Fetal Hemoglobin in Sickle Hemoglobinopathies: High Hbf Genotypes and Phenotypes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Adult, Embryonic and Fetal Hemoglobin Are Expressed in Human Glioblastoma Cells
514 INTERNATIONAL JOURNAL OF ONCOLOGY 44: 514-520, 2014 Adult, embryonic and fetal hemoglobin are expressed in human glioblastoma cells MARWAN EMARA1,2, A. ROBERT TURNER1 and JOAN ALLALUNIS-TURNER1 1Department of Oncology, University of Alberta and Alberta Health Services, Cross Cancer Institute, Edmonton, AB T6G 1Z2, Canada; 2Center for Aging and Associated Diseases, Zewail City of Science and Technology, Cairo, Egypt Received September 7, 2013; Accepted October 7, 2013 DOI: 10.3892/ijo.2013.2186 Abstract. Hemoglobin is a hemoprotein, produced mainly in Introduction erythrocytes circulating in the blood. However, non-erythroid hemoglobins have been previously reported in other cell Globins are hemo-containing proteins, have the ability to types including human and rodent neurons of embryonic bind gaseous ligands [oxygen (O2), nitric oxide (NO) and and adult brain, but not astrocytes and oligodendrocytes. carbon monoxide (CO)] reversibly. They have been described Human glioblastoma multiforme (GBM) is the most aggres- in prokaryotes, fungi, plants and animals with an enormous sive tumor among gliomas. However, despite extensive basic diversity of structure and function (1). To date, hemoglobin, and clinical research studies on GBM cells, little is known myoglobin, neuroglobin (Ngb) and cytoglobin (Cygb) repre- about glial defence mechanisms that allow these cells to sent the vertebrate globin family with distinct function and survive and resist various types of treatment. We have tissue distributions (2). During ontogeny, developing erythro- shown previously that the newest members of vertebrate blasts sequentially express embryonic {[Gower 1 (ζ2ε2), globin family, neuroglobin (Ngb) and cytoglobin (Cygb), are Gower 2 (α2ε2), and Portland 1 (ζ2γ2)] to fetal [Hb F(α2γ2)] expressed in human GBM cells. -

Identification of Novel HPFH-Like Mutations by CRISPR Base Editing That Elevates the Expression of Fetal Hemoglobin
bioRxiv preprint doi: https://doi.org/10.1101/2020.06.30.178715; this version posted July 1, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Identification of novel HPFH-like mutations by CRISPR base editing that elevates the expression of fetal hemoglobin Nithin Sam Ravi1,3, Beeke Wienert4,8,9, Stacia K. Wyman4, Jonathan Vu4, Aswin Anand Pai2, Poonkuzhali Balasubramanian2, Yukio Nakamura7, Ryo Kurita10, Srujan Marepally1, Saravanabhavan Thangavel1, Shaji R. Velayudhan1,2, Alok Srivastava1,2, Mark A. DeWitt 4,5, Jacob E. Corn4,6, Kumarasamypet M. Mohankumar1* 1. Centre for Stem Cell Research (a unit of inStem, Bengaluru), Christian Medical College Campus, Bagayam, Vellore-632002, Tamil Nadu, India. 2. Department of Haematology, Christian Medical College Hospital, IDA Scudder Rd, Vellore-632002, Tamil Nadu, India. 3. Sree Chitra Tirunal Institute for Medical Sciences and Technology, Thiruvananthapuram - 695 011, Kerala, India. 4. Innovative Genomics Institute, University of California Berkeley, Berkeley, California- 94704, USA. 5. Department of Microbiology, Immunology, and Molecular Genetics, University of California - Los Angeles, Los Angeles, California - 90095, USA. 6. Institute of Molecular Health Sciences, Department of Biology, ETH Zurich, Otto-Stern- Weg, Zurich - 8092, Switzerland. 7. Cell Engineering Division, RIKEN BioResource Center, Tsukuba, Ibaraki 305-0074, Japan. 8. Institute of Data Science and Biotechnology, Gladstone Institutes, San Francisco, California -94158, USA 9. School of Biotechnology and Biomolecular Sciences, University of New South Wales - 2052, Sydney, Australia. 10. Research and Development Department Central Blood Institute Blood Service Headquarters Japanese Red Cross Society, 2-1-67 Tatsumi, Tokyo, 135-8521, Japan *Corresponding Author: Dr. -

