Identification of Novel HPFH-Like Mutations by CRISPR Base Editing That Elevates the Expression of Fetal Hemoglobin
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hypoxia-Induced Alpha-Globin Expression in Syncytiotrophoblasts Mimics the Pattern Observed in Preeclamptic Placentas
International Journal of Molecular Sciences Article Hypoxia-Induced Alpha-Globin Expression in Syncytiotrophoblasts Mimics the Pattern Observed in Preeclamptic Placentas Zahra Masoumi 1,* , Lena Erlandsson 1, Eva Hansson 1, Mattias Magnusson 2, Eva Mezey 3 and Stefan R. Hansson 1,4 1 Department of Clinical Sciences Lund, Division of Obstetrics and Gynecology, Lund University, SE-22184 Lund, Sweden; [email protected] (L.E.); [email protected] (E.H.); [email protected] (S.R.H.) 2 Department of Molecular Medicine and Gene Therapy, Lund University, SE-22184 Lund, Sweden; [email protected] 3 Adult Stem Cell Section, National Institute of Dental and Craniofacial Research, National Institutes of Health, 9000 Rockville Pike, Bethesda, MD 20892, USA; [email protected] 4 Skåne University Hospital, SE-22184 Lund, Sweden * Correspondence: [email protected] Abstract: Preeclampsia (PE) is a pregnancy disorder associated with placental dysfunction and elevated fetal hemoglobin (HbF). Early in pregnancy the placenta harbors hematopoietic stem and progenitor cells (HSPCs) and is an extramedullary source of erythropoiesis. However, globin ex- pression is not unique to erythroid cells and can be triggered by hypoxia. To investigate the role of the placenta in increasing globin levels previously reported in PE, flow cytometry, histological and Citation: Masoumi, Z.; Erlandsson, immunostaining and in situ analyses were used on placenta samples and ex vivo explant cultures. L.; Hansson, E.; Magnusson, M.; Our results indicated that in PE pregnancies, placental HSPC homing and erythropoiesis were not Mezey, E.; Hansson, S.R. affected. Non-erythroid alpha-globin mRNA and protein, but not gamma-globin, were detected in Hypoxia-Induced Alpha-Globin Expression in Syncytiotrophoblasts syncytiotrophoblasts and stroma of PE placenta samples. -

1 Diocese of Harrisburg Geography of Pennsylvania
7/2006 DIOCESE OF HARRISBURG GEOGRAPHY OF PENNSYLVANIA Red Italics – Reference to Diocesan History Outcome: The student knows and understands the geography of Pennsylvania. Assessment: The student will apply the geographic themes of location, place and region to Pennsylvania today. Skills/Objectives Suggested Teaching/Learning Strategies Suggested Assessment Strategies The student will be able to: 1a. Using student desk maps, locate and highlight the 1a. On a blank map of the U.S., outline the state of state, trace rivers and their tributaries and circle Pennsylvania, label the capital, hometown and two 1. Locate and identify the state of Pennsylvania, its major cities. largest cities. capital, major cities and his hometown. On a blank map of the U.S. outline the state of 1b. Using the globe, determine the latitude and longitude Pennsylvania; within the state of Pennsylvania, outline of the state. the Diocese of Harrisburg. Label the city where the cathedral is located. 1c. Using a Pennsylvania highway map, design a road tour visiting major Pennsylvania cities. 1b. Using the desk map, identify the latitude line closest Using a Pennsylvania highway map with the counties to the hometown and two other towns of the same within the Diocese of Harrisburg marked, do the parallel of latitude. Identify the longitude nearest the following: first, in each county, identify a city or town state capital and two other towns on the same with a Catholic church; then design a road tour visiting meridian of longitude. each of these cities or towns. Using a desk map, place marks on the most-western, the most-northern, and the most-eastern tips of the Diocese 1d. -

