RNA Interference Screening Identifies a Novel Role for PCTK1/ CDK16 in Medulloblastoma with C-Myc Amplification
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Profiling Data
Compound Name DiscoveRx Gene Symbol Entrez Gene Percent Compound Symbol Control Concentration (nM) JNK-IN-8 AAK1 AAK1 69 1000 JNK-IN-8 ABL1(E255K)-phosphorylated ABL1 100 1000 JNK-IN-8 ABL1(F317I)-nonphosphorylated ABL1 87 1000 JNK-IN-8 ABL1(F317I)-phosphorylated ABL1 100 1000 JNK-IN-8 ABL1(F317L)-nonphosphorylated ABL1 65 1000 JNK-IN-8 ABL1(F317L)-phosphorylated ABL1 61 1000 JNK-IN-8 ABL1(H396P)-nonphosphorylated ABL1 42 1000 JNK-IN-8 ABL1(H396P)-phosphorylated ABL1 60 1000 JNK-IN-8 ABL1(M351T)-phosphorylated ABL1 81 1000 JNK-IN-8 ABL1(Q252H)-nonphosphorylated ABL1 100 1000 JNK-IN-8 ABL1(Q252H)-phosphorylated ABL1 56 1000 JNK-IN-8 ABL1(T315I)-nonphosphorylated ABL1 100 1000 JNK-IN-8 ABL1(T315I)-phosphorylated ABL1 92 1000 JNK-IN-8 ABL1(Y253F)-phosphorylated ABL1 71 1000 JNK-IN-8 ABL1-nonphosphorylated ABL1 97 1000 JNK-IN-8 ABL1-phosphorylated ABL1 100 1000 JNK-IN-8 ABL2 ABL2 97 1000 JNK-IN-8 ACVR1 ACVR1 100 1000 JNK-IN-8 ACVR1B ACVR1B 88 1000 JNK-IN-8 ACVR2A ACVR2A 100 1000 JNK-IN-8 ACVR2B ACVR2B 100 1000 JNK-IN-8 ACVRL1 ACVRL1 96 1000 JNK-IN-8 ADCK3 CABC1 100 1000 JNK-IN-8 ADCK4 ADCK4 93 1000 JNK-IN-8 AKT1 AKT1 100 1000 JNK-IN-8 AKT2 AKT2 100 1000 JNK-IN-8 AKT3 AKT3 100 1000 JNK-IN-8 ALK ALK 85 1000 JNK-IN-8 AMPK-alpha1 PRKAA1 100 1000 JNK-IN-8 AMPK-alpha2 PRKAA2 84 1000 JNK-IN-8 ANKK1 ANKK1 75 1000 JNK-IN-8 ARK5 NUAK1 100 1000 JNK-IN-8 ASK1 MAP3K5 100 1000 JNK-IN-8 ASK2 MAP3K6 93 1000 JNK-IN-8 AURKA AURKA 100 1000 JNK-IN-8 AURKA AURKA 84 1000 JNK-IN-8 AURKB AURKB 83 1000 JNK-IN-8 AURKB AURKB 96 1000 JNK-IN-8 AURKC AURKC 95 1000 JNK-IN-8 -

Human Kinases Info Page
Human Kinase Open Reading Frame Collecon Description: The Center for Cancer Systems Biology (Dana Farber Cancer Institute)- Broad Institute of Harvard and MIT Human Kinase ORF collection from Addgene consists of 559 distinct human kinases and kinase-related protein ORFs in pDONR-223 Gateway® Entry vectors. All clones are clonal isolates and have been end-read sequenced to confirm identity. Kinase ORFs were assembled from a number of sources; 56% were isolated as single cloned isolates from the ORFeome 5.1 collection (horfdb.dfci.harvard.edu); 31% were cloned from normal human tissue RNA (Ambion) by reverse transcription and subsequent PCR amplification adding Gateway® sequences; 11% were cloned into Entry vectors from templates provided by the Harvard Institute of Proteomics (HIP); 2% additional kinases were cloned into Entry vectors from templates obtained from collaborating laboratories. All ORFs are open (stop codons removed) except for 5 (MST1R, PTK7, JAK3, AXL, TIE1) which are closed (have stop codons). Detailed information can be found at: www.addgene.org/human_kinases Handling and Storage: Store glycerol stocks at -80oC and minimize freeze-thaw cycles. To access a plasmid, keep the plate on dry ice to prevent thawing. Using a sterile pipette tip, puncture the seal above an individual well and spread a portion of the glycerol stock onto an agar plate. To patch the hole, use sterile tape or a portion of a fresh aluminum seal. Note: These plasmid constructs are being distributed to non-profit institutions for the purpose of basic -

PRODUCTS and SERVICES Target List