Apoptotic Cells Inflammasome Activity During the Uptake of Macrophage
Downloaded from http://www.jimmunol.org/ by guest on September 29, 2021 is online at: average * The Journal of Immunology , 26 of which you can access for free at: 2012; 188:5682-5693; Prepublished online 20 from submission to initial decision 4 weeks from acceptance to publication April 2012; doi: 10.4049/jimmunol.1103760 http://www.jimmunol.org/content/188/11/5682 Complement Protein C1q Directs Macrophage Polarization and Limits Inflammasome Activity during the Uptake of Apoptotic Cells Marie E. Benoit, Elizabeth V. Clarke, Pedro Morgado, Deborah A. Fraser and Andrea J. Tenner J Immunol cites 56 articles Submit online. Every submission reviewed by practicing scientists ? is published twice each month by Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts http://jimmunol.org/subscription http://www.jimmunol.org/content/suppl/2012/04/20/jimmunol.110376 0.DC1 This article http://www.jimmunol.org/content/188/11/5682.full#ref-list-1 Information about subscribing to The JI No Triage! Fast Publication! Rapid Reviews! 30 days* Why • • • Material References Permissions Email Alerts Subscription Supplementary The Journal of Immunology The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2012 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. This information is current as of September 29, 2021. The Journal of Immunology Complement Protein C1q Directs Macrophage Polarization and Limits Inflammasome Activity during the Uptake of Apoptotic Cells Marie E. -

Genome Editing of HBG1/2 Promoter Leads to Robust Hbf Induction in Vivo While Editing of BCL11A Erythroid Enhancer Shows Erythroid Defect
Genome Editing of HBG1/2 Promoter Leads to Robust HbF Induction In Vivo While Editing of BCL11A Erythroid Enhancer Shows Erythroid Defect Kai-Hsin Chang, Minerva Sanchez, Jack Heath, Edouard de Dreuzy, Scott Haskett, Abigail Vogelaar, Kiran Gogi, Jen DaSilva, Tongyao Wang, Andrew Sadowski, Gregory Gotta, Jamaica Siwak, Ramya Viswanathan, Katherine Loveluck, Hoson Chao, Eric Tillotson, Aditi Chalishazar, Abhishek Dass, Frederick Ta, Emily Brennan, Diana Tabbaa, Eugenio Marco, John Zuris, Deepak Reyon, Meltem Isik, Ari Friedland, Terence Ta, Fred Harbinski, Georgia Giannoukos, Sandra Teixeira, Christopher Wilson, Charlie Albright, Haiyan Jiang Editas Medicine, Inc., Cambridge, MA 60th Annual Meeting and Exposition of American Society of Hematology December 2, 2018, San Diego, CA © 2018 Editas Medicine Overview Etiology of Sickle Cell Disease In Vivo Study Design to Evaluate Two Approaches to Increase Fetal Hemoglobin (HbF) Expression Effect of Downregulating BCL11A Expression by Targeting its Erythroid Enhancer Editing Cis-regulatory Elements in b-Globin Locus Conclusion © 2018 Editas Medicine Etiology of Sickle Cell Disease Amino Hemoglobin Low DNA Acid Tetramer O2 a a a a b-globin GLU G A G b b b b HbA a a a a a a a a s s s VAL b b b -globin G T G bs bs bs bs bs bs HbS HbS Fiber • Sickle cell disease (SCD) is caused by a single mutation E6V of the b-globin chain, leading to polymerization of hemoglobin (Hb) and formation of sickle hemoglobin (HbS) fibers when deoxygenated. • Symptoms include anemia, acute chest syndrome, pain crises, and an array of other complications. • Patients suffer significant morbidity and early mortality. -

Identifying Genetic Variants and Pathways Associated with Extreme Levels of Fetal Hemoglobin in Sickle Cell Disease in Tanzania
Nkya et al. BMC Medical Genetics (2020) 21:125 https://doi.org/10.1186/s12881-020-01059-1 RESEARCH ARTICLE Open Access Identifying genetic variants and pathways associated with extreme levels of fetal hemoglobin in sickle cell disease in Tanzania Siana Nkya1,2*, Liberata Mwita2, Josephine Mgaya2, Happiness Kumburu3, Marco van Zwetselaar3, Stephan Menzel4, Gaston Kuzamunu Mazandu5,6,7* , Raphael Sangeda2,8, Emile Chimusa5 and Julie Makani2 Abstract Background: Sickle cell disease (SCD) is a blood disorder caused by a point mutation on the beta globin gene resulting in the synthesis of abnormal hemoglobin. Fetal hemoglobin (HbF) reduces disease severity, but the levels vary from one individual to another. Most research has focused on common genetic variants which differ across populations and hence do not fully account for HbF variation. Methods: We investigated rare and common genetic variants that influence HbF levels in 14 SCD patients to elucidate variants and pathways in SCD patients with extreme HbF levels (≥7.7% for high HbF) and (≤2.5% for low HbF) in Tanzania. We performed targeted next generation sequencing (Illumina_Miseq) covering exonic and other significant fetal hemoglobin-associated loci, including BCL11A, MYB, HOXA9, HBB, HBG1, HBG2, CHD4, KLF1, MBD3, ZBTB7A and PGLYRP1. Results: Results revealed a range of genetic variants, including bi-allelic and multi-allelic SNPs, frameshift insertions and deletions, some of which have functional importance. Notably, there were significantly more deletions in individuals with high HbF levels (11% vs 0.9%). We identified frameshift deletions in individuals with high HbF levels and frameshift insertions in individuals with low HbF. CHD4 and MBD3 genes, interacting in the same sub-network, were identified to have a significant number of pathogenic or non-synonymous mutations in individuals with low HbF levels, suggesting an important role of epigenetic pathways in the regulation of HbF synthesis. -