Reception of Low Frequency Time Signals
Reprinted from I-This reDort show: the Dossibilitks of clock svnchronization using time signals I 9 transmitted at low frequencies. The study was madr by obsirvins pulses Vol. 6, NO. 9, pp 13-21 emitted by HBC (75 kHr) in Switxerland and by WWVB (60 kHr) in tha United States. (September 1968), The results show that the low frequencies are preferable to the very low frequencies. Measurementi show that by carefully selecting a point on the decay curve of the pulse it is possible at distances from 100 to 1000 kilo- meters to obtain time measurements with an accuracy of +40 microseconds. A comparison of the theoretical and experimental reiulb permib the study of propagation conditions and, further, shows the drsirability of transmitting I seconds pulses with fixed envelope shape. RECEPTION OF LOW FREQUENCY TIME SIGNALS DAVID H. ANDREWS P. E., Electronics Consultant* C. CHASLAIN, J. DePRlNS University of Brussels, Brussels, Belgium 1. INTRODUCTION parisons of atomic clocks, it does not suffice for clock For several years the phases of VLF and LF carriers synchronization (epoch setting). Presently, the most of standard frequency transmitters have been monitored accurate technique requires carrying portable atomic to compare atomic clock~.~,*,3 clocks between the laboratories to be synchronized. No matter what the accuracies of the various clocks may be, The 24-hour phase stability is excellent and allows periodic synchronization must be provided. Actually frequency calibrations to be made with an accuracy ap- the observed frequency deviation of 3 x 1o-l2 between proaching 1 x 10-11. It is well known that over a 24- cesium controlled oscillators amounts to a timing error hour period diurnal effects occur due to propagation of about 100T microseconds, where T, given in years, variations. -

Serum Albumin OS=Homo Sapiens
Protein Name Cluster of Glial fibrillary acidic protein OS=Homo sapiens GN=GFAP PE=1 SV=1 (P14136) Serum albumin OS=Homo sapiens GN=ALB PE=1 SV=2 Cluster of Isoform 3 of Plectin OS=Homo sapiens GN=PLEC (Q15149-3) Cluster of Hemoglobin subunit beta OS=Homo sapiens GN=HBB PE=1 SV=2 (P68871) Vimentin OS=Homo sapiens GN=VIM PE=1 SV=4 Cluster of Tubulin beta-3 chain OS=Homo sapiens GN=TUBB3 PE=1 SV=2 (Q13509) Cluster of Actin, cytoplasmic 1 OS=Homo sapiens GN=ACTB PE=1 SV=1 (P60709) Cluster of Tubulin alpha-1B chain OS=Homo sapiens GN=TUBA1B PE=1 SV=1 (P68363) Cluster of Isoform 2 of Spectrin alpha chain, non-erythrocytic 1 OS=Homo sapiens GN=SPTAN1 (Q13813-2) Hemoglobin subunit alpha OS=Homo sapiens GN=HBA1 PE=1 SV=2 Cluster of Spectrin beta chain, non-erythrocytic 1 OS=Homo sapiens GN=SPTBN1 PE=1 SV=2 (Q01082) Cluster of Pyruvate kinase isozymes M1/M2 OS=Homo sapiens GN=PKM PE=1 SV=4 (P14618) Glyceraldehyde-3-phosphate dehydrogenase OS=Homo sapiens GN=GAPDH PE=1 SV=3 Clathrin heavy chain 1 OS=Homo sapiens GN=CLTC PE=1 SV=5 Filamin-A OS=Homo sapiens GN=FLNA PE=1 SV=4 Cytoplasmic dynein 1 heavy chain 1 OS=Homo sapiens GN=DYNC1H1 PE=1 SV=5 Cluster of ATPase, Na+/K+ transporting, alpha 2 (+) polypeptide OS=Homo sapiens GN=ATP1A2 PE=3 SV=1 (B1AKY9) Fibrinogen beta chain OS=Homo sapiens GN=FGB PE=1 SV=2 Fibrinogen alpha chain OS=Homo sapiens GN=FGA PE=1 SV=2 Dihydropyrimidinase-related protein 2 OS=Homo sapiens GN=DPYSL2 PE=1 SV=1 Cluster of Alpha-actinin-1 OS=Homo sapiens GN=ACTN1 PE=1 SV=2 (P12814) 60 kDa heat shock protein, mitochondrial OS=Homo -