PRODUCTS AND SERVICES Target list Kinase Products P.1-11 Kinase Products Biochemical Assays P.12 "QuickScout Screening Assist™ Kits" Kinase Protein Assay Kits P.13 "QuickScout Custom Profiling & Panel Profiling Series" Targets P.14 "QuickScout Custom Profiling Series" Preincubation Targets Cell-Based Assays P.15 NanoBRET™ TE Intracellular Kinase Cell-Based Assay Service Targets P.16 Tyrosine Kinase Ba/F3 Cell-Based Assay Service Targets P.17 Kinase HEK293 Cell-Based Assay Service ~ClariCELL™ ~ Targets P.18 Detection of Protein-Protein Interactions ~ProbeX™~ Stable Cell Lines Crystallization Services P.19 FastLane™ Structures ~Premium~ P.20-21 FastLane™ Structures ~Standard~ Kinase Products For details of products, please see "PRODUCTS AND SERVICES" on page 1~3. Tyrosine Kinases Note: Please contact us for availability or further information. Information may be changed without notice. Expression Protein Kinase Tag Carna Product Name Catalog No. Construct Sequence Accession Number Tag Location System HIS ABL(ABL1) 08-001 Full-length 2-1130 NP_005148.2 N-terminal His Insect (sf21) ABL(ABL1) BTN BTN-ABL(ABL1) 08-401-20N Full-length 2-1130 NP_005148.2 N-terminal DYKDDDDK Insect (sf21) ABL(ABL1) [E255K] HIS ABL(ABL1)[E255K] 08-094 Full-length 2-1130 NP_005148.2 N-terminal His Insect (sf21) HIS ABL(ABL1)[T315I] 08-093 Full-length 2-1130 NP_005148.2 N-terminal His Insect (sf21) ABL(ABL1) [T315I] BTN BTN-ABL(ABL1)[T315I] 08-493-20N Full-length 2-1130 NP_005148.2 N-terminal DYKDDDDK Insect (sf21) ACK(TNK2) GST ACK(TNK2) 08-196 Catalytic domain -

The Discovery of Novel Gsk3 Substrates and Their Role in the Brain
THE DISCOVERY OF NOVEL GSK3 SUBSTRATES AND THEIR ROLE IN THE BRAIN James Robinson A DISSERTATION IN FULFILMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY St. Vincent’s Clinical School Facility of Medicine The University of New South Wales May 2015 ORIGINALITY STATEMENT ‘I hereby declare that this submission is my own work and to the best of my knowledge it contains no materials previously published or written by another person, or substantial proportions of material which have been accepted for the award of any other degree or diploma at UNSW or any other educational institution, except where due acknowledgement is made in the thesis. Any contribution made to the research by others, with whom I have worked at UNSW or elsewhere, is explicitly acknowledged in the thesis. I also declare that the intellectual content of this thesis is the product of my own work, except to the extent that assistance from others in the project's design and conception or in style, presentation and linguistic expression is acknowledged.’ Signed ……………………………………………… Date ………………………………………………… ii |Page Abstract Bipolar Disorder (BD) is a debilitating disease that dramatically impairs people’s lives and severe cases can lead to exclusion from society and suicide. There are no clear genetic or environmental causes, and current treatments suffer from limiting side effects. Therefore, alternative intervention strategies are urgently required. Our approach is to determine mechanisms of action of current drug therapies, in the hope that they will lead to discovery of next generation therapeutic targets. Lithium has been the mainstay treatment for BD for over 50 years, although its mechanism of action is not yet clear. -

Cdks in Sarcoma: Mediators of Disease and Emerging Therapeutic Targets