Impact of Rare and Common Genetic Variants on Diabetes Diagnosis by Hemoglobin A1c in Multi-Ancestry Cohorts
bioRxiv preprint doi: https://doi.org/10.1101/643932; this version posted May 28, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. 1 Impact of rare and common genetic variants on diabetes 2 diagnosis by hemoglobin A1c in multi-ancestry cohorts: 3 The Trans-Omics for Precision Medicine Program 4 5 Authors 6 Chloé Sarnowski,1,35,37,* Aaron Leong,2,3,4,35,38,** Laura M Raffield,5 Peitao Wu,1 Paul S de 7 Vries,6 Daniel DiCorpo,1 Xiuqing Guo,7 Huichun Xu,8 Yongmei Liu,9 Xiuwen Zheng,10 Yao 8 Hu,11 Jennifer A Brody,12 Mark O Goodarzi,13 Bertha A Hidalgo,14 Heather M Highland,15 9 Deepti Jain,10 Ching-Ti Liu,1 Rakhi P Naik,16 James A Perry,17 Bianca C Porneala,2 Elizabeth 10 Selvin,18 Jennifer Wessel,19,20 Bruce M Psaty,12,21,22 Joanne E Curran,23 Juan M Peralta,23 John 11 Blangero,23 Charles Kooperberg,11 Rasika Mathias,18,24 Andrew D Johnson,25,26 Alexander P 12 Reiner,11,27 Braxton D Mitchell,8,28 L Adrienne Cupples,1,25 Ramachandran S Vasan,25,29,30 13 Adolfo Correa,31,32 Alanna C Morrison,6 Eric Boerwinkle,6,33 Jerome I Rotter,7 Stephen S Rich,34 14 Alisa K Manning,2,3,4 Josée Dupuis,1,25,36 James B Meigs,2,3,4,36 on behalf of the Trans-Omics for 15 Precision Medicine (TOPMed) Diabetes and TOPMed Hematology and Hemostasis working 16 groups and the NHLBI TOPMed Consortium. -
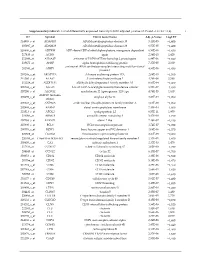
Supplementary Table S1. List of Differentially Expressed
Supplementary table S1. List of differentially expressed transcripts (FDR adjusted p‐value < 0.05 and −1.4 ≤ FC ≥1.4). 1 ID Symbol Entrez Gene Name Adj. p‐Value Log2 FC 214895_s_at ADAM10 ADAM metallopeptidase domain 10 3,11E‐05 −1,400 205997_at ADAM28 ADAM metallopeptidase domain 28 6,57E‐05 −1,400 220606_s_at ADPRM ADP‐ribose/CDP‐alcohol diphosphatase, manganese dependent 6,50E‐06 −1,430 217410_at AGRN agrin 2,34E‐10 1,420 212980_at AHSA2P activator of HSP90 ATPase homolog 2, pseudogene 6,44E‐06 −1,920 219672_at AHSP alpha hemoglobin stabilizing protein 7,27E‐05 2,330 aminoacyl tRNA synthetase complex interacting multifunctional 202541_at AIMP1 4,91E‐06 −1,830 protein 1 210269_s_at AKAP17A A‐kinase anchoring protein 17A 2,64E‐10 −1,560 211560_s_at ALAS2 5ʹ‐aminolevulinate synthase 2 4,28E‐06 3,560 212224_at ALDH1A1 aldehyde dehydrogenase 1 family member A1 8,93E‐04 −1,400 205583_s_at ALG13 ALG13 UDP‐N‐acetylglucosaminyltransferase subunit 9,50E‐07 −1,430 207206_s_at ALOX12 arachidonate 12‐lipoxygenase, 12S type 4,76E‐05 1,630 AMY1C (includes 208498_s_at amylase alpha 1C 3,83E‐05 −1,700 others) 201043_s_at ANP32A acidic nuclear phosphoprotein 32 family member A 5,61E‐09 −1,760 202888_s_at ANPEP alanyl aminopeptidase, membrane 7,40E‐04 −1,600 221013_s_at APOL2 apolipoprotein L2 6,57E‐11 1,600 219094_at ARMC8 armadillo repeat containing 8 3,47E‐08 −1,710 207798_s_at ATXN2L ataxin 2 like 2,16E‐07 −1,410 215990_s_at BCL6 BCL6 transcription repressor 1,74E‐07 −1,700 200776_s_at BZW1 basic leucine zipper and W2 domains 1 1,09E‐06 −1,570 222309_at -