Adult, Embryonic and Fetal Hemoglobin Are Expressed in Human Glioblastoma Cells
514 INTERNATIONAL JOURNAL OF ONCOLOGY 44: 514-520, 2014 Adult, embryonic and fetal hemoglobin are expressed in human glioblastoma cells MARWAN EMARA1,2, A. ROBERT TURNER1 and JOAN ALLALUNIS-TURNER1 1Department of Oncology, University of Alberta and Alberta Health Services, Cross Cancer Institute, Edmonton, AB T6G 1Z2, Canada; 2Center for Aging and Associated Diseases, Zewail City of Science and Technology, Cairo, Egypt Received September 7, 2013; Accepted October 7, 2013 DOI: 10.3892/ijo.2013.2186 Abstract. Hemoglobin is a hemoprotein, produced mainly in Introduction erythrocytes circulating in the blood. However, non-erythroid hemoglobins have been previously reported in other cell Globins are hemo-containing proteins, have the ability to types including human and rodent neurons of embryonic bind gaseous ligands [oxygen (O2), nitric oxide (NO) and and adult brain, but not astrocytes and oligodendrocytes. carbon monoxide (CO)] reversibly. They have been described Human glioblastoma multiforme (GBM) is the most aggres- in prokaryotes, fungi, plants and animals with an enormous sive tumor among gliomas. However, despite extensive basic diversity of structure and function (1). To date, hemoglobin, and clinical research studies on GBM cells, little is known myoglobin, neuroglobin (Ngb) and cytoglobin (Cygb) repre- about glial defence mechanisms that allow these cells to sent the vertebrate globin family with distinct function and survive and resist various types of treatment. We have tissue distributions (2). During ontogeny, developing erythro- shown previously that the newest members of vertebrate blasts sequentially express embryonic {[Gower 1 (ζ2ε2), globin family, neuroglobin (Ngb) and cytoglobin (Cygb), are Gower 2 (α2ε2), and Portland 1 (ζ2γ2)] to fetal [Hb F(α2γ2)] expressed in human GBM cells. -
Technical Note, Appendix: an Analysis of Blood Processing Methods to Prepare Samples for Genechip® Expression Profiling (Pdf, 1
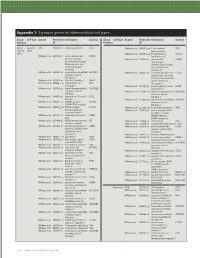
Appendix 1: Signature genes for different blood cell types. Blood Cell Type Source Probe Set Description Symbol Blood Cell Type Source Probe Set Description Symbol Fraction ID Fraction ID Mono- Lympho- GSK 203547_at CD4 antigen (p55) CD4 Whitney et al. 209813_x_at T cell receptor TRG nuclear cytes gamma locus cells Whitney et al. 209995_s_at T-cell leukemia/ TCL1A Whitney et al. 203104_at colony stimulating CSF1R lymphoma 1A factor 1 receptor, Whitney et al. 210164_at granzyme B GZMB formerly McDonough (granzyme 2, feline sarcoma viral cytotoxic T-lymphocyte- (v-fms) oncogene associated serine homolog esterase 1) Whitney et al. 203290_at major histocompatibility HLA-DQA1 Whitney et al. 210321_at similar to granzyme B CTLA1 complex, class II, (granzyme 2, cytotoxic DQ alpha 1 T-lymphocyte-associated Whitney et al. 203413_at NEL-like 2 (chicken) NELL2 serine esterase 1) Whitney et al. 203828_s_at natural killer cell NK4 (H. sapiens) transcript 4 Whitney et al. 212827_at immunoglobulin heavy IGHM Whitney et al. 203932_at major histocompatibility HLA-DMB constant mu complex, class II, Whitney et al. 212998_x_at major histocompatibility HLA-DQB1 DM beta complex, class II, Whitney et al. 204655_at chemokine (C-C motif) CCL5 DQ beta 1 ligand 5 Whitney et al. 212999_x_at major histocompatibility HLA-DQB Whitney et al. 204661_at CDW52 antigen CDW52 complex, class II, (CAMPATH-1 antigen) DQ beta 1 Whitney et al. 205049_s_at CD79A antigen CD79A Whitney et al. 213193_x_at T cell receptor beta locus TRB (immunoglobulin- Whitney et al. 213425_at Homo sapiens cDNA associated alpha) FLJ11441 fis, clone Whitney et al. 205291_at interleukin 2 receptor, IL2RB HEMBA1001323, beta mRNA sequence Whitney et al. -