International Journal of Molecular Sciences Review CDKs in Sarcoma: Mediators of Disease and Emerging Therapeutic Targets Jordan L Kohlmeyer 1,2, David J Gordon 3 , Munir R Tanas 4, Varun Monga 5 , Rebecca D Dodd 5 and Dawn E Quelle 1,2,4,* 1 Molecular Medicine Graduate Program, Carver College of Medicine, University of Iowa, Iowa City, IA 52242, USA; [email protected] 2 The Department of Neuroscience and Pharmacology, Carver College of Medicine, University of Iowa, 2-570 Bowen Science Bldg., Iowa City, IA 52242, USA 3 The Department of Pediatrics, Carver College of Medicine, University of Iowa, Iowa City, IA 52242, USA; [email protected] 4 The Department of Pathology, Carver College of Medicine, University of Iowa, Iowa City, IA 52242, USA; [email protected] 5 The Department of Internal Medicine, Carver College of Medicine, University of Iowa, Iowa City, IA 52242, USA; [email protected] (V.M.); [email protected] (R.D.D.) * Correspondence: [email protected]; Tel.: +319-353-5749; Fax: +319-335-8930 Received: 6 April 2020; Accepted: 22 April 2020; Published: 24 April 2020 Abstract: Sarcomas represent one of the most challenging tumor types to treat due to their diverse nature and our incomplete understanding of their underlying biology. Recent work suggests cyclin-dependent kinase (CDK) pathway activation is a powerful driver of sarcomagenesis. CDK proteins participate in numerous cellular processes required for normal cell function, but their dysregulation is a hallmark of many pathologies including cancer. The contributions and significance of aberrant CDK activity to sarcoma development, however, is only partly understood. -

Targeted MS Quantitation Assays for Signal Transduction Protein Pathways
Targeted MS quantitation assays for signal transduction protein pathways Paul Haney R&D Platform Manager Thermo Scientific Protein Research Products Rockford, IL Why Targeted Quantitative Proteomics via Mass Spec? . Measure protein abundance and protein isoforms (e.g. splice variants, PTMs) without the need for antibodies . Avoid immuno-based cross- reactivity during multiplexing. Validate relative quantitation data from discovery proteomic experiments 2 How Do You Select Peptides for Targeted MS Assays? List of Proteins Spectral library Discovery data repositories In silico prediction Pinpoint 1.2 Hypothesis Experimental List of target peptides and transitions 3 How Do You Select Peptides for Targeted MS Assays? List of Proteins Spectral library Discovery data repositories In silico prediction Pinpoint 1.2 Hypothesis Experimental Validation Assay results List of target peptides and transitions 4 Tools for Target Peptide Identification and Scheduling . Active-site probes for enzyme subclass enrichment . Rapid recombinant heavy protein expression using human cell-free extracts . Peptide retention time calibration mixture for chromatography QC and targeted method 100 80 60 acquisition scheduling Intensity 40 20 0 5 10 15 20 25 30 5 Tools for Target Peptide Identification and Scheduling . Active-site probes for enzyme subclass enrichment . Rapid recombinant heavy protein expression using human cell-free extracts • Stergachis, A. & MacCoss, M. (2011) Nature Methods (submitted) . Peptide retention time calibration mixture for 100 80 chromatography -

Supp Material.Pdf