Hemoglobin Binding to Aß and HBG2 SNP Association Suggest a Role In
Neurobiology of Aging 29 (2008) 185–193 Hemoglobin binding to A and HBG2 SNP association suggest a role in Alzheimer’s disease Rodney T. Perry a,∗, Debra A. Gearhart b, Howard W. Wiener a, Lindy E. Harrell c, James C. Barton d, Abdullah Kutlar e, Ferdane Kutlar e, Ozan Ozcan f,g, Rodney C.P. Go a, William D. Hill f,h,i,∗∗ a Department of Epidemiology, University of Alabama at Birmingham, Birmingham, AL 35294-0022, USA b Department of Pharmacology & Toxicology, Medical College of Georgia, Augusta, GA 30912-2300, USA c Alzheimer’s Disease Center, Department of Neurology, University of Alabama at Birmingham, Birmingham, AL 35294-0017, USA d Southern Iron Disorders Center, Birmingham, AL 35209, USA e Department of Medicine, Medical College of Georgia, Augusta, GA 30912-2000, USA f Department of Cellular Biology & Anatomy, Medical College of Georgia, Augusta, GA 30912-2000, USA g Department of Biology, Paine College, Augusta, GA 30912-2000, USA h Department of Neurology, Institute of Molecular Medicine and Genetics, Medical College of Georgia, Augusta, GA 30912-2000, USA i Augusta Veterans Administration Medical Center, Augusta, GA 30912-2000, USA Received 16 March 2006; received in revised form 4 August 2006; accepted 11 October 2006 Available online 8 December 2006 Abstract From a normal human brain phage display library screen we identified the gamma (A)-globin chain of fetal hemoglobin (Hb F) as a protein that bound strongly to A1-42. We showed the oxidized form of adult Hb (metHb A) binds with greater affinity to A1-42 than metHb F. -

3'HS1 CTCF Binding Site in Human Β-Globin Locus Regulates Fetal
bioRxiv preprint doi: https://doi.org/10.1101/2021.05.18.444713; this version posted May 18, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. 3’HS1 CTCF binding site in human β-globin locus regulates fetal hemoglobin expression Pamela Himadewi1,8, Xue Qing David Wang1,8, Fan Feng3,8, Haley Gore1, Yushuai Liu1, Lei Yu2, Jie Liu3, Ryo Kurita4, Yukio Nakamura5,6, Gerd Pfeifer1, and Xiaotian Zhang1,7. 1. Center for Epigenetics, Van Andel Research Institute, Grand Rapids, MI, USA 2. Cell and Development Biology, University of Michigan, Ann Arbor, MI, USA 3. Department of Computational Biology, University of Michigan, Ann Arbor, MI, USA 4. Department of Research and Development, Central Blood Institute, Japanese Red Cross Society,Tokyo,Japan. 5. Cell Engineering Division, RIKEN BioResource Research Center, Tsukuba, Japan 6. Faculty of Medicine, University of Tsukuba, Tsukuba, Japan 7. Current Address: Department of Pathology, University of Michigan 8. These authors contributed equally to the work Correspondence should be directed to: Xiaotian Zhang [email protected] bioRxiv preprint doi: https://doi.org/10.1101/2021.05.18.444713; this version posted May 18, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Summary Mutations in the adult β-globin gene can lead to a variety of hemoglobinopathies, including sickle cell disease and β-thalassemia. -

