible for changes in animal communities over time: rates of local species extinction, turnover, and coloni- zation. Our approach to estimation is based on capture±recapture models for closed animal populations that permit heterogeneity in detection probabilities among the different species in the sampled community. We have developed a computer program, COMDYN, to compute many of these estimators and associated bootstrap variances. -

Addressing the Mammoth in the Room: the Ethical and Political Implications of De- Extinction
Addressing the Mammoth in the room: The ethical and political implications of de- extinction (Ashlock, 2013) Lowieke Vermeulen (S4374452) Political Science: Political Theory Radboud University, Nijmegen, Netherlands Supervisor: prof. dr. Marcel Wissenburg Date: August 12, 2019 Word count: 23590 1 Table of Contents Chapter 1: Introduction...............................................................................................................3 1.2 Thesis structure............................................................................................................................6 Chapter 2: De-extinction and species selection..........................................................8 2.1 Extinction........................................................................................................................................9 2.2 Approaches to de-extinction.................................................................................................10 2.2.1 Back-breeding.........................................................................................................................10 2.2.2 Cloning.......................................................................................................................................12 2.2.3 Genetic engineering..............................................................................................................12 2.2.4 Mixed approaches..................................................................................................................13 2.3 -

International Chronostratigraphic Chart
INTERNATIONAL CHRONOSTRATIGRAPHIC CHART www.stratigraphy.org International Commission on Stratigraphy v 2014/02 numerical numerical numerical Eonothem numerical Series / Epoch Stage / Age Series / Epoch Stage / Age Series / Epoch Stage / Age Erathem / Era System / Period GSSP GSSP age (Ma) GSSP GSSA EonothemErathem / Eon System / Era / Period EonothemErathem / Eon System/ Era / Period age (Ma) EonothemErathem / Eon System/ Era / Period age (Ma) / Eon GSSP age (Ma) present ~ 145.0 358.9 ± 0.4 ~ 541.0 ±1.0 Holocene Ediacaran 0.0117 Tithonian Upper 152.1 ±0.9 Famennian ~ 635 0.126 Upper Kimmeridgian Neo- Cryogenian Middle 157.3 ±1.0 Upper proterozoic Pleistocene 0.781 372.2 ±1.6 850 Calabrian Oxfordian Tonian 1.80 163.5 ±1.0 Frasnian 1000 Callovian 166.1 ±1.2 Quaternary Gelasian 2.58 382.7 ±1.6 Stenian Bathonian 168.3 ±1.3 Piacenzian Middle Bajocian Givetian 1200 Pliocene 3.600 170.3 ±1.4 Middle 387.7 ±0.8 Meso- Zanclean Aalenian proterozoic Ectasian 5.333 174.1 ±1.0 Eifelian 1400 Messinian Jurassic 393.3 ±1.2 7.246 Toarcian Calymmian Tortonian 182.7 ±0.7 Emsian 1600 11.62 Pliensbachian Statherian Lower 407.6 ±2.6 Serravallian 13.82 190.8 ±1.0 Lower 1800 Miocene Pragian 410.8 ±2.8 Langhian Sinemurian Proterozoic Neogene 15.97 Orosirian 199.3 ±0.3 Lochkovian Paleo- Hettangian 2050 Burdigalian 201.3 ±0.2 419.2 ±3.2 proterozoic 20.44 Mesozoic Rhaetian Pridoli Rhyacian Aquitanian 423.0 ±2.3 23.03 ~ 208.5 Ludfordian 2300 Cenozoic Chattian Ludlow 425.6 ±0.9 Siderian 28.1 Gorstian Oligocene Upper Norian 427.4 ±0.5 2500 Rupelian Wenlock Homerian -