A B Young mouse Old mouse skin skin p65 p65 levels: 1.0 1.0 1.0 1.3 Human kidney Medulla Human kidney Cortex Human muscle (Arm/Leg) Human fibroblasts Human muscle (Abdomen) Human brain NFKBIA p105 CHUK NFKB1 IL1R1 PTPLAD1 MYD88 p50 NKIRAS1 IL1A p105 levels: 1.0 1.3 1.3 1.1 TRAF6 p50 levels: 1.0 1.1 1.6 1.2 TLR8 MALT1 MAP3K7 IκBα TNFRSF10A STAT1 IκBα levels: 1.0 1.1 0.8 0.9 CARD10 IRAK1 CARD14 ACTIN TNFRSF10B TNFRSF1A IKBKB MAP3K1 TIAF1 TNFAIP3 MAP3K7IP1 FADD B pathway genes (Biocarta) κ NKIRAS2 TNF NF- TLR1 ZNF675 TBK1 TLR6 MAP3K14 RIPK1 TRADD IKBKG TLR4 TLR3 RELA Genes induced with age (P<0.05) Genes repressed with age (P<0.05) Supplementary Figure 1. Diverse mechanisms of increased NF-κB activity with age. (A) Western analysis of two independent young and old mouse skin samples shows that NF-κB subunits p65, p50, and p105 weakly increase with age, while IκBα protein expression weakly decreases with age. Quantification was performed with ImageJ (http://rsb.info.nih.gov/ij/). (B) Expression of 38 genes found in NF-κB signaling pathways (BioCarta Molecular Pathways: http://www.biocarta.com) were anlayzed in six human tissues. One-sided t-tests identified genes whose expression significantly changes with age (P<0.05) in the indicated tissue. Some genes showed consistent change in multiple tissues, while many genes showed tissue specific effects. A B Input IP α-ER -ER -ER -ER -ER ∆ SP SP SP SP SP + LacZ + NFKB1 -ER ∆ ∆ ∆ ∆ GFP GFP NFKB1 NFKB1 GFP GFP NFKB1 NFKB1 4-OHT: 4-OHT 0h - + - + - + - + NFKB1∆SP-ER p65 p50 WB 4-OHT 2h RelB cRel ∆SP NFKB1 -ER localization p52 C D -ER SP ∆ -SR α B κ I GFP ∆SP NFKB1 NFKB1 - -ER -ER + 4-OHT κ α SP SP GFP + TNF + Antibody GFP ER I B -SR ∆ ∆ -SR + 4-OHT NFKB1∆SP-ER α B TNF: κ - + + - + GFP NFKB1 NFKB1 I -p50 -p65 -cRel -p52 -RelB 4-OHT: ACTIN ---- α α α α α - - + + + +TNF -ER -ER + 4-OHT SP SP ∆ ∆ B responsive genes -SR κ α B- κ NFKB1 I GFP NFKB1 NF-κB : IRF 147 NF- NF-κB : CCANNAGRKGGC NF-κB : GATA4 AP1 : ZID AP2-α : POU1F1 : RREB1 Average exp. -

Preview of “Supplement Table S4.Xls”
Annotations for all recurrently lost regions cytoband 1p36.12 3p14.2 3q13.13 q value 0,01352 0,1513 0,085307 residual q value 0,01352 0,1513 0,085307 wide peak boundaries chr1:23457835-23714048 chr3:60396160-60637030 chr3:110456383-110657226 genes in wide peak E2F2 FHIT DPPA4 ID3 DPPA2 TCEA3 DDEFL1 TBC1D19 PI4K2B STIM2 GBA3 KCNIP4 DKFZp761B107 C4orf28 GPR125 FLJ45721 hsa-mir-218-1 CXCL3 GRSF1 HNRPD HTN1 HTN3 IBSP IGFBP7 IGJ IL8 CXCL10 KDR CXCL9 AFF1 MUC7 NKX6-1 PF4 PF4V1 PKD2 POLR2B PPEF2 PPAT PPBP PRKG2 MAPK10 PTPN13 REST CXCL6 CXCL11 CXCL5 SPINK2 SPP1 SRP72 STATH SULT1E1 UGT2B4 UGT2B7 UGT2B10 UGT2B15 UGT2B17 SPARCL1 VDP SLC4A4 HERC3 GENX-3414 CDKL2 TMPRSS11D ABCG2 ADAMTS3 CLOCK CEP135 G3BP2 HNRPDL ENAM FAM13A1 CXCL13 PAICS UGT2B11 HPSE NMU SMR3B NPFFR2 UGT2A1 CCNI hsa-mir-491 hsa-mir-31 4p15.32 4q13.1 4q13.1 (continued) 6q14.1 0,14888 0,17878 0,093693 0,14888 0,17878 0,093693 chr4:17969802-29966659 chr4:55756035-90966136 chr6:76830186-107898353 CCKAR AFM SEC31A AIM1 DHX15 AFP RUFY3 BCKDHB RBPSUH ALB WDFY3 PRDM1 SOD3 AMBN LPHN3 CCNC SLIT2 ANXA3 DKFZP564O0823 CGA SLC34A2 AREG RCHY1 CNR1 PPARGC1A ART3 ANKRD17 EPHA7 KIAA0746 BMP3 BRDG1 GABRR1 ANAPC4 BTC SMR3A GABRR2 SLA/LP CCNG2 ASAHL GRIK2 LGI2 SCARB2 COQ2 HTR1B TBC1D19 CDS1 SULT1B1 HTR1E PI4K2B CENPC1 TMPRSS11E IMPG1 STIM2 CSN1S1 MRPS18C ME1 GBA3 CSN2 COPS4 NT5E KCNIP4 CSN3 HSD17B11 PGM3 DKFZp761B107 DCK HERC5 POU3F2 C4orf28 DMP1 PLAC8 PREP GPR125 DSPP NUDT9 SIM1 FLJ45721 EPHA5 NUP54 ELOVL4 hsa-mir-218-1 EREG UGT2B28 MAP3K7 FGF5 ODAM TPBG GC HERC6 TTK GK2 SDAD1 RNGTT GNRHR UBE1L2 TBX18 -

Inhibition of Akt/Protein Kinase B Signaling by Naltrindole in Small Cell Lung Cancer Cells