The Normal Structure and Regulation of Human Globin Gene Clusters
CHAPTER 3 The Normal Structure and Regulation of Human Globin Gene Clusters Bernard G. Forget and Ross C. Hardison The genes encoding the different globin chains of hemoglobin are members of an ancient gene family. In this chapter we will review the structural features of the globin genes, with particular attention to the sequences needed for proper regulation of gene expression. Some of these have been well- conserved during mammalian evolution and therefore are likely to provide a common function in many mammals. Others are only found in higher primates, and may play roles in lineage-specific regulation. We will first describe the structural characteristics of the human globin genes and then provide a comparative analysis of the genomic contexts, regulatory regions and evolutionary conservation of features present in the globin gene clusters. NUMBER AND CHROMOSOMAL LOCALIZATION OF HUMAN GLOBIN GENES Hemoglobin is a heterotetramer that contains two polypeptide subunits related to the α-globin gene subfamily (referred to here as α-like globins) and two polypeptide subunits related to the β-globin gene subfamily (β-like globins). Globin polypeptides bind heme, which in turn allows the hemoglobin in erythrocytes to bind oxygen reversibly and transport it from the lungs to respiring tissues. In humans, as in all vertebrate species studied, different α-like and β-like globin chains are synthesized at Chapter 3 The Normal Structure and Regulation of the Globin Gene Clusters progressive stages of development to produce hemoglobins characteristic of primitive (embryonic) and definitive (fetal and adult) erythroid cells (Figure 3.1). Before precise knowledge of globin gene organization was gained by gene mapping and molecular cloning, a general picture of the number and arrangement of the human globin genes emerged from the genetic analysis of normal and abnormal hemoglobins and their pattern of inheritance. -

Evolution of Hemoglobin and Its Genes
Downloaded from http://perspectivesinmedicine.cshlp.org/ on October 2, 2021 - Published by Cold Spring Harbor Laboratory Press Evolution of Hemoglobin and Its Genes Ross C. Hardison Center for Comparative Genomics and Bioinformatics, Huck Institute of Genome Sciences, Department of Biochemistry and Molecular Biology, The Pennsylvania State University, University Park, Pennsylvania 16802 Correspondence: [email protected] Insights into the evolution of hemoglobins and their genes are an abundant source of ideas regarding hemoglobin function and regulation of globin gene expression. This article pre- sents the multiple genes and gene families encoding human globins, summarizes major events in the evolution of the hemoglobin gene clusters, and discusses how these studies provide insights into regulation of globin genes. Although the genes in and around the a-like globin gene complex are relatively stable, the b-like globin gene clusters are more dynamic, showing evidence of transposition to a new locus and frequent lineage-specific expansions and deletions. The cis-regulatory modules controlling levels and timing of gene expression are a mix of conserved and lineage-specific DNA, perhaps reflecting evolutionary constraint on core regulatory functions shared broadly in mammals and adaptive fine-tuning in different orders of mammals. wide range of animals, vertebrate and inver- globin genes, and attempts to modulate globin Atebrate, use hemoglobins to transport oxy- gene expression are a fundamental approach to gen, carrying it from lungs, gills, or -

Thalassemia, a Human Blood Disorder
This is a repository copy of Thalassemia, a human blood disorder. White Rose Research Online URL for this paper: https://eprints.whiterose.ac.uk/177841/ Version: Published Version Article: Shafique, F., Ali, S., Almansouri, T.S. et al. (6 more authors) (2021) Thalassemia, a human blood disorder. Brazilian Journal of Biology, 83. e246062. ISSN 1519-6984 https://doi.org/10.1590/1519-6984.246062 Reuse This article is distributed under the terms of the Creative Commons Attribution (CC BY) licence. This licence allows you to distribute, remix, tweak, and build upon the work, even commercially, as long as you credit the authors for the original work. More information and the full terms of the licence here: https://creativecommons.org/licenses/ Takedown If you consider content in White Rose Research Online to be in breach of UK law, please notify us by emailing [email protected] including the URL of the record and the reason for the withdrawal request. [email protected] https://eprints.whiterose.ac.uk/ ISSN 1519-6984 (Print) ISSN 1678-4375 (Online) Original Article Thalassemia, a human blood disorder Talassemia, uma doença do sangue humano F. Shafiquea,b , S. Alic , T. Almansourid,e , F. Van Eedenb , N. Shafia , M. Khalidf , S. Khawajag , S. Andleeba and M. ul Hassanh aUniversity of Azad Jammu and Kashmir, Faculty of Science, Department of Zoology, Muzaffarabad, Pakistan bUniversity of Sheffield, Faculty of Science, Department of Biomedical Science, Sheffield, United Kingdom cGovernment College University Lahore, Faculty of Science,