2009 Geologic Time Scale Cenozoic Mesozoic Paleozoic Precambrian Magnetic Magnetic Bdy
2009 GEOLOGIC TIME SCALE CENOZOIC MESOZOIC PALEOZOIC PRECAMBRIAN MAGNETIC MAGNETIC BDY. AGE POLARITY PICKS AGE POLARITY PICKS AGE PICKS AGE . N PERIOD EPOCH AGE PERIOD EPOCH AGE PERIOD EPOCH AGE EON ERA PERIOD AGES (Ma) (Ma) (Ma) (Ma) (Ma) (Ma) (Ma) HIST. HIST. ANOM. ANOM. (Ma) CHRON. CHRO HOLOCENE 65.5 1 C1 QUATER- 0.01 30 C30 542 CALABRIAN MAASTRICHTIAN NARY PLEISTOCENE 1.8 31 C31 251 2 C2 GELASIAN 70 CHANGHSINGIAN EDIACARAN 2.6 70.6 254 2A PIACENZIAN 32 C32 L 630 C2A 3.6 WUCHIAPINGIAN PLIOCENE 260 260 3 ZANCLEAN 33 CAMPANIAN CAPITANIAN 5 C3 5.3 266 750 NEOPRO- CRYOGENIAN 80 C33 M WORDIAN MESSINIAN LATE 268 TEROZOIC 3A C3A 83.5 ROADIAN 7.2 SANTONIAN 271 85.8 KUNGURIAN 850 4 276 C4 CONIACIAN 280 4A 89.3 ARTINSKIAN TONIAN C4A L TORTONIAN 90 284 TURONIAN PERMIAN 10 5 93.5 E 1000 1000 C5 SAKMARIAN 11.6 CENOMANIAN 297 99.6 ASSELIAN STENIAN SERRAVALLIAN 34 C34 299.0 5A 100 300 GZELIAN C5A 13.8 M KASIMOVIAN 304 1200 PENNSYL- 306 1250 15 5B LANGHIAN ALBIAN MOSCOVIAN MESOPRO- C5B VANIAN 312 ECTASIAN 5C 16.0 110 BASHKIRIAN TEROZOIC C5C 112 5D C5D MIOCENE 320 318 1400 5E C5E NEOGENE BURDIGALIAN SERPUKHOVIAN 326 6 C6 APTIAN 20 120 1500 CALYMMIAN E 20.4 6A C6A EARLY MISSIS- M0r 125 VISEAN 1600 6B C6B AQUITANIAN M1 340 SIPPIAN M3 BARREMIAN C6C 23.0 345 6C CRETACEOUS 130 M5 130 STATHERIAN CARBONIFEROUS TOURNAISIAN 7 C7 HAUTERIVIAN 1750 25 7A M10 C7A 136 359 8 C8 L CHATTIAN M12 VALANGINIAN 360 L 1800 140 M14 140 9 C9 M16 FAMENNIAN BERRIASIAN M18 PROTEROZOIC OROSIRIAN 10 C10 28.4 145.5 M20 2000 30 11 C11 TITHONIAN 374 PALEOPRO- 150 M22 2050 12 E RUPELIAN -
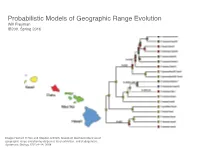
Probabilistic Models of Geographic Range Evolution Will Freyman 10 SYSTEMATIC BIOLOGY VOL
Probabilistic Models of Geographic Range Evolution Will Freyman 10 SYSTEMATIC BIOLOGY VOL. 57 IB200, Spring 2016 Downloaded from http://sysbio.oxfordjournals.org/ at University of California School Law (Boalt Hall) on April 12, 2016 Image: Richard H Ree and Stephen A Smith. Maximum likelihood inference of geographic range evolution by dispersal, local extinction, and cladogenesis. Systematic Biology, 57(1):4–14, 2008. FIGURE 3. Copyedited by: TRJ MANUSCRIPT CATEGORY: Article 2013 LANDIS ET AL.—BAYESIAN BIOGEOGRAPHY FOR MANY AREAS 3 A) B) There are an infinite number of biogeographic 1234011001 010 011 histories that can explain the observed geographic 011 001 ranges. When calculating the probability of the observed Biogeographic histories on geographic ranges at the tips of the phylogenetic tree, 010 a phylogeny: 5 011 it is unreasonable to condition on a specific history 001 101 of biogeographic change. After all, the past history 101 of biogeographic change is not observable. Instead, 6 111 011 the usual approach is to marginalize over all possible 111 histories of biogeographic change that could give rise 7 to the observed geographic ranges. The standard way 111 Downloaded from 101 to do this is to assume that events of colonization or local extinction occur according to a continuous- 8 101 time Markov chain (Ree et al. 2005). Marginalizing over histories of biogeographic change is accomplished C) D) 011101 010 011 011001 010 011 using two procedures. First, exponentiation of the 010 instantaneous-rate matrix, Q, gives the probability http://sysbio.oxfordjournals.org/ 011 010 011 011 011 011 111 111 001 density of all possible biogeographic changes along a 011 010 branch 111 010 101 011 011 111 011 001 Qt 111 101 011 p(y z t,Q) e− , 111 101 → ; = yz 001 % & 101 111 111 011 where y is the ancestral geographic range, z is the 011 001 current geographic range, and t is the duration of the 001 011 branch on the tree. -