[CANCER RESEARCH 64, 8723–8730, December 1, 2004] Inhibition of Akt/Protein Kinase B Signaling by Naltrindole in Small Cell Lung Cancer Cells Yulong L. Chen, P. Y. Law, and Horace H. Loh Department of Pharmacology, the University of Minnesota Medical School, Minneapolis, Minnesota ABSTRACT tors, leading to the activation of Akt/PKB signaling (8). Akt/PKB is the cellular homologue of the product of the v-akt oncogene (9) and The phosphatidylinositol 3-kinase-Akt/protein kinase B (PKB) survival has three isoforms: Akt-1, -2, and –3 (or PKB-␣,-, and –␥; ref. 10). signaling is very important for cancer cell survival and growth. Consti- Akt/PKB mediates a variety of biological functions, including glucose tutively active phosphatidylinositol 3-kinase-Akt/PKB signaling in small cell lung cancer (SCLC) is a major factor for the survival of SCLC cells. uptake, protein synthesis, and inhibition of apoptosis (8). Akt/PKB Inhibitors of this signaling pathway would be potential antitumor agents, can mediate cell survival and growth by regulating both post-transla- particularly for SCLC. Here we report that naltrindole, which has been tional mechanisms and gene transcription (11). The Akt/PKB kinase used as a classic ␦ opioid antagonist, inhibited growth and induced activity is regulated by phosphorylation on two regulatory sites, apoptosis in the three characteristic SCLC cell lines, NCI-H69, NCI-H345, threonine 308 in the activation loop of the catalytic domain and serine and NCI-H510. Naltrindole treatment reduced constitutive phosphoryla- 473 in the COOH-terminal regulatory domain (12). One of the Akt/ tion of Akt/PKB on serine 473 and threonine 308 in cells. -

Human CDK16 / PCTK1 (158-496, His-Tag) - Purified
OriGene Technologies, Inc. OriGene Technologies GmbH 9620 Medical Center Drive, Ste 200 Schillerstr. 5 Rockville, MD 20850 32052 Herford UNITED STATES GERMANY Phone: +1-888-267-4436 Phone: +49-5221-34606-0 Fax: +1-301-340-8606 Fax: +49-5221-34606-11 [email protected] [email protected] AR51243PU-N Human CDK16 / PCTK1 (158-496, His-tag) - Purified Alternate names: Cell division protein kinase 16, Cyclin-dependent kinase 16, PCTAIRE 1, PCTAIRE-1, PCTAIRE-motif protein kinase 1, PCTAIRE1, Serine/threonine-protein kinase PCTAIRE-1 Quantity: 0.5 mg Concentration: 1.0 mg/ml (determined by Bradford assay) Background: Cyclin-dependent kinase 16 isoform 1, also known as CDK16, belong to the CDK family of serine/threonine protein kinases. These proteins have a core kinase domain flanked by unique amino- and carboxy-terminal domains. CDK proteins are known to regulate the cell cycle. CDK16, which is expressed primarily in mammalian brain, interacts with a variety of proteins, and is thought to be part of a multiple signal transduction cascade. Uniprot ID: Q00536 NCBI: NP_006192 GeneID: 5127 Species: Human Source: E. coli Format: State: Liquid purified protein Purity: >95% by SDS - PAGE Buffer System: 20 mM Tris-HCl buffer (pH 8.0) containing 0.1M NaCl, 10% glycerol Description: Recombinant human CDK16 protein, fused to His-tag at N-terminus, was expressed in E.coli and purified by using conventional chromatography techniques. AA Sequence: MGSSHHHHHH SSGLVPRGSH MGSGFGKLET YIKLDKLGEG TYATVYKGKS KLTDNLVALK EIRLEHEEGA PCTAIREVSL LKDLKHANIV TLHDIIHTEK SLTLVFEYLD KDLKQYLDDC GNIINMHNVK LFLFQLLRGL AYCHRQKVLH RDLKPQNLLI NERGELKLAD FGLARAKSIP TKTYSNEVVT LWYRPPDILL GSTDYSTQID MWGVGCIFYE MATGRPLFPG STVEEQLHFI FRILGTPTEE TWPGILSNEE FKTYNYPKYR AEALLSHAPR LDSDGADLLT KLLQFEGRNR ISAEDAMKHP FFLSLGERIH KLPDTTSIFA LKEIQLQKEA SLRSSSMPDS GRPAFRVVDT EF Molecular weight: 41.1 kDa (362aa) confirmed by MALDI-TOF Storage: Store undiluted at 2-8°C for one week or (in aliquots) at -20°C to -80°C for longer. -