Molecular Epidemiology and Hematologic Characterization of Δβ
Jiang et al. BMC Medical Genetics (2020) 21:43 https://doi.org/10.1186/s12881-020-0981-x RESEARCH ARTICLE Open Access Molecular epidemiology and hematologic characterization of δβ-thalassemia and hereditary persistence of fetal hemoglobin in 125,661 families of greater Guangzhou area, the metropolis of southern China Fan Jiang1,2, Liandong Zuo2, Dongzhi Li2, Jian Li2, Xuewei Tang2, Guilan Chen2, Jianying Zhou2, Hang Lu2 and Can Liao1,2* Abstract Background: Individuals with δβ-thalassemia/HPFH and β-thalassemia usually present with intermedia or thalassemia major. No large-scale survey on HPFH/δβ-thalassemia in southern China has been reported to date. The purpose of this study was to examine the molecular epidemiology and hematologic characteristics of these disorders in Guangzhou, the largest city in Southern China, to offer advice for thalassemia screening programs and genetic counseling. Methods: A total of 125,661 couples participated in pregestational thalassemia screening. 654 subjects with fetal hemoglobin (HbF) level ≥ 5% were selected for further investigation. Gap-PCR combined with Multiplex ligation dependent probe amplification (MLPA) was used to screen for β-globin gene cluster deletions. Gene sequencing for the promoter region of HBG1 /HBG2 gene was performed for all those subjects. Results: A total of 654 individuals had hemoglobin (HbF) levels≥5, and 0.12% of the couples were found to be heterozygous for HPFH/δβ-thalassemia, including Chinese Gγ (Aγδβ)0-thal, Southeast Asia HPFH (SEA-HPFH), Taiwanese deletion and Hb Lepore–Boston–Washington. The highest prevalence was observed in the Huadu district and the lowest in the Nansha district. Three cases were identified as carrying β-globin gene cluster deletions, which had not been previously reported. -

Tigersharc Embedded Processor ADSP-TS101S
TigerSHARC Embedded Processor ADSP-TS101S FEATURES BENEFITS 300 MHz, 3.3 ns instruction cycle rate Provides high performance Static Superscalar DSP opera- 6M bits of internal—on-chip—SRAM memory tions, optimized for telecommunications infrastructure 19 mm × 19 mm (484-ball) CSP-BGA or 27 mm × 27 mm and other large, demanding multiprocessor DSP (625-ball) PBGA package applications Dual computation blocks—each containing an ALU, a multi- Performs exceptionally well on DSP algorithm and I/O bench- plier, a shifter, and a register file marks (see benchmarks in Table 1 and Table 2) Dual integer ALUs, providing data addressing and pointer Supports low overhead DMA transfers between internal manipulation memory, external memory, memory-mapped peripherals, Integrated I/O includes 14-channel DMA controller, external link ports, other DSPs (multiprocessor), and host port, 4 link ports, SDRAM controller, programmable flag processors pins, 2 timers, and timer expired pin for system integration Eases DSP programming through extremely flexible instruc- 1149.1 IEEE compliant JTAG test access port for on-chip tion set and high-level language-friendly DSP architecture emulation Enables scalable multiprocessing systems with low commu- On-chip arbitration for glueless multiprocessing with up to nications overhead 8 TigerSHARC processors on a bus COMPUTATIONAL BLOCKS PROGRAM SEQUENCER DATA ADDRESS GENERATION INTERNAL MEMORY 6 MEMORY MEMORY MEMORY JTAG PORT SHIFTER PC BTB IRQ INTEGER 32 32 INTEGER M0 M1 M2 JALU KALU ADDR 64K × 32 64K × 32 64K × 32 SDRAM CONTROLLER -