The Upper Ordovician Glaciation in Sw Libya – a Subsurface Perspective
J.C. Gutiérrez-Marco, I. Rábano and D. García-Bellido (eds.), Ordovician of the World. Cuadernos del Museo Geominero, 14. Instituto Geológico y Minero de España, Madrid. ISBN 978-84-7840-857-3 © Instituto Geológico y Minero de España 2011 ICE IN THE SAHARA: THE UPPER ORDOVICIAN GLACIATION IN SW LIBYA – A SUBSURFACE PERSPECTIVE N.D. McDougall1 and R. Gruenwald2 1 Repsol Exploración, Paseo de la Castellana 280, 28046 Madrid, Spain. [email protected] 2 REMSA, Dhat El-Imad Complex, Tower 3, Floor 9, Tripoli, Libya. Keywords: Ordovician, Libya, glaciation, Mamuniyat, Melaz Shugran, Hirnantian. INTRODUCTION An Upper Ordovician glacial episode is widely recognized as a significant event in the geological history of the Lower Paleozoic. This is especially so in the case of the Saharan Platform where Upper Ordovician sediments are well developed and represent a major target for hydrocarbon exploration. This paper is a brief summary of the results of fieldwork, in outcrops across SW Libya, together with the analysis of cores, hundreds of well logs (including many high quality image logs) and seismic lines focused on the uppermost Ordovician of the Murzuq Basin. STRATIGRAPHIC FRAMEWORK The uppermost Ordovician section is the youngest of three major sequences recognized widely across the entire Saharan Platform: Sequence CO1: Unconformably overlies the Precambrian or Infracambrian basement. It comprises the possible Upper Cambrian to Lowermost Ordovician Hassaouna Formation. Sequence CO2: Truncates CO1 along a low angle, Type II unconformity. It comprises the laterally extensive and distinctive Lower Ordovician (Tremadocian-Floian?) Achebayat Formation overlain, along a probable transgressive surface of erosion, by interbedded burrowed sandstones, cross-bedded channel-fill sandstones and mudstones of Middle Ordovician age (Dapingian-Sandbian), known as the Hawaz Formation, and interpreted as shallow-marine sediments deposited within a megaestuary or gulf. -

Decoupled Evolution of Soft and Hard Substrate Communities During the Cambrian Explosion and Great Ordovician Biodiversification Event
Decoupled evolution of soft and hard substrate communities during the Cambrian Explosion and Great Ordovician Biodiversification Event Luis A. Buatoisa,1, Maria G. Mánganoa, Ricardo A. Oleab, and Mark A. Wilsonc aDepartment of Geological Sciences, University of Saskatchewan, Saskatoon, SK, Canada S7N 5E2; bEastern Energy Resources Science Center, US Geological Survey, Reston, VA 20192; and cDepartment of Geology, The College of Wooster, Wooster, OH 44691 Edited by Steven M. Holland, University of Georgia, Athens, GA, and accepted by Editorial Board Member David Jablonski May 6, 2016 (received for review November 21, 2015) Contrasts between the Cambrian Explosion (CE) and the Great shed light on the natures of both radiations. Because there is still Ordovician Biodiversification Event (GOBE) have long been recog- controversy regarding Sepkoski’s nonstandardized curves of nized. Whereas the vast majority of body plans were established Phanerozoic taxonomic diversity (13–15), a rarefaction analysis as a result of the CE, taxonomic increases during the GOBE were was performed in an attempt to standardize diversity data. The manifested at lower taxonomic levels. Assessing changes of ichno- aims of this paper are to document the contrasting ichnodiversity diversity and ichnodisparity as a result of these two evolutionary and ichnodisparity trajectories in soft and hard substrate com- events may shed light on the dynamics of both radiations. The early munities during these two evolutionary events and to discuss the Cambrian (series 1 and 2) displayed a dramatic increase in ichnodi- possible underlying causes of this decoupled evolution. versity and ichnodisparity in softground communities. In contrast to this evolutionary explosion in bioturbation structures, only a few Results Cambrian bioerosion structures are known. -

Phanerozoic Phanerozoic & Precambrian
Geologic TimeScale Geologic Time Scale 2012 Foundation ISBN 978-0-44-459425-9 ISBN 978-0-44-459425-9 PHANEROZOIC PHANEROZOIC & PRECAMBRIAN y h 18 t d d d δ i c O SeaSea SeaSea AgeAge o PolarityPolarity o PolarityPolarity o i AgeAgeg AgeAgeg i AgeAgeg o ar LisiekiLisieki & ri a a AgeAge / Stage EpochEpoch AgeAge / Stage levellevel EpochEpoch AgeAge / Stage l levellevel EonEon EraEra PeriodPeriod ((Ma)Ma) p ra er er ChronChron Raymo,Raymo, 2005 CChronhron o (Ma)(Ma) -100- 0 100 200m200m (Ma)(Ma) -100 0 100 200m200m (Ma)(Ma) Era Er Period P Epoch E Era Er Period Pe Era E Period P 3453 4 5 Polarity P 0 00.0118.0118 HoloceneHolocene 2 0 Quat.Quat.PleistocenePleistocene see Quaternary detail C1C1 254.22 ChanghsingianChanggghsingian 2552255 Cenozoic Paleogene TTarantianarantian 4 Plioceneocene Piacenzian/P C2 LopingianLopingian 0.13 5.33 Zanclean 5 C3 259.8WuchiapingianW Cretaceous 6 7.257.25 MessinianMessinian 226060 100 CC44 10 e TortonianTortonian CaCapitanianpitanian Mesozoic Jurassic 11.6311.63 2262655 Guada-Guada- 265.1 200 8 13.8213.82 SerravallianSSllierravallian Triassic C5 lupianlupian 268268.8.8 WoWordianrdian 1010 15 15.9715.97 LanghianLLhianghig an Permian ocen 227070 eogene hes 272.272.33 Roadian 300 ian Carboniferous n BurdigalianBurdigalian Neogene N 1212 Miocene Mi 20.4420.44 u 20 227575 r C6 KKungurianungurian Devonian IonianIonian 23.0323.03 AquitanianAqquitanian rm 400 Paleozoic B 0.50.5 Brunhes 21,000 years Quaternary 2279.379.3 Silurian 1414 2525 228080 255 Ma M Permian P i ChattianChattian C7/9C7/9 Permian Pe Ordovician -