Stimulation of the Insulin/Mtor Pathway Delays Cone Death in a Mouse Model of Retinitis Pigmentosa
ARTICLES Stimulation of the insulin/mTOR pathway delays cone death in a mouse model of retinitis pigmentosa Claudio Punzo1, Karl Kornacker2 & Constance L Cepko1,3 Retinitis pigmentosa is an incurable retinal disease that leads to blindness. One puzzling aspect concerns the progression of the disease. Although most mutations that cause retinitis pigmentosa are in rod photoreceptor–specific genes, cone photoreceptors also die as a result of such mutations. To understand the mechanism of non-autonomous cone death, we analyzed four mouse models harboring mutations in rod-specific genes. We found changes in the insulin/mammalian target of rapamycin pathway that coincided with the activation of autophagy during the period of cone death. We increased or decreased the insulin level and measured the survival of cones in one of the models. Mice that were treated systemically with insulin had prolonged cone survival, whereas depletion of endogenous insulin had the opposite effect. These data suggest that the non-autonomous cone death in retinitis pigmentosa could, at least in part, be a result of the starvation of cones. Retinitis pigmentosa is a type of inherited retinal degeneration. It To determine the common underlying mechanism for cone death in is currently untreatable and usually leads to blindness. With over retinitis pigmentosa, we compared four mouse models harboring 40 reintitis pigmentosa genes identified, it is the most common type mutations in rod-specific genes (Pde6b–/– (ref. 11), Pde6g–/– (ref. 12), of retinal degeneration caused by a single disease allele (RetNet, Rho–/– (ref. 13) and P23H14, which carries a Rho transgene that has an http://www.sph.uth.tmc.edu/Retnet/). -
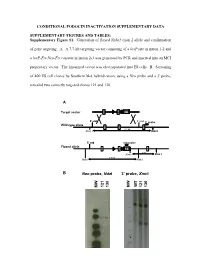
Supplementary Figure S1. Generation of Floxed Nphs2 Exon 2 Allele and Confirmation
CONDITIONAL PODOCIN INACTIVATION SUPPLEMENTARY DATA SUPPLEMENTARY FIGURES AND TABLES: Supplementary Figure S1. Generation of floxed Nphs2 exon 2 allele and confirmation of gene targeting. A. A 7.7-kb targeting vector consisting of a loxP site in intron 1-2 and a loxP-Frt-Neo-Frt cassette in intron 2-3 was generated by PCR and inserted into an MCI proprietary vector. The linearized vector was electroporated into ES cells. B. Screening of 400 ES cell clones by Southern blot hybridization, using a Neo probe and a 3' probe, revealed two correctly targeted clones 121 and 130. A Target vector 1 2 NEO 5’ end 3’ end 3’ probe Wild-type allele 1 2 3 9.2 kb Xmn I Xmn I 5’ end neo probe Floxed allele 1 2 NEO 3 5 kb Xmn I Xmn I 7.8 kb Nde I Nde I B Neo probe, NdeI 3’ probe, XmnI WT 121 130 121 130 MW MW - 1 - Supplementary Figure S2. Generation of Nphs2lox2/-,Cre+ mice and excision of exon 2 upon Cre recombinase induction. A. Triallelic Nphs2lox2/-,Cre+ mice were obtained by mating phenotypically normal Nphs2lox2/lox2 mice with Nphs2+/-,Cre+ mice. Mendelian inheritance of these alleles was observed. B. Genotypes were verified by multiplex PCR of tail genomic DNA. C. Cre recombinase activity was induced upon tamoxifen administration, leading to excision of the floxed exon 2 of the Nphs2 gene. D. Cre recombinase activity in the kidney was verified by PCR using a set of forward and reverse primers designed around exon 2 and demonstrating a 692-bp product (before Cre) and a 316-bp product (after Cre) using genomic DNA extracted from the renal cortex.