Normal, C=Critical
Patient Report |FINAL Client: Example Client ABC123 Patient: Patient, Example 123 Test Drive Salt Lake City, UT 84108 DOB 7/22/1970 UNITED STATES Gender: Female Patient Identifiers: 01234567890ABCD, 012345 Physician: Doctor, Example Visit Number (FIN): 01234567890ABCD Collection Date: 00/00/0000 00:00 Hemoglobin Evaluation Reflexive Cascade ARUP test code 2005792 Hemoglobin A 97.1 % (Ref Interval: 95.0-97.9) Hemoglobin A2 2.6 % (Ref Interval: 2.0-3.5) Hemoglobin F 0.3 % (Ref Interval: 0.0-2.1) REFERENCE INTERVAL: Hemoglobin F Access complete set of age- and/or gender-specific reference intervals for this test in the ARUP Laboratory Test Directory (aruplab.com). Hemoglobin S 0.0 % (Ref Interval: 0.0-0.0) Hemoglobin C 0.0 % (Ref Interval: 0.0-0.0) Hemoglobin E 0.0 % (Ref Interval: 0.0-0.0) Hemoglobin - Other 0.0 % (Ref Interval: 0.0-0.0) Sickle Cell Solubility Not Performed Hemoglobin, Capillary Electrophoresis Performed Hemoglobin Evaluation See Note Beta Globin Full Gene Sequencing Not Applicable H=High, L=Low, *=Abnormal, C=Critical Patient: Patient, Example ARUP Accession: 21-048-400496 Patient Identifiers: 01234567890ABCD, 012345 Visit Number (FIN): 01234567890ABCD Page 1 of 4 | Printed: 3/11/2021 11:41:09 AM 4848 Patient Report |FINAL Beta Globin (HBB) Del/Dup Result Not Applicable Alpha Thalassemia HBA1 and HBA2 Seq Not Applicable Gamma Globin (HBG1 and HBG2) Sequencing Not Applicable Hemoglobin Cascade Interpretation See Note H=High, L=Low, *=Abnormal, C=Critical Patient: Patient, Example ARUP Accession: 21-048-400496 Patient Identifiers: 01234567890ABCD, 012345 Visit Number (FIN): 01234567890ABCD Page 2 of 4 | Printed: 3/11/2021 11:41:09 AM 4848 Patient Report |FINAL RESULT Normal hemoglobin evaluation. -

Apoptotic Cells Inflammasome Activity During the Uptake of Macrophage
Downloaded from http://www.jimmunol.org/ by guest on September 29, 2021 is online at: average * The Journal of Immunology , 26 of which you can access for free at: 2012; 188:5682-5693; Prepublished online 20 from submission to initial decision 4 weeks from acceptance to publication April 2012; doi: 10.4049/jimmunol.1103760 http://www.jimmunol.org/content/188/11/5682 Complement Protein C1q Directs Macrophage Polarization and Limits Inflammasome Activity during the Uptake of Apoptotic Cells Marie E. Benoit, Elizabeth V. Clarke, Pedro Morgado, Deborah A. Fraser and Andrea J. Tenner J Immunol cites 56 articles Submit online. Every submission reviewed by practicing scientists ? is published twice each month by Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts http://jimmunol.org/subscription http://www.jimmunol.org/content/suppl/2012/04/20/jimmunol.110376 0.DC1 This article http://www.jimmunol.org/content/188/11/5682.full#ref-list-1 Information about subscribing to The JI No Triage! Fast Publication! Rapid Reviews! 30 days* Why • • • Material References Permissions Email Alerts Subscription Supplementary The Journal of Immunology The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2012 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. This information is current as of September 29, 2021. The Journal of Immunology Complement Protein C1q Directs Macrophage Polarization and Limits Inflammasome Activity during the Uptake of Apoptotic Cells Marie E. -