Determining Species Expansion and Extinction Possibilities Using Probabilistic and Graphical Models
Ekológia (Bratislava) Vol. 34, No. 2, p. 111–120, 2015 DOI:10.1515/eko-2015-0012 DetermininG species EXpansion AND EXtinction possibilities UsinG probabilistic AND Graphical models RAJESH CHATURVEDI, P. RAMA CHANDRA PRASAD Lab for Spatial Informatics, International Institute of Information Technology, Gachibowli, Hyderabad - 500032, India; e-mail: [email protected], [email protected] Abstract Chaturvedi R., Prasad R.Ch.: Determining species expansion and extinction possibilities using probabilistic and graphical models. Ekológia (Bratislava), Vol. 34, No. 2, p. 111–120, 2015. Survival of plant species is governed by a number of functions. The participation of each function in species survival and the impact of the contrary behaviour of the species vary from function to function. The probability of extinction of species varies in all such scenarios and has to be calculated separately. Secondly, species follow different patterns of dispersal and localisation at different stages of occupancy state of the site, therefore, the scenarios of competition for resources with climatic shifts leading to deterioration and loss of biodiversity resulting in extinction needs to be studied. Furthermore, most po- ssible deviations of species from climax community states needs to be calculated before species become extinct due to sudden environmental disruption. Globally, various types of anthropogenic disturbances threaten the diversity of biological systems. The impact of these anthropogenic activities needs to be analysed to identify extinction patterns with respect to these activities. All the analyses mentioned abo- ve have been tried to be achieved through probabilistic or graphical models in this study. Key words: biodiversity, disturbances, climate change, endangered, risks, ecological models. -

(Re)Proposal of Three Cambrian Subsystems and Their Geochronology
Article 273 by Ed Landing1*, Gerd Geyer2, Mark D. Schmitz3, Thomas Wotte4, and Artem Kouchinsky5 (Re)proposal of three Cambrian Subsystems and their Geochronology 1 New York State Museum, 222 Madison Avenue, Albany, NY, USA; *Corresponding author, E-mail: [email protected] 2 Lehrstuhl für Geodynamik und Geomaterialforschung, Institut für Geographie und Geologie, Bayerische Julius-Maximilians Universität Würzburg, Am Hubland, D-97074 Würzburg, Germany 3 Department of Geosciences, Boise State University, 1910 University Drive, Boise, Idaho 83725, USA 4 Department of Palaeontology, TU Bergakademie Freiberg, Bernhard-von-Cotta-Straße, D-09599 Freiberg, Germany 5 Department of Palaeontology, Swedish Museum of Natural History, Box 50007, SE-104 05, Stockholm, Sweden (Received: April 29, 2020; Revised accepted: September 24, 2020) https://doi.org/10.18814/epiiugs/2020/020088 The Cambrian is anomalous among geological systems 2018). Following work by the International Subcommission on Cam- as many reports divide it into three divisions of indetermi- brian Stratigraphy (ISCS), these key biotic developments can be related nate rank. This use of “lower”, “middle”, and “upper” has to the chronostratigraphic subdivision of the Cambrian System into been a convenient way to subdivide the Cambrian despite four global series and ten stages (e.g., Babcock, 2005; Babcock and agreement it consists of four global series. Traditional divi- Peng, 2007) complemented by an increasingly precise U-Pb (numeri- sions of the system into regional series (Lower, Middle, cal) geochronology (Fig. 1). This decision at the 2004 ISCS meeting Upper) reflected local biotic developments not interpro- meant that the traditional subdivisions of the Cambrian into regional “Lower,” “Middle,” and “Upper” series (discussed below) were no lon- vincially correlatable with any precision.