Time Signal Stations 1By Michael A
122 Time Signal Stations 1By Michael A. Lombardi I occasionally talk to people who can’t believe that some radio stations exist solely to transmit accurate time. While they wouldn’t poke fun at the Weather Channel or even a radio station that plays nothing but Garth Brooks records (imagine that), people often make jokes about time signal stations. They’ll ask “Doesn’t the programming get a little boring?” or “How does the announcer stay awake?” There have even been parodies of time signal stations. A recent Internet spoof of WWV contained zingers like “we’ll be back with the time on WWV in just a minute, but first, here’s another minute”. An episode of the animated Power Puff Girls joined in the fun with a skit featuring a TV announcer named Sonny Dial who does promos for upcoming time announcements -- “Welcome to the Time Channel where we give you up-to- the-minute time, twenty-four hours a day. Up next, the current time!” Of course, after the laughter dies down, we all realize the importance of keeping accurate time. We live in the era of Internet FAQs [frequently asked questions], but the most frequently asked question in the real world is still “What time is it?” You might be surprised to learn that time signal stations have been answering this question for more than 100 years, making the transmission of time one of radio’s first applications, and still one of the most important. Today, you can buy inexpensive radio controlled clocks that never need to be set, and some of us wear them on our wrists. -

Genome Editing of HBG1/2 Promoter Leads to Robust Hbf Induction in Vivo While Editing of BCL11A Erythroid Enhancer Shows Erythroid Defect
Genome Editing of HBG1/2 Promoter Leads to Robust HbF Induction In Vivo While Editing of BCL11A Erythroid Enhancer Shows Erythroid Defect Kai-Hsin Chang, Minerva Sanchez, Jack Heath, Edouard de Dreuzy, Scott Haskett, Abigail Vogelaar, Kiran Gogi, Jen DaSilva, Tongyao Wang, Andrew Sadowski, Gregory Gotta, Jamaica Siwak, Ramya Viswanathan, Katherine Loveluck, Hoson Chao, Eric Tillotson, Aditi Chalishazar, Abhishek Dass, Frederick Ta, Emily Brennan, Diana Tabbaa, Eugenio Marco, John Zuris, Deepak Reyon, Meltem Isik, Ari Friedland, Terence Ta, Fred Harbinski, Georgia Giannoukos, Sandra Teixeira, Christopher Wilson, Charlie Albright, Haiyan Jiang Editas Medicine, Inc., Cambridge, MA 60th Annual Meeting and Exposition of American Society of Hematology December 2, 2018, San Diego, CA © 2018 Editas Medicine Overview Etiology of Sickle Cell Disease In Vivo Study Design to Evaluate Two Approaches to Increase Fetal Hemoglobin (HbF) Expression Effect of Downregulating BCL11A Expression by Targeting its Erythroid Enhancer Editing Cis-regulatory Elements in b-Globin Locus Conclusion © 2018 Editas Medicine Etiology of Sickle Cell Disease Amino Hemoglobin Low DNA Acid Tetramer O2 a a a a b-globin GLU G A G b b b b HbA a a a a a a a a s s s VAL b b b -globin G T G bs bs bs bs bs bs HbS HbS Fiber • Sickle cell disease (SCD) is caused by a single mutation E6V of the b-globin chain, leading to polymerization of hemoglobin (Hb) and formation of sickle hemoglobin (HbS) fibers when deoxygenated. • Symptoms include anemia, acute chest syndrome, pain crises, and an array of other complications. • Patients suffer significant morbidity and early mortality.